Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.62 no.2 San José abr./jun. 2014
Intra-specific variability in life-history traits of Anadara tuberculosa (Mollusca: Bivalvia) in the mangrove ecosystem of the Southern coast of Ecuador
Variaciones en la historia de Anadara tuberculosa (Mollusca: Bivalvia) en el ecosistema de manglar de la costa Sur de Ecuador
Variaciones en la historia de Anadara tuberculosa (Mollusca: Bivalvia) en el ecosistema de manglar de la costa Sur de Ecuador
*Dirección para correspondencia:
Abstract
Anadara tuberculosa is one of the most important bivalves along the Western Pacific coast because of its commercial value. Nevertheless, the variability in growth, longlife span, natural mortality and reproductive parameters of this mangrove cockle has not yet been described. The aim of this study was to analyze these lifehistory traits in three areas of the Southern coast of Ecuador. Empirical and length-based methods were used to estimate these biological parameters. Body size data were collected from the commercial fishery between 2004 and 2011 in landing ports near to the Archipelago of Jambeli [Puerto Bolivar (PB), Puerto Jeli (PJ) and Puerto Hualtaco (PH)]. The von Bertalanffy growth parameters for combined sex were estimated between 70.87 to 93.45mm for L∞ and 0.22 to 0.80/year for k. The growth indices (Φ’) ranged from 3.17 to 3.85, while the overall growth performance (OGP) ranged from 5.03 to 5.82. The mean of long-life span (tmax), size and age at maturity (L50% and t50%) were estimated in 7.71±2.53years, 39.13±2.24mm and 1.46±0.56years for PB; 9.51±2.85years, 37.78±1.95mm and 1.37±0.41years for PJ and 5.81±2.11years, 39.73±3.31mm and 0.94±0.41years for PH. Natural mortality (M) ranged from 0.46 to 1.28/year. We concluded that significant intra-specific variation was observed in a temporal scale in Φ’ and OGP indices as well as L50% and M. Therefore, temporal changes in these life-history traits should be taken into account when assessing the status of the mangrove cockle fishery.
Key words: Anadara tuberculosa, growth, life span, size/age of maturity, Archipelago of Jambeli, Ecuador
Resumen
La pesquería de la concha prieta (Anadara tuberculosa) es una de las más importantes a lo largo de la costa del Pacifico. Las variaciones intra-especificas en el crecimiento, longevidad, mortalidad natural y parámetros reproductivos de la concha prieta, aún no han sido descritas. Por lo tanto, el objetivo de este estudio fue analizar esos parámetros biológicos en tres áreas al Sur de Ecuador. Métodos empíricos y basados en tallas fueron empleados para estimar los parámetros de historia de vida. Los datos de tallas analizados correspondieron al periodo 2004-2011 y son provenientes del muestreo de los desembarques de los puertos adyacentes al Archipiélago de Jambeli [Puerto Bolivar (PB), Puerto Jeli (PJ) y Puerto Hualtaco (PH)], al Sur de Ecuador. Los parámetros de crecimiento oscilaron entre 70.87 y 93.45mm de L∞ y 0.22 a 0.80/año de k. Mientras que los índices de crecimiento entre 3.17 a 3.85 de Φ’ y 5.03 a 5.82 de OGP. Los valores promedio de longevidad (tmax), talla y edad de madurez (L50% y t50%) fueron de 7.71±2.53años, 39.13±2.24mm y 1.46±0.56años en PB; 9.51±2.85años, 37.78±1.95mm y 1.37±0.41años en PJ y de 5.81±2.11años, 39.73±3.31mm y 0.94±0.41años en PH. Las estimaciones de mortalidad natural (M) oscilaron entre 0.46 y 1.28/año. Importantes cambios intra -especificos se observaron a escala temporal tanto en los índices Φ’ y OGP, así como en L50% y M. Por lo tanto, se sugiere considerar las variaciones temporales en los parámetros biológicos de A. tuberculosa, al momento de caracterizar el estatus de su pesquería y más aun si es sometida a altos niveles de extracción.
Palabras clave: Anadara tuberculosa, parámetros biológicos, Archipiélago de Jambeli, Ecuador.
The mangrove cockle Anadara tuberculosa (Sowerby, 1833) is the most representative species of the genus Anadara along the Western Pacific coast. Its distribution ranges from Mexico to Peru (MacKenzie, 2001). The common name of this species varies geographically. For example, in Ecuador it is called “concha prieta”, “concha negra” or “concha hembra”; but, in other locations such as Colombia and Costa Rica it is known as “piangua”.
A. tuberculosa lives mainly in or near the red mangrove tree roots (Rhizophora mangle). In natural population, A. tuberculosa is generally more abundant than A. similis (C.B. Adams, 1852) (Silva & Bonilla, 2001), and in the mangrove cockle fishery of Ecuador, the catch composition of these two species is dominated by A. tuberculosa. However, this proportion differs between landing ports (Mora, Moreno & Jurado, 2011; Flores & Morales, 2011; Mora, Flores, Moreno & Gilbert, 2012). Recently, Mora et al. (2011, 2012) have observed species proportions of 5:1 to 13:1 and 9:1 to 11:1 for A. tuberculosa/A. similis in the Archipelago of Jambeli, Ecuador. It is possible that a similar catch composition could be observed in the mangrove cockle fishery of the other countries.
Flores and Mora (2011) identified that the reproductive biology of A. tuberculosa has been the main topics of study [e.g. gonadal development stages, sex ratio] for this species. But they emphasized the lack of information regarding reproductive parameters such as size at maturity (L50%). Moreover, some life-history traits and the population dynamics of this species are still understudied along its geographic distribution, despite its commercial importance. The state of knowledge about A. tuberculosa life-history parameters is currently restricted to the reported by Flores (2011), and Flores and Mora (2011) for Ecuador, Felix-Pico et al. (2009) for Mexico and Gil-Agudelo et al. (2010) and Lucero et al. (2012) for Colombia.
Little attention has been paid to the analysis of the variability of the life-history traits of A. tuberculosa. The goal of this study was to characterize the intra-specific variability of growth, life span, natural mortality and reproductive parameters of A. tuberculosa on the Southern coast of Ecuador.
Materials and Methods
Data sources: Size data [Shell length (SL), mm] for the mangrove cockle, A. tuberculosa, were collected monthly based on a fishery-dependent sampling in landing ports closed to the Archipelago of Jambeli [Puerto Bolivar (PB) 3º15’40’’ S - 79º59’48’’ W, Puerto Jeli (PJ) 3º27’00’’ S - 80º03’06’’ W and Puerto Hualtaco (PH) 3º25’05’’ S - 80º05’0’’ W], Ecuador. SL for all mangrove cockles was measured along the anteriorposterior axis with a digital caliper and a precision of 0.05mm. The data corresponded to the time periods 2004-2005 and 2008-2011. The total number of mangrove cockle measured was 25 987 cockles in PB, 17 620 cockles in PJ and 24 164 cockles in PH. This information was used to estimate life history parameters based on length-methods and empirical relationships as an ad hoc approach.
Growth: The von Bertalanffy growth function (VBGF) was fitted to length-frequency distribution for each year using the NSLCA routine of the FISAT II (Gayanilo, Sparre & Pauly, 2005) according to the approach followed by Flores (2011) and Flores and Mora (2011) with some modifications. The response surface analysis was used to identify the parameters of the VBGF that best fitted the data. The initial values for asymptotic length [L∞ (mm)] were established considering the empirical relationship given by Pauly [1980 (+ 5% of maximum length, Lmax)]. First, Lmax and its 95% confidence interval were estimated based on Maximum Length Estimation (MLE) routine of FISAT II and then was added the 5% to this values. In contrast, the initial values for the Brody growth coefficient [k (mm/year)] were fixed to the range reported in literature (i.e., 0.20 to 0.80). The theoretical age at zero length [t0] parameter was estimated according to Pauly (1980).
The growth index phi prime (Φ’) was used to compare the growth performance due to the negative correlation between k and L∞ invalidates its individual comparison (Pauly & Munro, 1984). Additionally, the overall growth performance index (OGP) was calculated to the intra-specific comparison between the three mentioned regions and years. The OGP represents the growth rate at the point of inflexion of the size growth curve (Pauly, 1979). Jackknife technique was used to quantify descriptive estimators [mean, standard error and 95% confidence intervals] of the growth parameters and growth indices as well as the other life-history traits (Haddon, 2001).
Life span: The theoretical life span [tmax] was calculated by the inverse VBGF (Taylor, 1958) assuming t = 0 (because the larval period of this species is unknown):
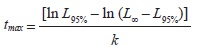
where L95% represents for this study the 95% of Lmax estimated in FISAT II and k and L∞ are as described above.
Reproductive parameters: The size at maturity (L50%) was calculated based on linear empirical relationships between the parameters L50% and L∞ reported in publications for Anadara genus. This approach is justified because these two parameters are positively correlated (Stamps, Mangel & Phillips, 1998) and there are not suitable data to estimate them directly. Similarly, the age at maturity (t50%) was obtained replacing the L50% in the VBGF and solved for t:
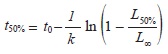
Natural mortality: Natural mortality rate (M) was calculated based on the empirical model proposed by Brey and Cage (1997) to benthic invertebrate populations according to:
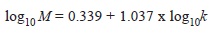
where M is the instantaneous rate of natural mortality and k is the Brody growth coefficient. This log-log equation had a significant coefficient of determination (r2=0.695; n=83). This empirical equation was used because it is the only equation available for estimating M in benthic invertebrates. The variance of M was estimated using a Monte Carlo method (Cubillos, Alarcon & Brante, 1999; Cubillos & Araya, 2007). This method gives alternative and equally probable values of M from a parametric sample of k according the expression:
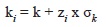
where ki are the alternative and equally probable values of k, zi is a random number with a standard normal distribution (mean=0, variance=1), and
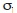 k is the standard deviation of k. A total of 200 simulations (Rmax) were performed to generate the values of k, which were then entered into the equation of Brey and Cage (1997). It allowed for comparing the estimations of M between years and zones.
k is the standard deviation of k. A total of 200 simulations (Rmax) were performed to generate the values of k, which were then entered into the equation of Brey and Cage (1997). It allowed for comparing the estimations of M between years and zones. Results
Growth: The average of the maximum shell length (Lmax) estimated in MLE routine of FISAT II was 78.79±5.27mm for PB, 77.75±5.04mm for PJ and 79.47±3.92mm for PH. This result did not differ from the average estimated for the data observed in each landing port. Estimates of VBGF growth parameters (L∞ and k) were different both in each year and landing port (Table 1). The highest k and growth indices values were observed during 2008 (Table 1).The growth analysis based on Φ’ and OGP, revealed a clear evidence of intra-specific variations among years (Table 2). Moreover, the analysis by time period showed an increment in growth rates between 2004-2005 and 2008-2011. This result was further confirmed when the length at age for one-year old individuals was analyzed by period. The individuals with the largest increase in length were those collected in PH during 2008 (Fig. 1).
Life span: The size for which the 95% of the Lmax differed by port. For instance, in PB the values of Lmax ranged from 64.32 to 82.61mm; while in PJ between 61.63 to 79.91mm, and in PH between 64.70 to 81.90mm. The mean tmax for the period 2004-2011 was 7.71±2.53years, 9.51±2.85years and 5.81±2.11years for the mentioned landing ports respectively. These mean values were different between landing ports but not statistically (ANOVA, p=0.15). Intra-specific variations were only observed for some years, especially in PB (ANOVA, p<0.0001).
Reproductive parameters: A positive and significant relationship was found between L50% and L∞ (Fig. 2), and the model explained 48% of the response variable. Different estimates of L50% and t50% were observed at each site (Table 3). However, neither L50% nor t50% were statistically different between landing’s port (ANOVA, p>0.05). A multiple comparison analysis revealed statistical differences between most of the years in L50% for each landing port (TUKEY, p<0.0001). In contrast, no differences were found in t50% among years (ANOVA, p>0.05). The average of L50% and t50% for the period 2004-2011 were 39.13±2.24mm and 1.46±0.56years for PB, 37.78±1.95mm and 1.37±0.41years for PJ and 39.73±3.31mm and 0.94±0.41years for PH, respectively.
Natural mortality: The estimates of M and its standard error are summarized in table 4. The calculated values ranged between 0.5 and 1.28/year. Hualtaco showed the highest values of M, whereas PB the lowest ones. The M values tended to increase in PJ from 2004 to 2010, but decreased sharply in 2011.
Discussion
In this study, clear differences in VBGF parameters were found across sites, but the range of the values observed were similar to those reported recently by Gil-Agudelo et al. (2010), Flores (2011), Flores and Mora (2011) and Lucero et al. (2012) in the Southeastern Pacific zone, specifically for the k parameter. The highest values of k were estimated in 2008, but this value seems to be overestimated as a consequence of convergence problems in the estimation process. The high values of k estimated in 2008 may also be due to the fact that the data set was collected mostly during the second semester of that year. Borda and Cruz (2004a) emphasized that the length-based methods are sensitive to the range of sizes used in the analysis. However, there is no evidence yet of effects of a truncated range of size on k estimates in A. tuberculosa using length-based methods. The k estimates for other years did not show evidence of bias, which can be attributed to relatively large sample size used.
In general, the k estimated values of this study and those reported for the Southeastern Pacific zone are lower than those estimated by Borda and Cruz (2004a) and Felix-Pico et al. (2009). Flores (2011) suggested that the differences found between these studies were a consequence of the shell length interval (SLI) and the length-based method used. To improve these estimations, Flores (2011) has recommended using the NSCLA algorithm with a jackknife technique and a SLI of 1.5 or 2.0mm. Most of the studies indicate that A. tuberculosa, has a low growth rate, which has been confirmed in experimental studies such as Villalobos and Baez (1983), Ortiz and Orellana (1997), Samaniego (2008) and Flores (2010). These analyses showed that this species, reaches its highest growth rate during the first two years of its life span. The same pattern was observed in this study. In PB, for example, the growth rate during the first two years ranged from 31 to 54mm; after that, the growth rate decreased.
At present, the most common methods used to assess inter and intra-specific growth variation are the growth rates [k/(y)] at which the asymptotic size is reached and the size at one year of the age (Francis, 1996; Wang & Milton, 2000). Nevertheless, Munro and Pauly (1983) demonstrated that growth indices can be also a suitable way to compare intra-specific growth. In this study, both growth indices and growth rates of year-old individuals showed evidences of mangrove cockle intraspecific variation, which were more significant in PB and PH. This is the first report where growth variability between areas is analyzed, but it is suggested to consider that the scale of this study analysis was the landing port. Growth variability is a well-known phenomenon in sedentary species because growth can change even on small-spatial scale (Caddy & Defeo, 2003). For example, Gil-Agudelo et al. (2010) found that growth parameters in A. tuberculosa differed across different sites. Thus, further investigations are needed to assess the effect of different spatial scales on growth using age data.
Bivalves are the most long-lived of molluscs and they are recognized to be a model for natural aging (Heller, 1990). Bivalve species exhibit a range of life span ranging from 1-2 years to hundreds of years (Philipp & Abele, 2010). This study used empirical and length-based methods to estimate longevity in A. tuberculosa. The mean longevity estimated here let us to conclude that the species has an intermediate long life span and with a considerable intra-specific variability. For instance, the range estimated was from 4 to 13 years old (without considering the growth estimate of 2008). This life history trait of A. tuberculosa is still understudied across its geographic distribution and this study represents its first formal estimates. In general, the estimated maximum values are close to those estimated for Baqueiro-Cárdenas and Aldana-Aranda [2003 (12 years)], Flores and Mora [2011 (10-11 years)] and Lucero et al. [2012 (12 years)].
In contrast to our estimations, even longer life span have been reported for A. tuberculosa in others areas, according to the estimate produce for Madrigal [1980 (20 years, with growth parameter estimated in Stern -Pirlot & Wolff, 2006)], Villalobos and Baez [1980 (33 years, with growth parameter estimated in Stern-Pirlot & Wolff, 2006)], Stern-Pirlot and Wolff [2006 (21 years)] and Flores [2011 (24-27 years)]. According to Miller [2004, cited in Stern-Pirlot & Wolff, 2006], it is common to find bivalves with 20 to 30 year life spans, and even more in the genus Anadara [e.g. A. broughtoni: 46 years (Zolotarev, 1980; cited in Gabaev & Olifirenko, 2001), 36 years (Zoaiko, 1981; cited in Gabaev & Olifirenko, 2001), 53 years (Gabaev & Olifirenko, 2001)].
At present, considerable uncertainty exists on maximal longevity of the genus Anadara. Thus, it is advisable to carry out further research in sclerochronology to explore the factors that could explain this variability.
In the Southeastern Pacific area, A. tuberculosa is characterized to have a reproductive cycle that extends throughout the year, but is variable from year to year. Based on biological data for the period of this study, it has been hypothesized that the reproductive season [the highest frequency percentage of mature and spawning stages] occurs between November and February in the Southern coast of Ecuador (Ayala, unpublished data). However, this should be verified by the use of histological techniques because of the uncertainty in the assignation of gonad stages. Given that an estimate of reproductive parameters for the coast of Ecuador has not yet been performed yet, the use of an empirical approach can be useful to construct proxies for L50% and t50%. In this study was also identified a positive and significative relationship such as Flores and Mora (2011), but with a lower percentage of explanation of the response variable.
Estimates of L50% were between 33 and 45mm. These are within the range reported for this species along its geographic distribution in the Pacific coast. In Mexico Perez-Medina (2005) reported a L50% of 36.50mm. Ampie and Cruz (1989) estimated values from 23 to 26mm in Costa Rica. Guilbert (2007) reported 32mm for Panama. In Colombia, many different values of L50% have been estimated. Squires et al. (1975) estimated 32mm, Borda and Cruz (2004b) 44mm, Gil-Agudelo et al. (2010) between 41 to 47mm, and lately, Lucero et al. (2012) 39.50mm. Estimates for L50% in Ecuador range between 37 to 42mm (Flores & Mora, 2011), whereas 41mm in Peru (Ishiyama & Terukina, 1989).
However, considering the overall period 2004-2011, the estimation of L50% were below the values reported by Gil-Agudelo et al. (2010) for the Colombian Pacific and slightly close to that reported previously by Flores and Mora (2011) for the Archipelago of Jambeli. In this context, the differences found across years and between each port support the evidence of important intra-specific variation for L50% in A. tuberculosa. On the other hand, the estimate of t50% represents the first formal report for this species along its geographical distribution. Based on the findings in this study, A. tuberculosa on the Southern coast of Ecuador reaches its age of maturity in after the first year of life and this parameter is less variable temporally.
As expected, natural mortality followed the variations in the growth coefficient, and therefore was consistent with longevity and L50%. Bases on the comparative analysis, we propose that cohorts of A. tuberculosa in Hualtaco have been characterized for a growth rate and natural mortality higher than that in the other ports. Estimates of M in this study were within the range of the values previously reported for the Southeastern pacific region.
Borda and Cruz (2004a) estimated values between 0.23 to 1.38/year using three different empirical models [Taylor, Tanaka and Rikhter & Efanov]. Vivar [1996; cited in Borda & Cruz, 2004a] and Gil-Agudelo et al. (2010) estimated values of 1.18/year and between 0.68 to 1.07/ year, using the Pauly’s method, respectively. However, these estimates were based on empirical models developed for fish. In contrast, the estimates in this study were based on models developed for invertebrates. This may reduce the uncertainty related to this demographic trait. However, because this is an important population parameter, it is recommended to perform studies using a more direct method.
Acknowledgments
The database used in this study is part of the information collected for the monitoring program of the mangrove cockle fishery in Ecuador. This program is supported by the National Fisheries Institute of Ecuador [Instituto Nacional de Pesca (INP)]. LF and EM thank the INP for the use of information. Also, many thanks to Juan Moreno, Fedra Solano, Viviana Jurado and Fernando Aguilar for their collaboration in the data collection. Our sincere gratitude to Yahira Piedrahita for her support to the mangrove cockle research program while she was Director of INP. Thanks to Natalia (Tala) Woodward (UW) and Nikita Gaibor for correcting and editing the English version and also to the referees for their comments to improve this manuscript.
References
Ampie, C. L. & Cruz, R. A. (1989). Tamaño y madurez sexual de Anadara tuberculosa (Bivalvia: Arcidae) en Costa Rica. Brenesia, 31, 21-24. [ Links ]
Baqueiro-Cárdenas, E. & Aldana-Aranda, D. (2003). Patrones en la biología poblacional de moluscos de importancia comercial en México. Revista de Biología Tropical, 51(4), 97-107. [ Links ]
Borda, C. A. & Cruz, R. (2004a). Crecimiento y tasa de mortalidad del bivalvo Anadara tuberculosa (Sowerby, 1833) en el Pacifico Colombiano. Revista de Investigaciones Marinas, 25, 177-184. [ Links ]
Borda, C. A. & Cruz, R. (2004b). Reproducción y reclutamiento del molusco Anadara tuberculosa (Sowerby, 1833) en el Pacifico Colombiano. Revista de Investigaciones Marinas, 25, 185-195. [ Links ]
Brey, T. & Gage, J. D. (1997). Interactions of growth and mortality in benthic invertebrate populations: empirical evidence for a mortality-growth continuum. Archive of Fishery and Marine Research, 45(1), 45-59. [ Links ]
Caddy, J. F. & Defeo, O. (2003). Enhancing or restoring the productivity of natural population of shellfish and other marine invertebrate resources. FAO Fisheries Technical Paper, No. 448. Rome: FAO. [ Links ]
Cubillos, L., Alarcón, R., & Brante, A. (1999). Empirical estimates of natural mortality for the Chilean hake (Merluccius gayi): evaluation of precision. Fisheries Research, 42, 147-153. [ Links ]
Cubillos, L. & Araya, M. (2007). Estimaciones empíricas de mortalidad natural en el bacalao de profundidad (Dissostichus eleginoides Smitt, 1898). Revista de Biología Marina y Oceanografía, 42(3), 287-297. [ Links ]
Félix-Pico, E. F., Ramírez-Rodríguez, M., & Holguín-Quiñones, O. (2009). Growth and Fisheries of the Black Ark Anadara tuberculosa, a Bivalve Mollusk, in Bahía Magdalena, Baja California Sur, Mexico. North American Journal of Fisheries Management, 29, 231-236. [ Links ]
Flores, L. (2010). Tasa de crecimiento de Anadara tuberculosa (Sowerby, 1833) (Bivalvia: Arcidae) en la Reserva Ecológica Manglares Cayapas-Mataje (REMACAM): un análisis basado en sistemas de cajas suspendidas. Revista de Ciencias del Mar y Limnologia, 4, 89-98. [ Links ]
Flores, L. (2011). Growth estimation of mangrove cockle (Anadara tuberculosa): application and assessment of length based method. Revista de Biología Tropical, 59(1), 159-175. [ Links ]
Flores, L. & Mora, E. (2011). Evaluando variaciones en la talla de Anadara tuberculosa y Anadara similis en el Archipiélago of Jambelí: Hay indicios de sobrepesca? Revista de Ciencias del Mar y Limnologia, 5(1), 33-49. [ Links ]
Flores, L. & Morales, M. (2011). La explotación de Anadara tuberculosa y Anadara similis en Puerto El Morro: un análisis después de una década sin monitoreo. Boletin Científico y Técnico, 21(4), 24-35. [ Links ]
Francis, R. I. C. (1996). Do herring grow faster than orange roughy? Fisheries Bulletin, 94, 783-786. [ Links ]
Gabaev, D. D. & Olifirenko, A. B. (2001). Growth, stock and production of the Anadara scapharca broughtoni in Peter the Great Bay, Sea of Japan. Oceanology, 41(3), 403-412. [ Links ]
Gayanilo, F. C. Jr., Sparre, P., & Pauly, D. (2005). The FAO-ICLARM Stock Assessment Tools II (FISAT II). Revised version. User’s guide. FAO Computerized Information Series (Fisheries), No 8. Rome: FAO. [ Links ]
Guilbert, A. (2007). State of the Anadara tuberculosa (Bivalvia: Arcidae) fishery in las Perlas Archipielago, Panama. (Master of Science thesis). School of Life Science, HeriotWatt University, Edinburgh. [ Links ]
Gil-Agudelo, D. L., Gualteros, W., Delgado, M. F., Lucero, C. H., Espinosa, S., Cortés, N., Palacio, C. J., Roldán, A. M., Zapata, L. A., Candelo, C., Cantera, J. R., Mayor, G., & Muñoz, S. (2010). Potencial productivo de las poblaciones naturales de la piangua Anadara tuberculosa y Anadara similis dentro de una perspectiva espacio-temporal en la costa Pacífica colombiana. (Informe Final. INVEMAR-ASCONAR-WWF-Universidad del Valle-UAESP-NN). Cali: Universidad del Valle. [ Links ]
Haddon, M. (2001). Modelling and quantitative methods in fisheries. New York: Chapman and Hall/CRC. [ Links ]
Heller, J. (1990). Longevity in molluscs. Malacologia, 31(2), 259-295. [ Links ]
Ishiyama, V. & Terukina, S. (1989). Reproduccion de Anadara tuberculosa y Anadara similis en la zona de Puerto Pizarro. Revista Biota, 95, 2-11. [ Links ]
Lucero, C., Cantera, J., & Neira, R. (2012). Pesquería y crecimiento de la piangua (Arcoida: Arcidae) Anadara tuberculosa en la Bahía de Málaga del Pacifico colombiano, 2005-2007. Revista de Biología Tropical, 60(1), 203-217. [ Links ]
MacKenzie, C. L. Jr. (2001). The Fisheries for Mangrove Cockles, Anadara spp., from Mexico to Peru, with Descriptions of their Habitats and Biology, the Fishermen’s Lives, and the Effects of Shrimp Farming. Marines Fisheries Review, 63, 1-39. [ Links ]
Mora, E., Moreno, J., & Jurado, V. (2011). Un análisis sobre la pesquería de la concha prieta en Ecuador, año 2010. Boletin Científico y Técnico, 21(2), 1-13. [ Links ]
Mora, E., Flores, L., Moreno, J., & Gilbert, G. (2012). La pesquería de la concha prieta (Anadara tuberculosa y Anadara similis) en los principales puertos de desembarque de Ecuador en el 2011. Boletin Científico y Técnico, 22(2), 1-16. [ Links ]
Munro, J. L. & Pauly, D. (1983). A simple method for comparison the growth of fishes and invertebrates. Fishbyte, 1, 5-6. [ Links ]
Ortiz, I. G. & Orellana, G. Y. (1997). Crecimiento de concha prieta (Anadara tuberculosa) en canastas sumergidas con tres densidades de población. (Tesis de Ingeniería en Acuicultura). Universidad Técnica de Machala, Machala, Ecuador. [ Links ]
Pauly, D. (1979). Gillsize and temperature as governing factors in fish growth: a generalization of the von Bertalanffy’s growth formula. Berichte aus dem Institut für Meereskunde an der Christian-Albrechts-Universität Kiel, 63, 1-156. [ Links ]
Pauly, D. (1980). A selection of simple methods for the assessment of tropical fish stocks. FAO Fisheries Circular, No. 729. Rome: FAO. [ Links ]
Pauly, D. & Munro, J. L. (1984). Once more on the comparison of growth in fish and invertebrates. Fishbyte, 2(1), 21. [ Links ]
Perez-Medina, D. (2005). Biología reproductiva de Anadara tuberculosa (Bivalvia: Arcidae) en el Estero Santo Domingo, B.C.S., Mexico. (Tesis de Maestría). Centro Interdisciplinario de Ciencias Marinas, Baja California Sur, México. [ Links ]
Philipp, E. & Abele, D. (2010). Master of longevity: lessons from long-lived bivalves – a mini-review. Gerontology, 56, 55-65. [ Links ]
Samaniego, E. T. (2008). Cultivo de concha prieta Anadara tuberculosa a diferentes densidades en estanques camaroneros utilizando canastas de cultivo. (Tesis de Ingeniería en Acuicultura). Universidad Técnica de Machala, Machala, Ecuador. [ Links ]
Silva, A. & Bonilla, R. (2001). Abundancia y morfometría de Anadara tuberculosa y A. similis (Mollusca: Bivalvia) en el Manglar de Purruja, Golfo Dulce, Costa Rica. Revista de Biología Tropical, 49 (2), 315-320. [ Links ]
Squires, H. J., Estevez, M., Barona, O., & Mora, O. (1975). Mangrove cockles, Anadara spp. (Mollusca: Bivalvia) of the Pacific coast of Colombia. Veliger, 18, 57-68. [ Links ]
Stamps, J. A., Mangel, M., & Phillips, J. A. (1998). A new look at relationships between size at maturity and asymptotic size. The American Naturalist, 152 (3), 470-479. [ Links ]
Stern-Pirlot, A. & Wolff, M. (2006). Population dynamics and fisheries potential of Anadara tuberculosa (Bivalvia: Arcidae) along the Pacific coast of Costa Rica. Revista de Biología Tropical, 54(1), 87-97. [ Links ]
Taylor, C. C. (1958). Cod growth and temperature. Journal du Conseil Permanent International pour l’Exploration de la Mer, 23, 366-370. [ Links ]
Villalobos, C. R. & Baez, A. L. (1983). Tasa de crecimiento y mortalidad de Anadara tuberculosa (Bivalvia: Arcidae) bajo dos sistemas de cultivo en el Pacifico de Costa Rica. Revista Latinoamerica de Acuicultura, 17, 9-18. [ Links ]
Wan, Y. & Milton, D. A. (2000). On comparison of growth curves: How do we test whether growth rates differ? Fisheries Bulletin, 98, 874-880. [ Links ]
Baqueiro-Cárdenas, E. & Aldana-Aranda, D. (2003). Patrones en la biología poblacional de moluscos de importancia comercial en México. Revista de Biología Tropical, 51(4), 97-107. [ Links ]
Borda, C. A. & Cruz, R. (2004a). Crecimiento y tasa de mortalidad del bivalvo Anadara tuberculosa (Sowerby, 1833) en el Pacifico Colombiano. Revista de Investigaciones Marinas, 25, 177-184. [ Links ]
Borda, C. A. & Cruz, R. (2004b). Reproducción y reclutamiento del molusco Anadara tuberculosa (Sowerby, 1833) en el Pacifico Colombiano. Revista de Investigaciones Marinas, 25, 185-195. [ Links ]
Brey, T. & Gage, J. D. (1997). Interactions of growth and mortality in benthic invertebrate populations: empirical evidence for a mortality-growth continuum. Archive of Fishery and Marine Research, 45(1), 45-59. [ Links ]
Caddy, J. F. & Defeo, O. (2003). Enhancing or restoring the productivity of natural population of shellfish and other marine invertebrate resources. FAO Fisheries Technical Paper, No. 448. Rome: FAO. [ Links ]
Cubillos, L., Alarcón, R., & Brante, A. (1999). Empirical estimates of natural mortality for the Chilean hake (Merluccius gayi): evaluation of precision. Fisheries Research, 42, 147-153. [ Links ]
Cubillos, L. & Araya, M. (2007). Estimaciones empíricas de mortalidad natural en el bacalao de profundidad (Dissostichus eleginoides Smitt, 1898). Revista de Biología Marina y Oceanografía, 42(3), 287-297. [ Links ]
Félix-Pico, E. F., Ramírez-Rodríguez, M., & Holguín-Quiñones, O. (2009). Growth and Fisheries of the Black Ark Anadara tuberculosa, a Bivalve Mollusk, in Bahía Magdalena, Baja California Sur, Mexico. North American Journal of Fisheries Management, 29, 231-236. [ Links ]
Flores, L. (2010). Tasa de crecimiento de Anadara tuberculosa (Sowerby, 1833) (Bivalvia: Arcidae) en la Reserva Ecológica Manglares Cayapas-Mataje (REMACAM): un análisis basado en sistemas de cajas suspendidas. Revista de Ciencias del Mar y Limnologia, 4, 89-98. [ Links ]
Flores, L. (2011). Growth estimation of mangrove cockle (Anadara tuberculosa): application and assessment of length based method. Revista de Biología Tropical, 59(1), 159-175. [ Links ]
Flores, L. & Mora, E. (2011). Evaluando variaciones en la talla de Anadara tuberculosa y Anadara similis en el Archipiélago of Jambelí: Hay indicios de sobrepesca? Revista de Ciencias del Mar y Limnologia, 5(1), 33-49. [ Links ]
Flores, L. & Morales, M. (2011). La explotación de Anadara tuberculosa y Anadara similis en Puerto El Morro: un análisis después de una década sin monitoreo. Boletin Científico y Técnico, 21(4), 24-35. [ Links ]
Francis, R. I. C. (1996). Do herring grow faster than orange roughy? Fisheries Bulletin, 94, 783-786. [ Links ]
Gabaev, D. D. & Olifirenko, A. B. (2001). Growth, stock and production of the Anadara scapharca broughtoni in Peter the Great Bay, Sea of Japan. Oceanology, 41(3), 403-412. [ Links ]
Gayanilo, F. C. Jr., Sparre, P., & Pauly, D. (2005). The FAO-ICLARM Stock Assessment Tools II (FISAT II). Revised version. User’s guide. FAO Computerized Information Series (Fisheries), No 8. Rome: FAO. [ Links ]
Guilbert, A. (2007). State of the Anadara tuberculosa (Bivalvia: Arcidae) fishery in las Perlas Archipielago, Panama. (Master of Science thesis). School of Life Science, HeriotWatt University, Edinburgh. [ Links ]
Gil-Agudelo, D. L., Gualteros, W., Delgado, M. F., Lucero, C. H., Espinosa, S., Cortés, N., Palacio, C. J., Roldán, A. M., Zapata, L. A., Candelo, C., Cantera, J. R., Mayor, G., & Muñoz, S. (2010). Potencial productivo de las poblaciones naturales de la piangua Anadara tuberculosa y Anadara similis dentro de una perspectiva espacio-temporal en la costa Pacífica colombiana. (Informe Final. INVEMAR-ASCONAR-WWF-Universidad del Valle-UAESP-NN). Cali: Universidad del Valle. [ Links ]
Haddon, M. (2001). Modelling and quantitative methods in fisheries. New York: Chapman and Hall/CRC. [ Links ]
Heller, J. (1990). Longevity in molluscs. Malacologia, 31(2), 259-295. [ Links ]
Ishiyama, V. & Terukina, S. (1989). Reproduccion de Anadara tuberculosa y Anadara similis en la zona de Puerto Pizarro. Revista Biota, 95, 2-11. [ Links ]
Lucero, C., Cantera, J., & Neira, R. (2012). Pesquería y crecimiento de la piangua (Arcoida: Arcidae) Anadara tuberculosa en la Bahía de Málaga del Pacifico colombiano, 2005-2007. Revista de Biología Tropical, 60(1), 203-217. [ Links ]
MacKenzie, C. L. Jr. (2001). The Fisheries for Mangrove Cockles, Anadara spp., from Mexico to Peru, with Descriptions of their Habitats and Biology, the Fishermen’s Lives, and the Effects of Shrimp Farming. Marines Fisheries Review, 63, 1-39. [ Links ]
Mora, E., Moreno, J., & Jurado, V. (2011). Un análisis sobre la pesquería de la concha prieta en Ecuador, año 2010. Boletin Científico y Técnico, 21(2), 1-13. [ Links ]
Mora, E., Flores, L., Moreno, J., & Gilbert, G. (2012). La pesquería de la concha prieta (Anadara tuberculosa y Anadara similis) en los principales puertos de desembarque de Ecuador en el 2011. Boletin Científico y Técnico, 22(2), 1-16. [ Links ]
Munro, J. L. & Pauly, D. (1983). A simple method for comparison the growth of fishes and invertebrates. Fishbyte, 1, 5-6. [ Links ]
Ortiz, I. G. & Orellana, G. Y. (1997). Crecimiento de concha prieta (Anadara tuberculosa) en canastas sumergidas con tres densidades de población. (Tesis de Ingeniería en Acuicultura). Universidad Técnica de Machala, Machala, Ecuador. [ Links ]
Pauly, D. (1979). Gillsize and temperature as governing factors in fish growth: a generalization of the von Bertalanffy’s growth formula. Berichte aus dem Institut für Meereskunde an der Christian-Albrechts-Universität Kiel, 63, 1-156. [ Links ]
Pauly, D. (1980). A selection of simple methods for the assessment of tropical fish stocks. FAO Fisheries Circular, No. 729. Rome: FAO. [ Links ]
Pauly, D. & Munro, J. L. (1984). Once more on the comparison of growth in fish and invertebrates. Fishbyte, 2(1), 21. [ Links ]
Perez-Medina, D. (2005). Biología reproductiva de Anadara tuberculosa (Bivalvia: Arcidae) en el Estero Santo Domingo, B.C.S., Mexico. (Tesis de Maestría). Centro Interdisciplinario de Ciencias Marinas, Baja California Sur, México. [ Links ]
Philipp, E. & Abele, D. (2010). Master of longevity: lessons from long-lived bivalves – a mini-review. Gerontology, 56, 55-65. [ Links ]
Samaniego, E. T. (2008). Cultivo de concha prieta Anadara tuberculosa a diferentes densidades en estanques camaroneros utilizando canastas de cultivo. (Tesis de Ingeniería en Acuicultura). Universidad Técnica de Machala, Machala, Ecuador. [ Links ]
Silva, A. & Bonilla, R. (2001). Abundancia y morfometría de Anadara tuberculosa y A. similis (Mollusca: Bivalvia) en el Manglar de Purruja, Golfo Dulce, Costa Rica. Revista de Biología Tropical, 49 (2), 315-320. [ Links ]
Squires, H. J., Estevez, M., Barona, O., & Mora, O. (1975). Mangrove cockles, Anadara spp. (Mollusca: Bivalvia) of the Pacific coast of Colombia. Veliger, 18, 57-68. [ Links ]
Stamps, J. A., Mangel, M., & Phillips, J. A. (1998). A new look at relationships between size at maturity and asymptotic size. The American Naturalist, 152 (3), 470-479. [ Links ]
Stern-Pirlot, A. & Wolff, M. (2006). Population dynamics and fisheries potential of Anadara tuberculosa (Bivalvia: Arcidae) along the Pacific coast of Costa Rica. Revista de Biología Tropical, 54(1), 87-97. [ Links ]
Taylor, C. C. (1958). Cod growth and temperature. Journal du Conseil Permanent International pour l’Exploration de la Mer, 23, 366-370. [ Links ]
Villalobos, C. R. & Baez, A. L. (1983). Tasa de crecimiento y mortalidad de Anadara tuberculosa (Bivalvia: Arcidae) bajo dos sistemas de cultivo en el Pacifico de Costa Rica. Revista Latinoamerica de Acuicultura, 17, 9-18. [ Links ]
Wan, Y. & Milton, D. A. (2000). On comparison of growth curves: How do we test whether growth rates differ? Fisheries Bulletin, 98, 874-880. [ Links ]
*Correspondencia a:
Luis Flores. Investigación de Recursos Bioacuáticos y su Ambiente, Instituto Nacional de Pesca, Letamendí 102 y La Ría, P.O. Box 09-01-15131, Guayaquil, Ecuador. Correo electrónico lflores@inp.gob.ec
Roberto Licandeo. Fisheries Centre, Aquatic Ecosystems Research Laboratory (AERL), University of British Columbia, Vancouver, Canada. Correo electrónico r.licandeo@fisheries.ubc.ca
Luis A. Cubillos. Programa COPAS Sur-Austral, Departamento de Oceanografía, Facultad de Ciencias Naturales y Oceanográficas, Universidad de Concepción, Casilla 160-C, Concepción, Chile. Correo electrónico lucubillos@udec.cl
Elba Mora. Investigación de Recursos Bioacuáticos y su Ambiente, Instituto Nacional de Pesca, Letamendí 102 y La Ría, P.O. Box 09-01-15131, Guayaquil, Ecuador. Correo electrónico emora@inp.gob.ec
1. Investigación de Recursos Bioacuáticos y su Ambiente, Instituto Nacional de Pesca, Letamendí 102 y La Ría, P.O. Box 09-01-15131, Guayaquil, Ecuador. Correo electrónico lflores@inp.gob.ec, emora@inp.gob.ec
2. Fisheries Centre, Aquatic Ecosystems Research Laboratory (AERL), University of British Columbia, Vancouver, Canada. Correo electrónico r.licandeo@fisheries.ubc.ca
3. Programa COPAS Sur-Austral, Departamento de Oceanografía, Facultad de Ciencias Naturales y Oceanográficas, Universidad de Concepción, Casilla 160-C, Concepción, Chile. Correo electrónico lucubillos@udec.cl
Received 10-iv-2013. Corrected 05-Xi-2013. Accepted 05-Xii-2013.












 uBio
uBio 

