Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.59 n.1 San José Mar. 2011
Phagodeterrence by Quassia amara (Simaroubaceae) wood extract fractions on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae
Francisco Soto1, Luko Hilje2, Gerardo A. Mora3 & Manuel Carballo2
1. Graduate School. Tropical Agricultural Research and Higher Education Center (CATIE). Turrialba, Costa Rica; fsoto@catie.ac.cr
2. Department of Agriculture and Agroforestry. Tropical Agricultural Research and Higher Education Center (CATIE). Turrialba, Costa Rica; lhilje@catie.ac.cr, mcarball@catie.ac.cr
3. Associate Researcher. Natural Products Research Center (CIPRONA). Universidad de Costa Rica. San José, Costa Rica; gerardo.moralopez@ucr.ac.cr
Dirección para correspondencia
Abstract
In Latin America and the Caribbean, precious wood species like mahoganies (Swietenia spp.) and cedars (Cedrela spp.) are seriously injured by the mahogany shootborer, Hypsipyla grandella (Zeller) (Lepidoptera: Pyralidae) larva, which bores into the main shoot of trees. In previous experiments focused on searching for a preventive method for managing this pest, a wood extract of bitterwood, Quassia amara L. ex Blom (Simaroubaceae) had been shown to cause phagodeterrence to larvae. Therefore, three fractions (water, methanol and diethyl ether) of a wood extract were tested for their phagodeterrence to larvae, by means of laboratory and greenhouse trials. Phagodeterrence was assessed by determining their effect on foliage consumption, mortality and signs of damage (number of orifices, sawdust piles, fallen shoots, number of tunnels and tunnel length) caused by larvae on Spanish cedar (C. odorata). Both the methanol and diethyl ether fractions caused phagodeterrence, by strongly reducing foliage consumption and signs of damage, while not causing larval mortality. The lowest concentration at which phagodeterrence was detected for the methanol fraction corresponded to 0.0625%, which is equivalent to a 1.0% of the bitterwood crude extract. However, results with the diethyl ether fraction were unsatisfactory, as none of the treatments differed from the solvent, possibly because of an adverse effect of the solvent on foliar tissues. Phagodeterrent principles from Q. amara derivatives may play an important role in dealing with H. grandella if they are complemented with other integrated pest management preventative tactics. Rev. Biol. Trop. 59 (1): 487-499. Epub 2011 March 01.
Key words: Hypsipyla grandella, mahogany shootborer, plant extracts, Quassia amara, bitterwood, phagodeterrence.
Resumen
En América Latina y el Caribe, algunas especies que son fuente de maderas preciosas, como las caobas (Swietenia spp.) y cedros (Cedrela spp.), son seriamente dañadas por la larva de Hypsipyla grandella, la cual barrena el brote principal de los árboles. En experimentos previos orientados hacia la búsqueda de un método preventivo para manejar esta plaga, un extracto de la madera de hombre grande, Quassia amara (Simaroubaceae), había demostrado causar fagodisuasión a la larva. Por tanto, tres fracciones de un extracto de la madera de dicho árbol (agua, metanol y éter dietílico) fueron evaluadas en cuanto a su efecto fagodisuasivo sobre las larvas, mediante experimentos de laboratorio e invernadero. Dicho efecto se determinó según el consumo de follaje, la mortalidad y los signos de daño (número de orificios, montículos, brotes caídos, número y longitud de túneles) causados por las larvas en arbolitos de cedro amargo (C. odorata). Tanto la fracción de metanol como la de éter dietílico causaron fagodisuasión, pues redujeron fuertemente el consumo de follaje y los signos de daño, sin matar las larvas. La menor concentración de la fracción metanólica a la cual se detectó fagodisuasión correspondió a 0.0625%, la cual equivale al 1.0% del extracto crudo de hombre grande. Por su parte, los resultados con la fracción de éter dietílico fueron insatisfactorios, ya que ninguna de las concentraciones difirió del disolvente, quizás debido a un efecto adverso del mismo sobre los tejidos foliares. Los principios fagodisuasivos de los derivados Q. amara podrían jugar un papel importante para manejar a H. grandella, si fueran complementados con otras tácticas preventivas de manejo integrado de plagas.
Palabras clave: Hypsipyla grandella, barrenador de meliáceas, extractos vegetales, Quassia amara, hombre grande, fagodisuasión.
The mahogany shootborer, Hypsipyla grandella (Zeller) (Lepidoptera: Pyralidae) is a key forest pest throughout the neotropics, as it attacks precious wood trees of the Meliaceae family. Its larva bores into terminal shoots of mahoganies (Swietenia spp.) and cedars (Cedrela spp.), as well as other related species (Schabel et al. 1999), breaking the apical dominance, which causes forking of the stems and excessive production of lateral branches; this can occur as early as the plantlet stage.
Likewise, since a density as low as one larva per shoot is high enough to cause irreversible damage and high economic losses, this pest has precluded attempts to establish commercial plantations of mahoganies and cedars in Latin America and the Caribbean.
In the search for a preventative approach to deal with this pest, it has been shown that some plant extracts can act as larval feeding deterrents (Hilje & Mora 2006), the main ones being methanol extracts of both the wood and leaves of the bitterwood tree (Quassia amara L. ex Blom, Simaroubaceae) (Mancebo et al. 2000, Soto et al. 2007), as well as leaf extracts of common rue (Ruta chalepensis L. Rutaceae) (Mancebo et al. 2001, Soto et al. 2007, Barboza et al. 2010).
Q. amara is a neotropical forest shrub or small tree, whose range extends from Mexico to Ecuador, including the Caribbean basin, where it normally grows in the forest understory, but it also grows easily in disturbed areas (Villalobos 1995). Its wood contains several quassinoids with insecticidal properties (Polonsky 1973), which explains why Q. amara was one of the botanical insecticides widely used before synthetic insecticides were developed (Metcalf et al. 1951).
In recent years there has been a renewed interest in promoting this plant as a useful resource for Mesoamerican communities, so that studies have been conducted on several ecological and silvicultural aspects of the species, as well as on potential markets for its products (Ocampo 1995, Ocampo & Díaz 2006).
Considering preliminary results by Mancebo et al. (2000), the objective of this research was to assess some fractions of Q. amara in regards of their phagodeterrent effect on H. grandella larvae, and to gain insight into more specific groups of substances causing such effect, under both laboratory and greenhouse conditions.
Materials and methods
Extract preparation: Plant material was collected from a single location and at the same time, in February 2000, within the Keköldi Indian Reservation (Talamanca, Costa Rica), in order to avoid undesirable variability due to geographic or seasonal differences. It was stored under appropriate conditions for about a month.
The hydroalcoholic extract was prepared at the Natural Products Research Center (CIPRONA) (San José, Costa Rica) from wood chips dried in an oven at 40ºC, ground and placed in 70% methanol in a suitable flask for 24h; the solvent was drained and the residue was treated again with methanol for 24h. The pooled extracts were filtered through a Whatman No. 4 filter paper, and concentrated at 40ºC using a rotary evaporator. The final residue was freeze-dried to eliminate any water remaining in the crude extract.
In addition, to obtain the fractions (water, methanol and diethyl ether), a column 31cm high and 4.5cm in diameter was packed with 100g of the synthetic resin Diaion HP-20 (Mitsubishi Chemical Industry Ltd., Tokyo, Japan). The resin was washed with water, methanol, and diethyl ether and then the order of washings was reversed. A maximum of 10g of the crude freeze-dried extract was placed on the column and eluted with 1L each of the solvents, starting with water and finishing with diethyl ether. The column was used as many times as necessary to completely process one batch of crude extract. The solvents were evaporated and freeze-dried, if necessary, to obtain the weight of each fraction, in order to establish the proper dose to be used in the bioassay.
Starting with 40g of the freeze-dried wood extract, the fractionation process produced 9.2, 2.5 and 0.56g of the freeze-dried water, methanol and diethyl ether fractions, respectively. The yield obtained for each fraction in terms of weight revealed its proportion in the crude extract. This composition determined the concentration at which every fraction was dissolved to be applied in the experiments, allowing an equivalent comparison among fractions, as well as between them and the crude extract. The freeze-dried crude extract and the corresponding fractions were kept hermetic, refrigerated and in darkness, to avoid their chemical decomposition or contamination by fungi.
Experiments: Both the crude extract and its three fractions were assessed for their feeding deterrence on H. grandella larvae through two laboratory (general and specific bioassays) and one greenhouse experiments, which took place at CATIE (Tropical Agricultural Research and Higher Education Center), in Turrialba, within the Caribbean watershed of Costa Rica, at 9°52’ N; 83°38’ W and 640 m.a.s.l., with annual averages of 22ºC, 2479mm (rainfall) and 87% RH. The whole experiment lasted for about five months.
Larvae were taken from a colony maintained at CATIE, where they are initially reared on tender foliage of Spanish cedar (Cedrela odorata) and later transferred to artificial diet (Vargas et al. 2001). Third-instar larvae, which had been fed exclusively on cedar foliage, were selected because the amount of foliar tissue they consume facilitates comparisons among treatments; also, their size allows easy handling, without harming them, and the duration of such instar is long enough as to monitor their feeding behavior while exposed to treated disks.
General bioassay: Each fraction was tested at the concentration it would have if the crude extract had been dissolved at 10% w/v (weight/volume), according to the yield from the fractioning process, such concentration was the highest one at which phagodeterrence on H. grandella larvae had been detected previously (Mancebo et al. 2000).
Thus, treatments corresponded to the following concentrations (w/v) for each fraction: 2.3 (water), 0.625 (methanol) and 0.14% (diethyl ether); 100ml of dissolution were prepared for each fraction, through weighting the specific amount of the freeze-dried fraction and dissolving it in its respective solvent (water, methanol or diethyl ether).
The absolute control treatment corresponded to 100ml of Q. amara crude extract dissolution at 10% (in water); it was diluted from a stock dissolution of 21.2%, which was the concentration produced from the extraction process. Relative control treatments corresponded to the pertinent solvents (water, methanol and diethyl ether).
All dissolutions were prepared the same day they were used, by mixing the freeze- dried extract of each one of the fractions in the corresponding solvent. The emulsifier Nu Film 17 (96% Di-1-p-menthene) (Millar Chemical Corp. Fertilizer, USA) was added to all of them at 0.03%, as well as to each one of the relative control treatments.
Disks of Spanish cedar tender foliage (2.3cm in diameter) were cut with a cork- borer from healthy leaves collected the same day that were used, in a way to avoid vein portions where small larvae could bore into. Disks were dipped in the respective treatment for 10s, and allowed to dry for 30min. Treated disks were placed individually in 30ml glass flasks, along with a third-instar H. grandella larva which had been deprived of food for 3h. A piece of paper towel was fastened with the lid of each flask and was moistened periodically, in order to keep the disk turgid.
Larvae to be included within the same block were taken from a single rearing box, by means of a fine hairbrush, as to guarantee genetic uniformity; instar III larvae were identified for its typical length (approximately 5mm) and whitish color.
A randomized complete block design, including sub-sampling, was used, involving four replicates. Each block was represented by a plastic tray, and the experimental unit consisted of eight larvae, each one being considered as a subsample. Larvae (subsamples) were randomized within each tray, as well as treatments within each block.
Trays were placed inside a Percival I-35L environmental chamber (Percival Scientific. Inc., Perry, Iowa) set at 25°C, 80-90% RH, and 8:16 (L:D) photoperiod. As trays were stacked, their position within the stack was changed on a daily basis in order for all of them to be equally exposed to light within the chamber; also, this allowed all trays to be evaluated at the same time between days.
After being exposed to the corresponding treatment for 24h, each larva was transferred to a flask containing about 6ml of artificial diet, where it was allowed to complete its development; larvae were transferred to other flasks in cases when the diet was not suitable for their development. Once larvae became pupae they were weighted in an electronic balance (Mettler PM 60) and placed individually in 20ml plastic jars until either adult emergence or their death (for 20 days since their appearance).
Phagodeterrence was determined by refusal of larvae to eat treated leaf disks, but once they were placed into flasks containing an artificial diet they continued feeding and completed their development, normally reaching the adult stage. Therefore, three types of variables were measured in response to Q. amara treatments: food consumption, mortality and developmental effects (instar duration and pupal weight).
Food consumption was assessed for each disk, by recording the percentage of foliar area which was consumed in 24h. This was done by means of a visual scale of the program Distrain 1.0 (Tomerlin & Howell 1988), for which the observer got adequate training in advance.
Mortality was determined for each larva every 24h, for a week, and the instar at which mortality occurred was recorded; cessation of movement and color change to black, as well as a soft and wet appearance, were the criteria used for judging mortality.
Developmental effects included time between larval instars to pupa, as well as pupal weight a day after pupation, time for larval molting, conversion into pupae and adult emergence. For the general bioassay, IV and V instars and pupa duration were determined, as well as time elapsing to adult emergence; instar change was detected by locating cephalic capsules inside the flasks. Pupae were weighted a day after larvae turned into pupae by taking them out of their silky cocoon.
Greenhouse experiment: Treatments were the same ones as in the previous section. A randomized complete block design was used, involving eight replicates, the experimental unit being represented by a single tree.
Seeds were obtained from a Spanish cedar progeny from Pococí, Limón, Costa Rica. Cedar plants, 50-75cm in height and 6-month age, were planted in 2 000ml pots and placed on the ground. Each plant was leaf-pruned, leaving only the topmost eight compound leaves ("branches").
Treatments were applied on the terminal portion (main shoot plus three youngest "branches") of trees. These were sprayed outside the greenhouse, to prevent contamination between treatments, by means of a DeVilbiss 15 hand-sprayer, with an adjustable tip (The DeVilbiss, Somerset, Pennsylvania), which was connected to a GAST air pump, model DOAP104- AA (GAST Manufacturing Corp. Benton Harbor, Michigan), under a constant pressure (0.7kg/cm2). Three first-instar H. grandella larvae were placed on the terminal portion of each tree, with a fine hairbrush, 30min later. Such instar larvae were used not only for more closely mimicking what occurs during colonization under field conditions, but also because variables to be measured had to be recorded throughout H. grandella life cycle.
Variables included the number of orifices, sawdust piles and tunnels made by larvae. Orifices were counted on the main shoot as well as on the axils of branches, two days after larval inoculation. Sawdust piles (reddish, soft and moist mounds of sawdust, silk and excrement, resulting from larval feeding), as well as wilt shoots and fallen shoots and "branches" were recorded two days, one week and two weeks after larval inoculation. Tunnel number and length, as well as larvae and pupae inside them were counted four weeks after larval inoculation.
Specific bioassays: Treatments corresponded to those concentrations at which each fraction would be represented in the crude Q. amara extract if it were to be applied at the following doses: 0.1, 0.32, 1.0, 3.2 and 10% (w/v), and according to yields obtained in the fractioning process. Therefore, five treatments (concentrations, w/v) for each fraction were tested: 0.0062, 0.0200, 0.0620, 0.2000 and 0.6200% (methanol) and 0.0014, 0.004, 0.014, 0.044 and 0.1400% (diethyl ether).
Treatments were compared to the control (distilled water) and to the respective solvents, which corresponded to a mixture of 20% methanol in water and an emulsified mixture of 20% diethyl ether in water, for the respective fractions. Diluted solvents were so used in order to avoid some leaf disk phytotoxicity caused by pure solvents and observed from the specific bioassays.
Solutions were prepared by dilution, departing from the most concentrated fraction. Methanol fraction solutions were added with 0.03% of the emulsifier Nu Film 17 (96% a.i.), whereas diethyl ether fraction solutions were added with 0.03% of Citowett (99% alkoxilate) (BASF, Germany), as only the latter allowed an appropriate emulsification of diethyl ether and water. The rest of procedures were as in the general bioassay.
Analysis: Data were analyzed by means of ANOVA procedures and means were compared by the Tukey´s test (SAS Institute 1985), at a significance level of α=0.05. Data for orifice number, sawdust piles and fallen shoots were transformed by the logarithmic method, in order to meet the assumptions underlying ANOVA. In all cases, such assumptions were verified. Repeated ANOVA procedures were carried out in order to determine interaction between sawdust piles and the number of fallen shoots over time. In addition, regression analyses were performed for disk leaf consumption percentage in response to Q. amara extract concentrations.
Results
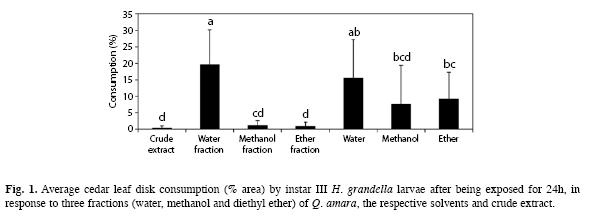
General bioassay: In terms of leaf disk consumption by H. grandella larvae, there were very large differences between treatments (F=19.39, d.f.=6, p<0.01) (Fig. 1). The lowest consumption averages were attained with the methanol and diethyl ether fractions along with the crude extract (0.34-1.16%) and the highest (15- 20%) with the water fraction and the water control treatment.
Moreover, there was a strong contrast in consumption between the diethyl ether fraction and the respective solvent (p<0.05), but not between the methanol fraction and its corresponding solvent. Also, there were no differences (p>0.05) between any of the solvents.
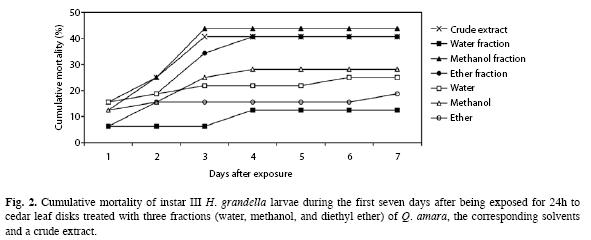
Regarding larval mortality, there were no differences between treatments for the first (F=0.67, d.f.=3, p>0.05) and second days (F=1.20, d.f.=3, p>0.05) (Fig. 2), when larvae were exposed to disks and when they were in the artificial diet, respectively. Differences became evident between days three and seven, with the methanol (44%) and water fractions (12%) occupying the extremes.
In general, mortality values due to the methanol fraction were closely followed (p>0.05) by those of the crude extract and the diethyl ether fraction, both attaining 41%. Moreover, in terms of cumulative mortality until pupation occurred (total mortality), the highest values (62 and 66%) were attained with the diethyl ether and methanol fractions, respectively, whereas the lowest one (22%) corresponded to the water fraction.
Developmental times for both fourth (3.2- 4.5 days) and fifth larval (3.6-4.5 days) instars, as well as for pupae (10.0-11.8 days), did not differ (p>0.05) among treatments. Also, neither pupal weight (0.08-0.11g) nor the time elapsed from experiment start to adult emergence (35.8- 45.2 days) differed between treatments.
Greenhouse experiment: Concerning the number of orifices two days after tree inoculation with larvae, there were large differences between treatments (F=16.33, d.f.=6, p<0.01) (Fig. 3A). No orifices were found on trees treated with either the methanol fraction or the crude extract, whereas they were rare on those treated with the diethyl ether fraction, so that they did not differ (p>0.05) among them. However, all of them differed from the corresponding solvents, which in turn did not differ from the water fraction. Among the solvents, there were differences between trees treated with water and diethyl ether.
In terms of the number of sawdust piles, there were large differences in the treatments (F=69.11, d.f.=6, p<0.01), as well as among evaluation dates (F=12.43, d.f.=2, p<0.01).
Although interactions between treatments and evaluation dates showed strong differences (F=3.18, d.f.=12, p<0.01), the trend of the three evaluation dates was consistent throughout the evaluation period. Sawdust piles were null on trees treated with the crude extract and almost null on those trees treated with the diethyl ether and methanol fractions. These treatments did not differ among them, but did from both the water fraction and the respective solvents (p<0.05) throughout the three evaluation periods. In turn, water did not differ from methanol, but did from diethyl ether. This trend was well reflected in the average of three evaluation dates (Fig. 3B), representing sawdust pile accumulation over time in the same tree.
Regarding the number of fallen shoots, there were large differences between treatments (F=75.91, d.f.=6, p<0.01), as well as between evaluation times (F=26.56, d.f.=2, p<0.01).
As in the case of sawdust piles, interactions between treatments and evaluation times showed strong differences (F=5.76, d.f.=12, p<0.01). Its trend among the three evaluation dates can be described as follows. Fallen shoots did not occur on trees treated with either the crude extract or the diethyl ether fraction, while barely appeared on those treated with the methanol fraction two weeks after inoculation. Such treatments did not differ between them, but they did (p<0.05) from the corresponding solvents, except for the diethyl ether fraction two days after inoculation and from the water fraction. In turn, solvents did not differ from the water fraction, even though no clear-cut trends were observed for all cases. However, the trend of treatments which had an effect on shoot falling was well represented by the average of the three evaluation dates (Fig. 3C).
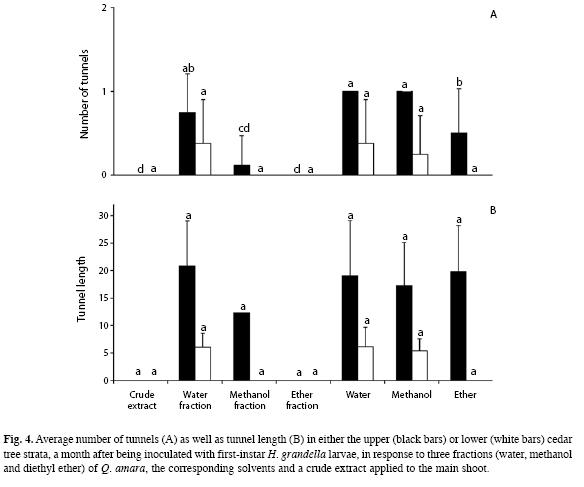
In terms of the number of tunnels present in the upper portion four weeks after the trees were inoculated with larvae, there were large differences between treatments (F=17.93, d.f.=6, p<0.01). Tunnels did not occur in trees with either the crude extract or the diethyl ether fraction, while barely appeared in those treated with the methanol fraction (Fig. 4A), which did not differ from trees treated with diethyl ether. The diethyl ether-treated trees did not differ from those treated with the water fraction.
Also, numbers of larvae and pupae found inside differed considerably (F=4.71, d.f.=6, p<0.01), ranging from an average of 0.88 in trees treated with water and methanol, to zero in those treated with either the crude extract or the diethyl ether fraction. Otherwise, there were no differences between treatments regarding the number of tunnels in the lower portion of the trees, nor in the numbers of larvae and pupae found inside them.
Concerning tunnel length, there were no differences between treatments (p>0.05) for neither the upper (F=0.29, d.f.=4, p>0.05) or the lower portion (F=0.03, d.f.=2, p>0.05), ranging from 0-20.8 and 0-6.1 for either portion, respectively (Fig. 4B). Tunnels in the lower portion were much shorter and non linear, resembling galleries with branches in several directions.
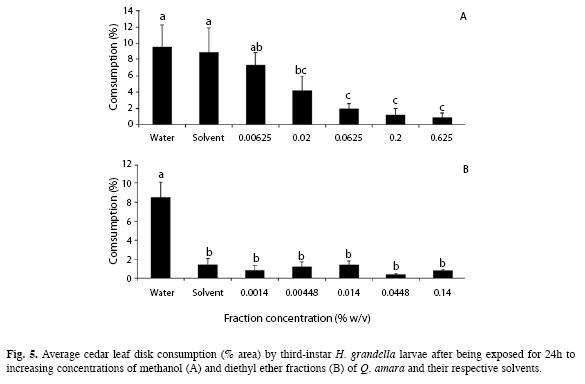
Specific bioassays: Bioassays included only the methanol and diethyl ether fractions, as they were the only ones showing distinct and consistent phagodeterrence in the two previous experiments. For the methanol fraction, in terms of leaf disk consumption by larvae, there were large differences between treatments (F=25.95, d.f.=6, p<0.01) (Fig. 5A). Consumption was significantly reduced at the four highest concentrations, with no differences (p>0.05) between them. The two control treatments (water and methanol) did not differ between them, nor with the lowest concentration of the fraction; the latter one did not differ from the 0.02% concentration.
A logarithmic model best fitted the response curve between leaf disk consumption and methanol fraction concentration (Y=2.27X-0.48, R2=0.97, p<0.01), represented by the equivalent values of crude extract concentration which would have been applied to coincide with the methanol fraction doses used in the experiment
In addition to leaf disk consumption, only mortality was further assessed, since the general bioassay analysis revealed that those variables associated with H. grandella larval and pupae development were not influenced by any treatment.
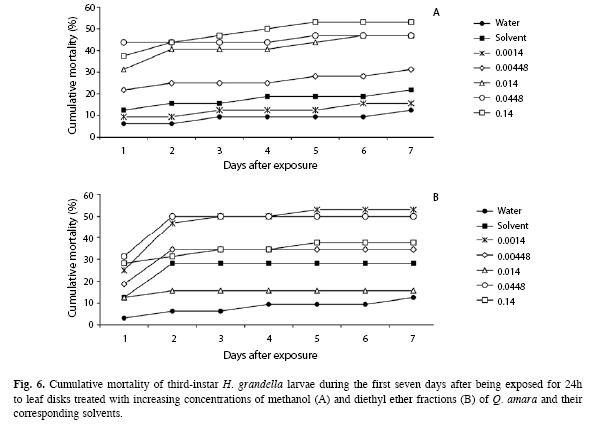
Concerning larval mortality, there were differences between treatments for both the first (larvae exposed to disks) and the rest of days (larvae in the artificial diet) (Fig. 6A), except in the last day. The highest cumulative mortality average (equivalent to 53% of the exposed larvae) was attained at the highest concentration (53.12%±23.66) from the fifth day on; it always differed from the control treatment up to the sixth day (9.38%±6.25, p<0.05), but not from the rest of treatments; it also differed from the lowest concentration from the fourth (12.50%±14.43) to the sixth days (15.62%±11.97).
Treatments corresponding to the second and third highest concentrations differed from the control treatment only on some days, but trends were rather erratic. The solvent never differed (p>0.05) from any of the Q. amara concentrations, whereas the lowest mortality values were achieved with the control treatment and the solvent, which did not differ (p>0.05) amongst them.
For the diethyl ether fraction, regarding leaf disk consumption, there were large differences between treatments (F=67.97, d.f.=6, p<0.01) (Fig. 5B). The lowest consumption averages were attained with all the concentrations and the solvent, with no differences (p>0.05) between them. It was not possible to obtain a clear- cut response curve between fraction concentration and leaf disk consumption, as with the methanol fraction, as no model fitted well this correlation.
In terms of mortality, there were differences during the whole period (Fig. 6B), except for the first day (when larvae were exposed to disks). The highest cumulative mortality average (equivalent to 53.12%±12.0) of the exposed larvae was attained with the lowest concentration from the fifth day on; it always differed from the control (12.5%±14.43, p<0.05), but not from the rest of treatments; only the lowest one and the 0.0448% concentrations consistently differed from the control treatment, whereas the other treatments showed erratic trends. The solvent never differed (p<0.05) from any of the concentrations, attaining 21.88%±21.35 for the last day.
Discussion
Findings herewith reported confirm previous results from Mancebo et al. (2000) concerning the presence of phagodeterrent principles for H. grandella larvae in Q. amara extracts, in which quassinoids such as quassin and neoquassin possibly predominate (Polonsky 1973) and there are also some carboline and related alkaloids (Barbetti et al. 1987). H. grandella larvae possess deterrent receptors in the medial and/or lateral sensilla styloconica located on the maxillae (Schoonhoven 1980). Leskinen et al. (1984) found that a type of quassin from Q. amara deters feeding by instar IV larvae of Epilachna varivestis (Coleoptera: Coccinellidae).
Moreover, testing of fractions to gain insight into more specific groups of substances responsible for feeding deterrence lent even more support to those findings. Strong feeding deterrence was attained when larvae were exposed to disks treated with the methanol and diethyl ether fractions. Water fraction lacked any activity. This finding provides the basis for future work on isolation, identification and characterization of individual compounds and the whole mix of each fraction. For example, quassin is soluble in polar solvents like methanol and non-polar solvents like benzene and acetone but not in other non-polar solvents, such as diethyl ether (Budavari 1989); consequently, it is likely that the methanol fraction contained quassin, which caused phagodeterrence.
Methanol and diethyl ether fractions did not differ from the crude extract, which showed a slightly higher deterrence. This suggests that for some plant species there is a possible synergism between substances present in the crude extract as a result of a joint biological effect among extract fractions. Warthen & Morgan (1990), summarizing findings from other authors, stated that when pure deterrent compounds are tested individually at the same concentration at which they occur in a plant, they are less active that the plant itself, but when all individual components are combined resulting deterrence is comparable to the effect produced by the plant itself.
Results were quite consistent in both laboratory and greenhouse experiments. Even though a direct toxic effect of some fractions can not be ruled out altogether, data do not substantiate any such effect.
In laboratory bioassays larvae barely consumed treated leaf disks with the methanol and diethyl ether fractions, as well as with the crude extract, showing low mortality once they were transferred to artificial diet. The observed mortality was probably due to starvation and weakness resulting from ingesting even very low amounts of treated foliage. In addition, developmental times for IV and V larval instars and pupae, pupal weight and the interval for adult emergence were not affected by any treatment.
In the greenhouse experiment null or very low numbers of orifices, sawdust piles, fallen shoots and tunnels were a clear-cut expression of phagodeterrence, leading to starvation and larval death. In this case, despite larvae had the opportunity to search for feeding on untreated sites within the tree, they were probably impaired and died; in fact, tunnels in the lower portion of the trees were detected only for the water fraction as well as for some solvents, which could be due to competition avoidance in the upper portion of the trees.
Furthermore, when determining by bioassays the minimum concentration at which the methanol and diethyl ether fractions would cause phagodeterrence to H. grandella larvae, there was a distinct dose-response trend for the former but not for the latter treatments.
For the methanol fraction, leaf disk consumption was lowest and not significantly different among the four highest concentrations, 0.02% being the lowest of them. Another way to express this is that the response curve for leaf disk consumption and methanol fraction concentration (represented as crude extract concentration) tended to be constant when approaching 0.0625% of the methanol fraction, which represents 1.0% of the crude extract.
For the diethyl ether fraction, leaf disk consumption trends were erratic, as none of the concentrations differed from the solvent. Even though the latter was prepared mixing 20% of diethyl ether in water and adding an emulsification agent, disks underwent dehydration, pigment lixiviation and a slight corrugation, which could turn tissues less edible to H. grandella larvae, precluding any possible effects of the substances present in the diethyl ether fraction to induce low consumption rates. Similarly, cumulated mortality during the period of observations showed erratic trends.
From a practical standpoint, feeding deterrence principles from Q. amara derivatives could play an important role for managing H. grandella. It would be possible to eventually formulate a commercial product to be deployed during the critical period to H. grandella damage, as this pest is especially critical during the first 5-8 years of tree development, depending on the region, site quality and stand management (Cibrián et al. 1995).
Therefore, during such a period, phagodeterrents could be complemented with other integrated pest management (IPM) preventative tactics, such as plant breeding, silvicultural practices and biological control (Newton et al. 1993, Speight 1997), to accomplish commercial and profitable lumber production of wood from mahogany and cedar species.
For the agrichemical industry to get involved into developing commercial deterrents based upon Q. amara derivatives, a number of factors seem to be quite favorable.
First, they offer a unique opportunity in terms of safety, as they would not pose risks to humans and other mammals. Q. amara is a traditional natural medicine in some neotropical countries (Ocampo 1995) and nowadays Q-assia is the brand of a pharmaceutical product for digestive problems, which is manufactured by Lisanatura, a local pharmaceutical company in Costa Rica.
Secondly, efforts from previous years in promoting the utilization of the Q. amara tree as an economic resource for local communities in Mesoamerica have given rise to promising and concrete initiatives.
For instance, since Q. amara can be established as a plantation tree, Bougainvillea S.A., a Costa Rican company, is making a systematic effort for domesticating the shrub, developing harvesting practices compatible with the conservation of the species and assurance of a high content of quassinoids, as well as optimizing the extraction conditions to produce an industrial extract for insecticidal purposes (Ocampo & Díaz 2006).
Finally, systemic effects of a Q. amara crude extract have been demonstrated (Soto et al. 2007), which offers a great potential for managing H. grandella. In fact, some systemic insecticides, such as methomyl and carbofuran, have been applied in pellet form at planting of Spanish cedar trees and provided complete control of H. grandella for several months (Allan et al. 1973, Wilkins et al. 1976).
But, in this case, it would be feasible to develop formulations suitable to be applied through either tree implants or microinjections that are commercially available. By doing so, exposure by people and wildlife, including pest natural enemies and pollinators, would be avoided, and water sources and soil would be protected as well, thus contributing in the development of sustainable plantation forestry systems.
Acknowledgments
This paper is a partial result of a CATIE’s M.Sc. thesis by the first author, which was funded by the Japan-IDB Scholarship Program. Thanks are due to Gilberto Páez and Johnny Pérez (CATIE) for their support in statistical aspects; Edgar Esquivel, Arturo Ramírez, and Guido Sanabria (CATIE), for their logistic support at both the Biotechnology and Entomology laboratories; Róger Villalobos (CATIE), for his help in collecting plant material; and Juan Carlos Brenes (CIPRONA), for preparing the extracts for the experiments.
References
Allan, G.G., R.I. Gara & R.M. Wilkins. 1973. The evaluation of some systemic insecticides for the control of larvae in Cedrela odorata L., p. 40-48. In P. Grijpma (ed.). Studies on the shootborer Hypsipyla grandella (Zeller). Lep. Pyralidae v. l. IICA Misc. Publ. No. 101. Turrialba, Costa Rica. [ Links ]
Barbetti, P., G. Grandolini, G. Fardella & I. Chiappini. 1987. Indole alkaloids from Quassia amara. Planta Med. 53: 289-290. [ Links ]
Barboza, J., L. Hilje, J. Durón, V. Cartín & M. Calvo. 2010. Fagodisuasión de un extracto de ruda (Ruta chalepensis, Rutaceae) y sus particiones sobre larvas de Hypsipyla grandella (Lepidoptera: Pyralidae). Rev. Biol. Trop. 58: 1-14. [ Links ]
Budavari, S. 1989. The Merck Index. Merck & Co. Rahway, New Jersey, USA. [ Links ]
Cibrián, D., J.T. Méndez, R. Campos, H.O. Yates III & J.E. Flores. 1995. Insectos forestales de México. Universidad Autónoma de Chapingo-Comisión Forestal de América del Norte (COFAN). Publication No. 6. [ Links ]
Hilje, L. & G.A. Mora. 2006. Promissory botanical repellents/ deterrents for managing two key tropical insect pests, the whitefly Bemisia tabaci and the mahogany shootborer Hypsipyla grandella, p. 379-403. In M. Rai & C. Carpinella (eds.). Naturally occurring bioactive compounds: a new and safe alternative for control of pests and diseases. Advances in phytomedicine. Elsevier, Amsterdam, Holanda. [ Links ]
Leskinen, V., J. Polonsky & S. Bhatnagar. 1984. Antifeedant activity of quassinoids. J. Chem. Ecol. 10: 1497-1507. [ Links ]
Mancebo, F., L. Hilje, G. Mora & R. Salazar. 2000. Antifeedant activity of Quassia amara (Simaroubaceae) extracts on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae. Crop. Prot. 19: 301-305. [ Links ]
Mancebo, F., L. Hilje, G. Mora, V. Castro & R. Salazar. 2001. Biological activity of Ruta chalepensis (Rutaceae) and Sechium pittieri (Cucurbitaceae) extracts on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae. Rev. Biol. Trop. 49: 501-508. [ Links ]
Metcalf, C.L., W.P. Flint & R.L. Metcalf. 1951. Destructive and useful insects. McGraw-Hill, New York, USA. [ Links ]
Newton, A., P. Baker, S. Ramnarine, J.F. Mesén & R.R.B. Leakey. 1993. The mahogany shoot borer: Prospects for control. For. Ecol. Mgmnt. 57: 301-328. [ Links ]
Ocampo, R.A. (ed.). 1995. Potencial de Quassia amara como insecticida natural. Serie Técnica CATIE. Informe Técnico No. 267. Turrialba, Costa Rica. [ Links ]
Ocampo, R.A. & R. Díaz. 2006. Cultivo, conservación e industrialización del hombre grande (Quassia amara). Litografía e Imprenta LIL, San José, Costa Rica. [ Links ]
Polonsky, J. 1973. Quassinoid bitter principles. Fort. Chem. Org. Nat. 30: 101-150. [ Links ]
SAS Institute. 1985. SAS user guide: Statistics, Version 5 ed. SAS Institute Inc. Cary, North Carolina, USA. [ Links ]
Schabel, H., L. Hilje, K.S.S. Nair & R.V. Varma. 1999. Economic entomology in tropical forest plantations: An update. J. Trop. For. Sci. 11: 303-315. [ Links ]
Schoonhoven, L.M. 1980. Perception of azadirachtin by some lepidopterous larvae, p. 105-108. In H. Schmutterer, K. Ascher & H. Rembold (eds.). Natural pesticides from the neem tree (Azadirachta indica A. Juss). GTZ, Eschborn, Germany. [ Links ]
Soto, F., L. Hilje, G. Mora, M.E. Aguilar & M. Carballo. 2007. Systemic activity of plant extracts in Cedrela odorata (Meliaceae) seedlings and their biological activity on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae. Agric. & For. Entomol. 9: 221-226. [ Links ]
Speight, M.R. 1997. Forest pests in the tropics: Current status and future threats, p. 207-227. In A.D. Watt, N.E. Stork & M.D. Hunter (eds.). Forest and insects. Chapman & Hall, London, England. [ Links ]
Tomerlin, J. & T. Howell. 1988. Distrain: A computer program for training people to estimate disease severity on cereal leaves. Plant Dis. 72: 455-459. [ Links ]
Vargas, C., P. Shannon, R. Taveras, F. Soto & L. Hilje. 2001. Un nuevo método para la cría masiva de Hypsipyla grandella. Man. Integ. Plagas No. 62, Hoja Técnica No. 39. p. 1-4. [ Links ]
Villalobos, R. 1995. Distribución de Quassia amara L. ex Blom en Costa Rica, y su relación con los contenidos de cuasina y neocuasina (insecticidas naturales) en sus tejidos. Mag. Sci. Thesis CATIE, Turrialba, Costa Rica. [ Links ]
Warthen, J.D. & E.D. Morgan. 1990. Insect feeding deterrents, p. 23-134. In E.D. Morgan & N.B. Mandava (eds.). CRC Handbook of natural pesticides, Vol. 6: Insect attractants and repellents. CRC, Boca Raton, Florida. [ Links ]
Wilkins, R., G. Allan & R. Gara. 1976. Protection of Spanish cedar with controlled release insecticides, p. 63-70. In J.L. Whitmore (ed.). Studies on the shootborer Hypsipyla grandella (Zeller). Lep. Pyralidae. Vol. III. IICA Misc. Publ. No. 101. IICA, Turrialba, Costa Rica. [ Links ]
Correspondencia a: Francisco Soto. Graduate School. Tropical Agricultural Research and Higher Education Center (CATIE). Turrialba, Costa Rica; fsoto@catie.ac.cr
Luko Hilje & Manuel Carballo. Department of Agriculture and Agroforestry. Tropical Agricultural Research and Higher Education Center (CATIE). Turrialba, Costa Rica; lhilje@catie.ac.cr, mcarball@catie.ac.cr
Gerardo A. Mora. Associate Researcher. Natural Products Research Center (CIPRONA). Universidad de Costa Rica. San José, Costa Rica; gerardo.moralopez@ucr.ac.cr
Received 26-III-2010. Corrected 26-VIII-2010. Accepted 27-IX-2010.