Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.56 no.4 San José dic. 2008
Antifeedant activity of botanical crude extracts and their fractions on Bemisia tabaci (Homoptera: Aleyrodidae) adults: III. Quassia amara (Simaroubaceae)
Guillermo Flores1, Luko Hilje2, Gerardo A. Mora3 & Manuel Carballo2
1. Ministry of Agriculture and Animal Husbandry (MAG), Cartago, Costa Rica.
2. Department of Agriculture and Agroforestry, Tropical Agricultural Research and Higher Education Center (CATIE), Turrialba, Costa Rica, Fax (506) 558-2043; lhilje@catie.ac.cr
3. Associate Researcher. Natural Products Research Center (CIPRONA), Universidad de Costa Rica. San José, Costa Rica.
Abstract: Bemisia tabaci is a key virus vector on several crops worldwide. Then, a preventive approach to deal with viral epidemics may be the deployment of repellents or phagodeterrents at earlier stages of plant development (critical period). Thus, the crude extract and four fractions thereof (water, water: methanol, methanol, and diethyl ether) of bitterwood (Quassia amara, Simaroubaceae) were tested for phagodeterrence to B. tabaci adults under greenhouse conditions, on tomato plants, in Costa Rica. Both restricted-choice and unrestricted-choice experiments showed that the crude extract and some fractions exerted such effect on the insect. In the former case (in sleeve cages), all fractions caused deterrence at doses as low as 0.1 % (methanol), 0.5 % (water: methanol and diethyl ether), and 1% (water). In the latter case (plants exposed in a greenhouse) the methanol fraction stood out, followed by the diethyl ether fraction. Nonetheless, none of the fractions performed better than the deterrent control (mineral oil). Rev. Biol. Trop. 56 (4): 2131-2146. Epub 2008 December 12.
Key words: Bemisia tabaci, plant extracts, bitterwood, Quassia amara, phagodeterrence.
Bemisia tabaci (Gennadius) is probably the main agricultural pest worldwide, especially affecting tropical and subtropical cropping systems (Brown and Bird, 1992; Brown, 1994). Estimated economic losses amount to several hundreds or even thousands of millions of dollars a year, worldwide (Oliveira et al., 2001). Moreover, B. tabaci has at least 19 well-documented races or biotypes (Perring, 2001) which may vary in their degree of association with particular host plants and the induction of specific systemic disorders (syndromes), as well as their reproductive potential and response to climatic changes.
B. tabaci is a highly polyphagous pest, which can develop or reproduce on over 500 different plant species, distributed in 74 families (Greathead, 1986), and affecting some 30 cash and staple crops worldwide, such as tomato, peppers, melon, watermelon, soybean, cotton, beans and cassava. Crop damage may occur directly through excessive sap removal, or indirectly by promoting the growth of sooty mold, inducing syndromes through feeding, or by vectoring plant viruses (Schuster et al., 1996).
There are some 17 begomoviruses (family Geminiviridae), formerly classified as gemi niviruses, affecting tomato in the Americas (Polston and Anderson, 1997). Their impact on yields depends on plant age at time of infection: the earlier the infection, the greater the severity of the disease and its effect on yield (Schuster et al., 1996). The critical period of susceptibility of tomato for several begomoviruses encompasses the first 50-60 days after emergence (Franke et al., 1983; Schuster et al., 1996). In addition, it has been documented that the action threshold is very low (0.3 adults/ plant) in Costa Rica (Hilje, 2001). Therefore, a preventive approach should be sought, focusing on this critical early period to minimize contact between the vector and the host plant.
In Costa Rica, Hilje (1993, 2001) promoted a two-phase scheme for integrated management of whitefly-borne begomoviruses in tomato, such as the tomato yellow mottle virus (ToYMoV) and the Sinaloa Tomato Leaf Curl virus (STLCV), emphasizing production of virus-free seedlings for 30 days under tunnels covered by fine nets, and post-transplant protection by combinations of cultural practices, including living ground covers, which could be complemented with repellents or deterrents.
It has been either shown or suggested that several types of biorational substances could repel or deter B. tabaci adults or other members of the Aleyrodidae family (veierov, 1996), including both cooking and mineral oils, as well as plant extracts, such as those from the bitterwood tree (Quassia amara L. ex Blom, Simaroubaceae). Therefore, the objective of this research was to confirm previous results (Cubillo et al., 1997; Hilje and Stansly, 2001), as well as to test some of its fractions to gain insight into more specific groups of substances causing phagodeterrence.
Materials and methods
Location: Greenhouse experiments were carried out at CATIE, in Turrialba, within the Caribbean watershed of Costa Rica, at 9° 52 N, 83° 38Wand 640 m.a.s.l., with annual averages of 22 ºC, 2479 mm (rainfall) and 87% RH.
Extract preparation: Plant material was collected from a single location and at the same time, within the Keköldi Indian Reservation (Talamanca, Costa Rica), in order to avoid variability due to geographic or seasonal differences.
Hydroalcoholic extracts were prepared at the Natural Products Research Center (CIPRONA), Universidad de Costa Rica, as follows: wood chips were dried in an oven at 40 ºC, ground and extracted with 70% methanol in a suitable flask for 24 h; the solvent was drained and the residue was extracted again with methanol for 24 h. The pooled extracts were filtered through a Whatman No. 4 filter paper, and concentrated at 40 ºC using a rotary evaporator. The final residue was freeze-dried to eliminate any water remaining in the crude extract.
To fractionate the crude extract, a column 31 cm high and 4.5 cm diameter was filled with 170 g of the synthetic resin Diaion HP-20 (Mitsubishi Chemical Industry). The resin was washed with portions of water, water: methanol (1:1), methanol, and diethyl ether and then the order was reversed, prior to placing the sample on the column. A maximum of 10 g of the crude freeze-dried extract per 100 g of stationary phase (synthetic resin) (17 g in total) was placed on the column and eluted with 1 L each of the solvents, starting with water and finishing with diethyl ether. The column was used three times to completely process one batch of 51 g of crude extract. The stationary phase was washed reversing the solvents each time it was reused. The solvents were evaporated and freeze-dried, if necessary, to give 18.04 g (35.37% of crude extract), 3.80 g (7.45% of crude extract), 18.10 g (35.49% of crude extract) and 3.43 g (6.73% of crude extract), respectively, for the water, water: methanol, methanol and diethyl ether fractions.
The dry weight of each fraction was used to determine its equivalent dose with respect to the weight of the crude extract. A description of the procedure used is shown in the next section.
Both the freeze-dried crude extract and the corresponding fractions were kept hermetic, refrigerated and in darkness, to avoid their chemical decomposition or contamination by fungi.
Preparation of fraction solutions: Initially the crude extract was evaluated for biological activity at 0.1, 0.5, 1.0, and 1.5% w/v (weight/volume); so, the fractions were evaluated at the corresponding concentrations, equivalent to those of the crude extract, according to the relative concentration of the fraction in the crude extract. In the text, we refer to the concentrations of the fractions as 0.1, 0.5, 1.0, and 1.5% w/v but these are equivalent concentrations. The real fraction concentration is different, as described below. To prepare the solutions, the amount required for each fraction, in accordance to the yield of the fractionation process, was weighed and dissolved into 100 mL of the respective solvent.
Thus, eight treatments (with four replicates each) were obtained, corresponding to four concentrations (0.1, 0.5, 1.0 and 1.5% w/v) of each of the four fractions. Therefore, such treatments (w/v) were: 0.03537%, 0.17685%, 0.3537% and 0.5306% (water fraction), 0.00745%, 0.03725%, 0.0745% and 0.11175% (water: methanol fraction), 0.03708%, 0.1854%, 0.3708% and 0.5562% (methanol fraction), and 0.00672%, 0.0336%, 0.0672% and 0.1008% (diethyl ether fraction).
A stock solution was prepared at the highest concentration; other solutions were prepared by dilution. In the case of the fractions with organic solvents (methanol and diethyl ether) it was necessary to change solvents, because of phytotoxicity. It was found that, when methanol or diethyl ether was used, necrosis of the leaves occurred. Thus, the methanol fraction was dissolved in 20% methanol in water, and the diethyl ether fraction was dissolved in 20% acetone in water. These mixtures were found to be the least toxic, most compatible solvents for each fraction. The solvent for the water: methanol fraction was maintained as 50% methanol in water.
All solutions were prepared just before the experiment was set up, with distilled water as a carrier.
Experiments: Both the crude extract and four fractions (water, water: methanol, methanol, and diethyl ether) were assessed for their feeding deterrence and oviposition response on B. tabaci adults of the A biotype. Insects were taken from a colony kept in a greenhouse at CATIE, and reared on a mixture of eggplant and tomato plants.
The crude extract and its fractions were tested by means of restricted-choice experiments (in a closed space), whereas only the most promising doses of each fraction were compared through an unrestricted-choice experiment (in an open space).
Restricted-choice experiments: In this case, adult whiteflies were exposed to a treated and an untreated potted tomato plant within a sleeve cage, so that they had to choose between two alternatives or remain flying and resting on the cage´s walls.
A first experiment was run with a crude freeze-dried extract, aiming at confirming previous results by Cubillo et al. (1997) and Hilje and Stansly (2001), as well as determining the minimum dose at which deterrence could be detected, to use it as reference for further experiments. Then an experiment was run for each of the four fractions, tested at the equivalent of such a dose along with the respective solvents, as controls, to rule out any effects of them on B. tabaci adults.
For these restricted-choice experiments, two pots, each one with a tomato plant, were placed in a sleeve cage. One of the plants was sprayed with either the crude extract, each one of its fractions or the control solvents, whereas the other was treated with distilled water. Experiments were run using a completely randomized design (as environmental conditions were quite homogenous inside the greenhouse where cages were located), with four replicates, the experimental unit being represented by each potted plant receiving a given treatment.
The crude extract, as well as each one of its fractions were tested in individual experiments at the doses of 0.1, 0.5, 1.0 and 1.5% w/v, or their equivalent ones (see above). They were compared with an insecticide (endosulfan), a control treatment (distilled water), and Aceite Agrícola 81 SC, which is an agricultural oil that strongly deters whitefly adults (Hilje, unpubl.), and the emulsifier Citowett. Endosulfan (Thiodan 35% CE; Hoechst, Germany) (350 g a.i./L) was used at its recommended commercial dose (2.5 mL/L water), as was Aceite Agrícola 81 SC (1.5% v/v) (Mobil Oil Corp., Memphis, Tennessee). Citowett (BASF, Germany) was added to all treatments at the same dosage (0.25 mL/L, i.e., 0.025%, which is half of what is commercially recommended) to improve adherence to foliage.
In all cases, treatments were applied to tomato plants (var. Hayslip) with three true-leaves. This was done with a Devilbiss 15 hand-sprayer, with an adjustable tip (The Devilbiss, Somerset, Pennsylvania), which was connected to an air pump, under a constant pressure (10 kg/cm2). Plants from each treatment were sprayed separately with each solution in an isolated room, for which they were placed on a table and thoroughly sprayed to run-off.
Treated plants were introduced into sleeve cages (30 cm x 30 cm x 45 cm, with walls made of wood, a fine net, and glass) 30 min after being sprayed. Fifty B. tabaci adults were collected from the colony with a hand aspirator and released into each cage. Release took place between 8:30-10:30 h; 2 min later, the aspirator flask was checked, in order to count and release additional adults to replace those which had died due to handling.
Unrestricted choice experiments: In this case, tomato plants were exposed to flying whiteflies inside a greenhouse where their colony is maintained, so that they had no restrictions in choosing where to settle.
These experiments were performed for the lowest doses causing deterrence in the previous experiments: diethyl ether (0.1%), water (0.5%) and water: methanol (0.5%). They were compared with the same controls, except for endosulfan, in order not to disturb colony development.
Potted tomato plants (treated as in the restricted-choice experiments) were placed on a bench and arranged in a randomized complete block design, with four replicates. The experimental unit was represented by each potted plant receiving a given treatment.
Analysis: For the restricted-choice experiments, counts were made on the foliage of the whole plant. The criterion to appraise feeding deterrence was the number of adults on each plant at 48 h, in combination with the number of those surviving within that interval. Oviposition response was appraised by counting the number of eggs laid over 48 h on one leaf per plant; eggs were counted under a stereo-microscope.
Mortality was determined by counting the total number of living adults in each cage (on both plants) at 48 h, and subtracting the sum from 50; in this case, the experimental unit was represented by each cage (both two potted plants), as adult whiteflies are very mobile and would be expected to make contact with both plants and get killed if substances present in a given treatments are toxic.
For the unrestricted-choice experiments counts were also made on the foliage of the whole plant 1, 2, 8 and 15 days after plant treatment. Nymph numbers (instars I and II) were counted at the end of the experiment by counting a 1 cm2 square on one of the leaves, under a stereo-microscope.
To determine feeding deterrence and oviposition response for both types of experiments, the number of either adults or eggs present on each plant within each cage was subjected to analysis of variance (ANOvA), and mean values were compared using a Duncan´s test (SAS Institute, 1985), at a significance level of α = 0.05. For the purpose of determining mortality, the total numbers of living adults in each cage were also subjected to ANOvA and mean values were compared using a Duncan´s test.
Since the coefficients of variation were high (> 20%) for data resulting from the unrestricted-choice experiments, they were transformed by the logarithmic method, in order to stabilize variance and meet the assumptions underlying ANOVA. In all cases, such assumptions were verified.
Results
Restricted-choice experiments: Because of the nature of the experiments, comparisons about feeding deterrence and oviposition response were valid only between both plants within each cage, and not between cages. However, for mortality, comparisons were valid between cages.
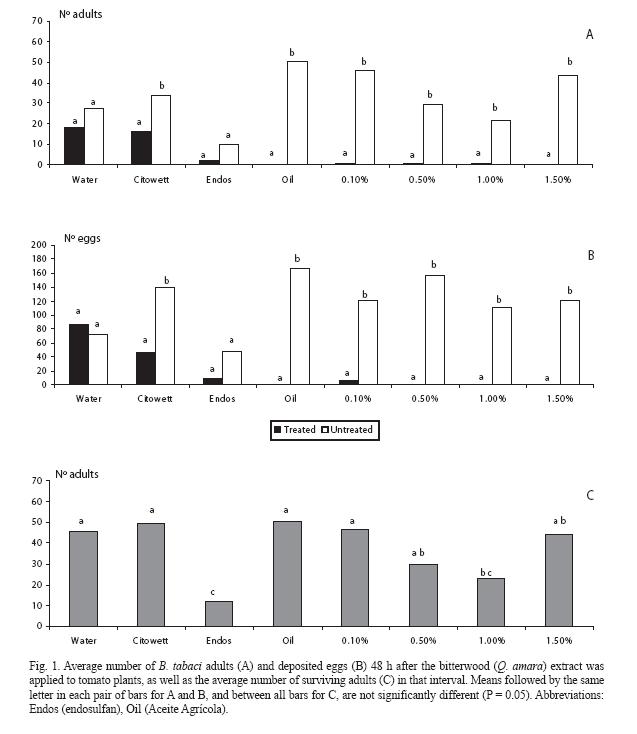
For the freeze-dried crude extract, there was a sharp contrast in the low number of adults that landed on the treated plant for all doses, as compared to the corresponding controls (p < 0.05) (Fig. 1A); the same also occurred with the mineral oil. In the extreme treatments (water and endosulfan) there was no difference between both plants (p > 0.05) but, in the latter case, adult numbers strongly decreased in both plants. In the Citowett treatment there was an unexpected difference between both plants (p < 0.05). Similar trends were observed for the oviposition response (Fig. 1B).
Regarding mortality, plants treated with endosulfan had the lowest number of surviving adults, in contrast with almost all the crude extract treatments (Fig. 1C), which in general did not differed among them (p > 0.05) nor with the water control.
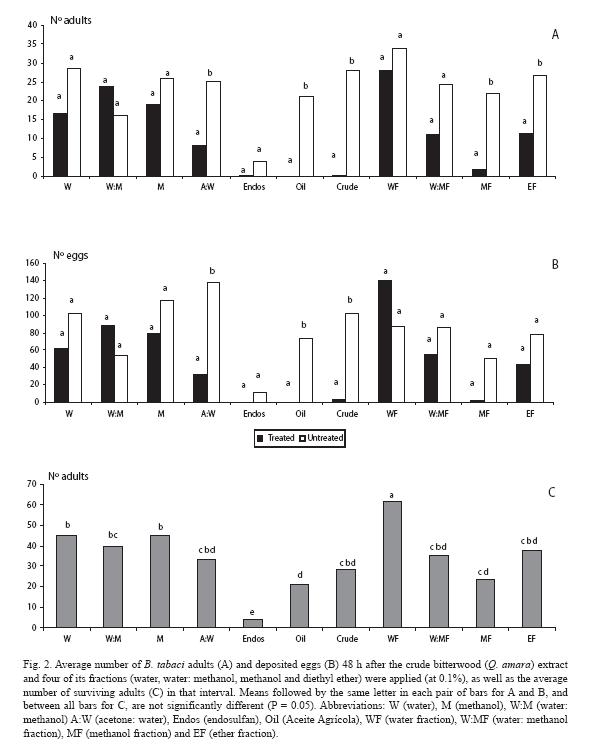
In the following experiment, the lowest dose of the crude extract causing deterrence (0.1%) was selected as a baseline reference. The fewest adults were counted on the plant treated with the methanol and ether fractions (Fig. 2A), as well as for the crude extract, the mineral oil, and one of the control treatments (acetone: water). Similar trends were observed for the oviposition response, except for the methanol and ether fractions (Fig. 2B); however, there were fewer eggs in those plants treated with these fractions, as well as with the water: methanol fraction. As for mortality, there was a sharp contrast between endosulfan and the rest of treatments (Fig. 2C).
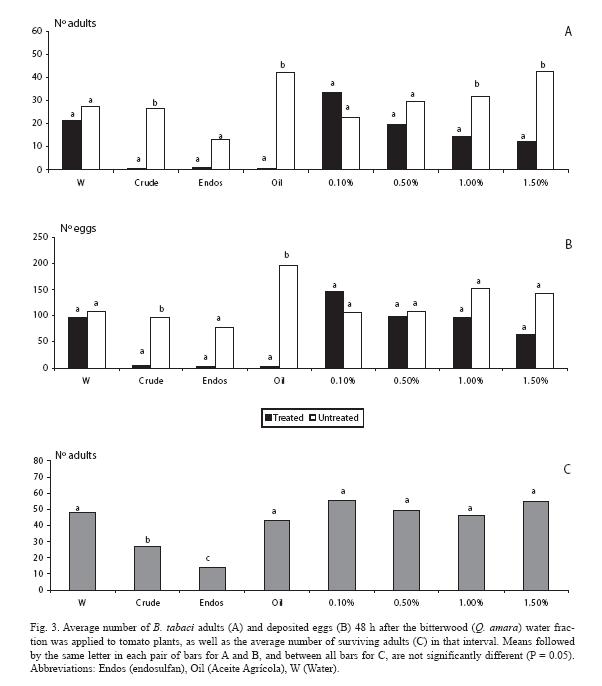
When analyzing the water fraction, there were fewer adults (p < 0.05) at the two highest doses (1.0 and 1.5%), as well as in the mineral oil and the crude extract (Fig. 3A). Trends held for the oviposition response, but at the two highest doses there were no differences (p > 0.05) (Fig. 3B), although there were fewer eggs in those plants treated with these fractions. Concerning mortality, endosulfan clearly contrasted with the rest of treatments (Fig. 3C).
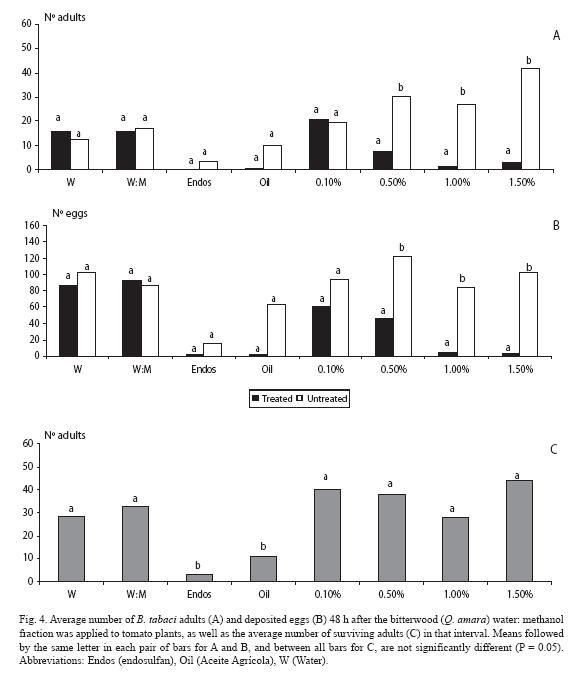
For the water: methanol fraction, fewer adults landed (p < 0.05) at the three highest doses (0.5, 1.0 and 1.5%) (Fig. 4A). In the case of the mineral oil there was an unexpected lack of difference between plants (p > 0.05). Trends exactly held for the oviposition response, but at the highest dose there was no difference (p > 0.05), although there were fewer eggs in the plant treated with that fraction (Fig. 4B). As for mortality, endosulfan clearly contrasted with the rest of treatments, except for the mineral oil (Fig. 4C).
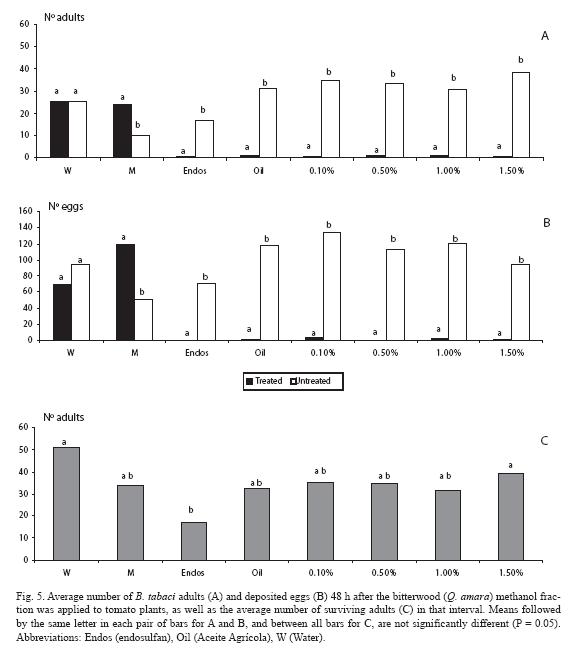
For the methanol fraction there was a sharp contrast in the low number of adults on the treated plant for all doses (p < 0.05), as well as with the mineral oil (Fig. 5A). In the water control treatment there was no difference between plants (p > 0.05). Unexpectedly, even more adults were counted on the solvent treated plants than on the untreated ones (p < 0.05). Similar trends were observed for the oviposition response (Fig. 5B). Concerning mortality (Fig. 5C), none of the four doses differed from the control treatments nor from the mineral oil (p > 0.05), whereas the three lowest doses (0.1, 0.5 and 1.0%) did not differed from endosulfan nor from the solvent and the mineral oil.
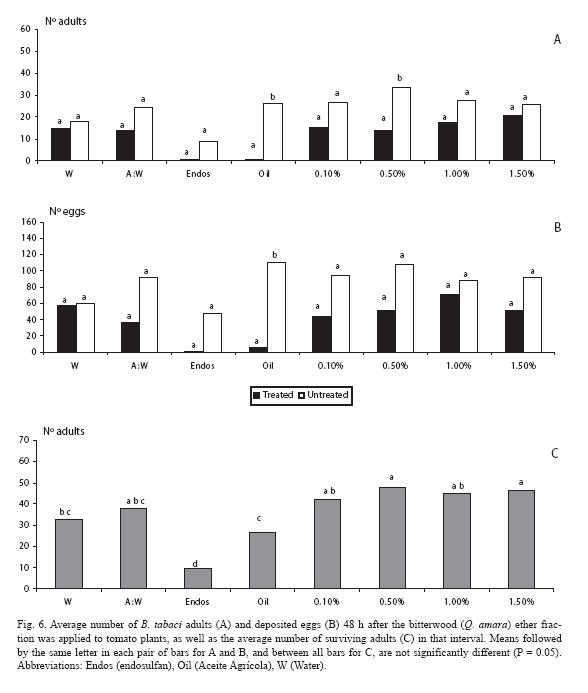
For the diethyl ether fraction, there were fewer adults (p < 0.05) only at one of the doses (0.5%), as it also occurred with the mineral oil (Fig. 6A). Such trends in general held for the oviposition response (Fig. 6B). As for mortality, endosulfan neatly contrasted with the rest of treatments (Fig. 6C).
Unrestricted choice experiments: In this case, comparisons about feeding deterrence and nymphal development were valid between treatments, as all of them were equally exposed to the same group of adult whiteflies.
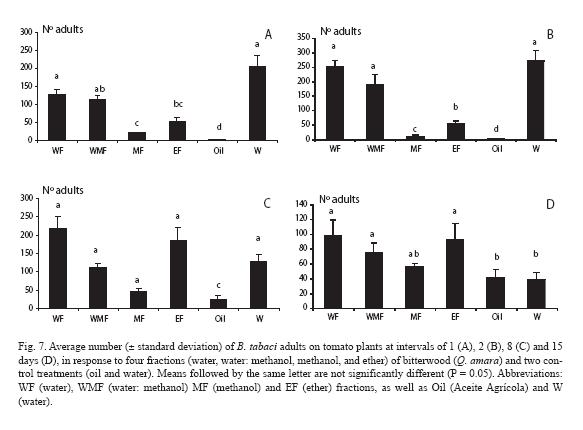
For the first count, 24 h after the fractions were sprayed, there was a very strong contrast between all treatments and the mineral oil, which yielded only one adult (Fig. 7A). The methanol fraction was the one that performed better, closely followed by the diethyl ether fraction. Such trends held exactly a day later, with adult numbers increasing for all treatments, whereas only two adults were counted in the mineral oil treatment (Fig. 7B). A week later (Fig. 7C) the same pattern was observed, but numbers varied somewhat, depending on the treatments. Finally, two weeks later in general there were no differences among treatments (p > 0.05) and adult numbers strongly decreased in the water control (Fig. 7D).
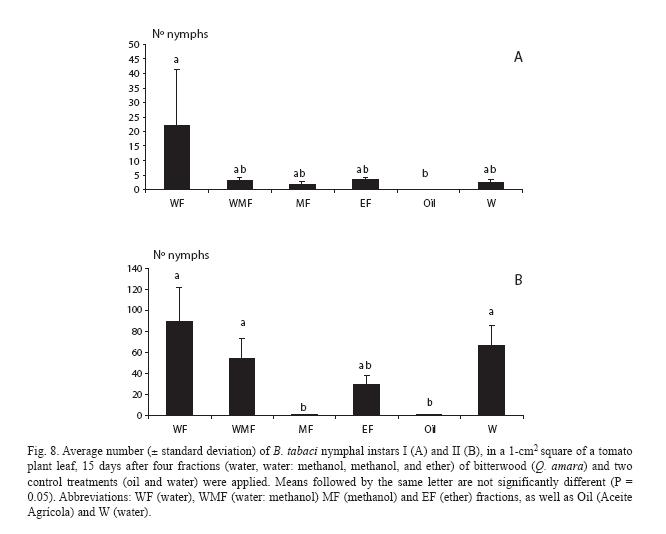
Regarding nymph numbers, for instar I they were lowest for plants treated with the mineral oil, even though they did not differ from the rest of the treatments, except for the water fraction (Fig. 8A). However, for instar II they strongly increased for all treatments, except the mineral oil and the methanol fraction (Fig. 8B).
Discussion
Aspects concerning B. tabaci adults ability to react to plant substances (Walker and Gordh 1989) as well as the types of allomones to which they could respond (Warthen and Morgan 1990) and how their effects could be recognized (Walker and Perring 1994) have been discussed elsewhere (Flores et al. 2008).
Our clear-cut findings with the freeze-dried crude extract confirm preliminary results from other authors (Cubillo et al., 1997; Hilje and Stansly 2001) concerning the existence of phagodeterrent principles for B. tabaci in bitterwood extracts. The lower number of landed adults on tomato plants after treating them with the extract was an expression of feeding deterrence. It would be expected that once adults land on a treated plant make contact with the deterrent substances, so that over time they tended to gather on the untreated plant. Feeding deterrence by bitterwood extracts has also been demonstrated for the mahogany shootborer, Hypsipyla grandella (Lepidoptera: Pyralidae), a key neotropical forest pest (Mancebo et al., 2000).
The coincident pattern of lower oviposition response on treated plants could be explained by the lower number of females present on them, rather than in terms of oviposition deterrence, although the latter cannot be ruled out. Likewise, nymph numbers were probably a direct expression of female abundance and egg-laying activity for each treatment.
Moreover, the experiment at which the lowest dose of the crude extract causing deterrence (0.1%) was compared to the fractions at the equivalent dose, revealed that the former exerted a more distinct phagodeterrent effect than any of the fractions, and suggests that there is some synergism between some substances present in the crude extract but not in any of the particular fractions. Warthen and Morgan (1990), summarizing findings from other authors, state that when pure deterrent compounds are tested individually at the same concentration at which they occur in a plant, they are less active than the plant itself, but when all individual components are combined there is an effect producing a deterrence comparable to the effect produced by the plant itself.
Nevertheless, testing of fractions to gain insight into more specific groups of substances responsible for phagodeterrence, showed that methanol and water: methanol fractions provoked a stronger response by whitefly adults, which occurred at doses as low as 0.1 and 0.5%, respectively, the same pattern holding for oviposition. Deterrence could be explained by the presence of quassinoids, such as quassin and neoquassins, which are common in Q. amara (Polonsky, 1973). These compounds are known to be more soluble in methanol and ethanol than in diethyl ether (The Merck Index, 2001). For example, Leskinen et al. (1984) found that quassin from Q. amara deters feeding by Epilachna varivestis (Coleoptera: Coccinellidae).
When tomato plants treated with each of the bitterwood fractions were exposed to flying whiteflies in the greenhouse, the methanol fraction performed better than the rest, closely followed by the ether fraction, but none of them did better than the mineral oil, a trend that lasted for only 48 h. Within a week, none of these fractions performed satisfactorily, which suggests that the deterrent principles decompose under the experimental conditions; low numbers in the water control were possibly due to plant deterioration, so that adults moved to neighbor plants.
Quassinoids are not volatile, but probably are decomposed by the air and/or light. These quassinoids are interchangeable, as quassin can be reduced to the neoquassins and the neoquassins oxidized to quassin, and can undergo several transformations catalized by mild acidic conditions (Polonsky, 1973). Other components of Q. amara which could have a similar activity are some indole alkaloids of the canthin-6-one type, mainly present in the leaves (Sáenz and Nassar, 1970) but which can be found in the wood (Barbetti et al., 1990; Coe and Anderson, 1996), especially if the preparation of the sample included some bark. These compounds can also be decomposed by air and/or light.
Adult mortality observed in treated plants, which was dose-independent, may be attributed either to an indirect effect of strong deterrence, causing heat stress, energy depletion or dehydration (veierov, 1996), or to direct toxicity of the crude extract or the fractions to very susceptible adults, as insecticidal properties of bitterwood have been widely documented (Metcalf et al., 1951).
In some of the experiments there were occasional inconsistencies regarding adult responses to some of the control treatments, as well as lack of the expected dose-dependent responses to some extract fractions. They could be explained in terms of a great deal of variability between replicates of each treatment, especially due to adult age and vigor, different sex ratios in each replicate, etc., which were factors not under control.
Deterrent activity by bitterwood derivatives would make it possible to eventually formulate a commercial product to be deployed during the critical period of susceptibility to begomoviruses and other viruses affecting tomato and other vegetables, to complement other tactics, as semiochemicals by themselves very seldom provide robust pest control (Pickett et al. 1997). In addition, they offer a unique opportunity for the agrichemical industry in terms of safety, as they would not pose risks to humans and other mammals. Bitterwood is a traditional natural medicine in some neotropical countries, and has given rise to Q-assia, the brand of a new pharmaceutical product for digestive problems recently released into the market by Lisanatura, a local pharmaceutical company in Costa Rica.
It has been shown that the aqueous extract of the dry wood of bitterwood was capable of preventing the formation of ulcers on rats, as induced by indomethacin, ethanol, or stress (Badilla et al., 1998). In a previous study, no sign of acute toxicity was observed at any oral dose of an aqueous extract of the wood; however, the intraperitoneal administration of 500 mg/kg, presented acute toxicity signs with a 24 h recovery, but the 1000 mg/kg dose was lethal to a 100% within 24 h (García et al. 1996). The crude methanol extract of the stem wood caused significant reduction in the weight of the testis, epididymis and seminal vesicle, but an increase in weight of the anterior pituitary gland. Quassin produced similar biological actions as the crude extract while the effects of 2-methoxycanthin-6-one did not seem to differ from those of the control (Raji and Bolarinwa, 1997). Quassin was also shown to inhibit the synthesis of testosterone in rat Leydig cells in a dose-dependent fashion (Njar et al. 1995).
Industrialization of products from Q. amara could become a model of wise utilization of neotropical forest biodiversity, and there is a growing interest in promoting the utilization of this shrub as an economic resource for indigenous communities in Central America. Therefore, several of its ecological, silvicultural, and marketing aspects have been researched in recent years (Ocampo, 1995). Its range extends from Mexico to Ecuador, including the Caribbean basin, where it normally grows in the forest understory, but it also grows easily in disturbed areas (villalobos, 1995). Since it can be established as a plantation tree, Bougainvillea S.A., a Costa Rican company (Rafael Ocampo 2007, personal communication) is making a systematic effort for domesticating the shrub, developing harvesting practices compatible with the conservation of the species and assurance of a high content of quassinoids, and optimizing the extraction conditions to produce an industrial extract for insecticidal purposes.
Acknowledgments
This paper is a partial result of a CATIEs M.Sc. thesis by the first author, which was partially funded by the Consejo Nacional de Investigaciones Científicas y Tecnológicas (CONICIT) and UMCRE, from the Instituto Costarricense de Electricidad (ICE). Thanks are due to Arturo Ramírez and Gustavo López (CATIE) for their logistical support, to Juan Carlos Brenes (CIPRONA) for preparing the extracts for the experiments and to Norman Farnsworth (Chief Editor of NAPRALERT), for granting access to the database.
Resumen
En el plano mundial, Bemisia tabaci es un importante vector de virus en numerosos cultivos. Un enfoque preventivo para enfrentar las epidemias virales podría ser el empleo de sustancias repelentes o fagodisuasivas en las etapas tempranas del desarrollo de las plantas (período crítico). Tanto el extracto crudo como cuatro fracciones (agua, agua: metanol, metanol, y éter dietílico). Evaluamos extractos del árbol llamado "hombre grande" (Quassiaamara, Simaroubaceae) fueron evaluadas en cuanto a su actividad fagodisuasiva sobre los adultos de B. tabaci, usando tomate, en condiciones de invernadero, utilizando plantas de tomate, en Turrialba, Costa Rica. Tanto los experimentos de escogencia irrestricta e irrestrictam, el extracto crudo y algunas fracciones mostraron dicha actividad. En los primeros experimentos (en jaulas de manga), todas las fracciones causaron fagodisuasión a dosis de apenas 0.1% (metanol), 0.5% (agua: metanol y éter dietílico) y 1% (agua). En los segundos (plantas expuestas dentro de un invernadero) sobresalió la fracción metanólica, seguida por la de éter dietílico. Sin embargo, ninguna de las fracciones superó al testigo fagodisuasivo (aceite mineral).
Palabras clave: Bemisia tabaci, extractos vegetales, hombre grande, Quassia amara, fagodisuasión.
Received 16-II-2007. Corrected 24-III-2008. Accepted 31-VII-2008.
References
Badilla, B., T. Miranda, G. Mora & K. vargas. 1998. Actividad gastrointestinal del extracto acuoso bruto de Quassia amara (Simarubaceae). Rev. Biol. Trop. (Costa Rica) 46(2): 203-210. [ Links ]
Barbetti, P., G. Grandolini, G. Fardella & I. Chiappini. 1987. Indole alkaloids from Quassia amara. Planta Med. 53: 289-290. [ Links ]
Brown, J.K. 1994. Current status of Bemisia tabaci as a plant pest and virus vector in agroecosystems worldwide. FAO Plant Prot. Bull. 42: 3-32. [ Links ]
Brown, J.K. & J. Bird. 1992. Whitefly-transmitted geminiviruses in the Americas and the Caribbean Basin: Past and present. Plant Dis. 76: 220-225. [ Links ]
Coe, F.G. & G.J. Anderson. 1996. Screening of medicinal plants used by the Garifuna of Eastern Nicaragua for bioactive compounds. J. Ethnopharmacol. 53: 29-50. [ Links ]
Cubillo, D., G. Sanabria & L. Hilje. 1997. Mortalidad de adultos de Bemisia tabaci con extractos de hombre grande (Quassia amara). Man. Integr. Plagas (Costa Rica) 45: 25-29. [ Links ]
Flores, G., L. Hilje, G.A. Mora & M. Carballo. 2008. Antifeedant activity of botanical crude extracts and their fractions on Bemisia tabaci (Homoptera: Aleyrodidae) adults: I. Gliricidia sepium (Fabaceae). Rev. Biol. Trop. 56 (4): 2099-2113. [ Links ]
Franke, G., L. van Balen & E. Debrot. 1983. Efecto de la época de infección por el mosaico amarillo sobre el rendimiento del tomate. Rev. Fac. Agron. Univ. Zulia (venezuela) 6: 741-743. [ Links ]
García, M., S.M. González & L. Pazos. 1996. Actividad farmacológica del extracto acuoso de madera de Quassia amara (Simarubaceae) en ratas y ratones albinos. Rev. Biol. Trop. (Costa Rica) 44/45: 47-50 [ Links ]
Greathead A.H. 1986. Host plants. p. 17-26. In M.J.W. Cock (ed.). Bemisia tabaci. A Literature Survey. CAB Int. Inst. of Biol. Control, Silwood Park, UK. [ Links ]
Hilje, L. 1993. Un esquema conceptual para el manejo integrado de la mosca blanca (Bemisia tabaci) en el cultivo del tomate. Man. Integr. Plagas (Costa Rica) 29: 51-57. [ Links ]
Hilje, L. 2001. Avances hacia el manejo sostenible del complejo Bemisia tabaci-geminivirus en tomate, en Costa Rica. Man. Integr. Plagas (Costa Rica) 61: 70-81. [ Links ]
Hilje, L. & P.A. Stansly. 2001. Development of crop associations for managing geminiviruses vectored by whiteflies in tomatoes. Final Report. U.S. Dept. Agr. (USDA). CATIE. Turrialba, Costa Rica. 134 p. [ Links ]
Leskinen, V., J. Polonsky & S. Bhatnagar. 1984. Antifeedant activity of quassinoids. J. Chem. Ecol. 10 (10): 14971507. [ Links ]
Mancebo, F., L. Hilje, G.A. Mora & R. Salazar. 2000. Antifeedant activity of Quassia amara (Simaroubaceae) extracts on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae. Crop Prot. 19: 301-305. [ Links ]
Metcalf, C.L., W.P. Flint & R.L. Metcalf. 1951. Destructive and useful insects. 3 ed. McGraw-Hill, New York. 1208 p. [ Links ]
Njar, V.C., T.O. Alao, J.I. Okogun, Y. Raji, A.F. Bolarinwa & E.U. Nduka. 1995. Antifertility activity of Quassia amara: quassin inhibits the steroidogenesis in rat Leydig cells in vitro. Planta Med. Apr. 61:180-182. [ Links ]
Ocampo, R.A. (Ed.), 1995. Potencial de Quassia amara como insecticida natural. Serie Técnica. Informe Técnico No. 267. CATIE. Turrialba, Costa Rica. 185 p. [ Links ]
Oliveira, M.R.V., T.J. Henneberry & P. Anderson. 2001. History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot. 20: 709-723. [ Links ]
Perring, T.M. 2001. The Bemisia tabaci species concept. Crop Prot. 20: 725-737. [ Links ]
Pickett, J.A., L.J. Wadhams & C.M. Woodcock. 1997. Developing sustainable pest control from chemical ecology. Agric. Ecosyst. Environ. 64: 149-156. [ Links ]
Polonsky, J., 1973. Quassinoid bitter principles. Fortsch. Chem. Org. Naturst. 30: 101-150. [ Links ]
Polston, J.E. & P.K. Anderson. 1997. The emergence of whitefly-transmitted geminiviruses in tomato in the Western Hemisphere. Plant Dis. 81: 1358-1369. [ Links ]
Raji, Y. & A.F. Bolarinwa. 1997. Antifertility activity of Quassia amara in male rats -in vivo study. Life Sci. 61: 1067-74. [ Links ]
Sáenz, J.A. & M. Nassar.1970. Phytochemical screening of Costa Rican plants: Alkaloid analysis. Iv. Rev. Biol. Trop. 18: 129-138. [ Links ]
SAS Institute, 1985. SAS User Guide: Statistics, version 6 Edition, SAS Institute Inc, Cary, North Carolina. 956 p. [ Links ]
Schuster, D.J., P.A. Stansly & J.E. Polston. 1996. Expressions of plant damage of Bemisia. p. 153-165. In D. Gerling & R.T. Mayer (eds.). Bemisia 1995: Taxonomy, Biology, Damage, Control and Management. Intercept, Andover, UK. [ Links ]
The Merck Index (13th Edition). Merck & Co., Inc., Whitehouse Station, New Jersey, USA. 2001. Monographs 6491 and 8115. [ Links ]
Veierov, D., 1996. Physically and behaviorally active formulations for control of Bemisia. p. 557-576. In D. Gerling & R.T. Mayer (eds.). Bemisia 1995: Taxonomy, Biology, Damage, Control and Management. Intercept, Andover, UK. [ Links ]
Villalobos, R., 1995. Distribución de Quassia amara L. ex Blom en Costa Rica, y su relación con los contenidos de cuasina y neocuasina (insecticidas naturales) en sus tejidos. M.Sc. Thesis. CATIE. Turrialba, Costa Rica. 207 p. [ Links ]
Walker, G.P. & G. Gordh. 1989. The occurrence of apical labial sensilla in the Aleyrodidae and evidence for a contact chemosensory function. Entomol. Exp. Appl. 51: 215-224. [ Links ]
Walker, G.P. & T.M. Perring. 1994. Feeding and oviposition behavior of whiteflies (Homoptera: Aleyrodidae) interpreted from AC electronic feeding monitor waveforms. Ann. Entomol. Soc. Amer. 87: 363-374. [ Links ]
Warthen, J.D. & E.D. Morgan. 1990. Insect feeding deterrents. p. 23-134. In E.D. Morgan & N.B. Mandava (eds.). CRC Handbook of Natural Pesticides, Vol. 6: Insect Attractants and Repellents. CRC Press, Boca Raton, Florida. [ Links ]












 uBio
uBio