Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.55 no.3-4 San José sep./dic. 2007
Prey composition in the carnivorous plants Utricularia inflata and U. gibba (Lentibulariaceae) from Paria Peninsula, Venezuela
Elizabeth Gordon1 & Sergio Pacheco2
1 Instituto de Zoología Tropical, Escuela de Biología, Facultad de Ciencias, Universidad Central de Venezuela, A.P. 47058, Caracas 1041, Venezuela; egordon@strix.ciens.ucv.ve
2 Escuela de Biología, Facultad de Ciencias, Universidad Central de Venezuela, A.P. 47058, Caracas 1041, Venezuela
Abstract: Carnivorous aquatic plants, genus Utricularia (Lentibulariaceae), capture small aquatic organisms, such as rotifers, copepods, and cladocerans, by means of anatomical structures named bladders. The present study aimed to determine prey size and composition in U. gibba and U. inflata, which were collected from a small lake and an herbaceous wetland, respectively, located in Paria Peninsula (Sucre State, Venezuela). Water pH, conductivity, dissolved oxygen, and salinity were measured in situ at each sampling location, and water samples were collected to determine N-Kjeldahl, total-P, Na+, K+, Ca++, Mg++, and Cl-. Fifty bladders from each plant species were measured and their contents were analyzed. N-Kjeldahl and total-P values were similar in both sites, and were also similar to values reported for eutrophic ecosystems, although Na+, K+, Ca++, Mg++ concentrations and in situ water parameter values were higher in the herbaceous wetland. Bladder content showed the following zooplankton groups: rotifers, cladocerans, copepods, annelids, rhizopodeans, and insects; and the following phytoplankton divisions: Bacillariophyta, Chlorophyta, Cyanophyta, and Euglenophyta. U. inflata presented smaller and fewer bladders, but higher abundance and total algal and animal morphospecies richness than U. gibba. Prey composition similarity at the taxon level between the two carnivorous species was low. Rev. Biol. Trop. 55 (3-4): 795-803. Epub 2007 December, 28.
Key words: Lentibulariaceae, Utricularia, carnivorous plant, prey composition, prey capture, plankton, Venezuela.
Most carnivorous plants live in mineral-poor and acidic environments, and absorb a portion of their nutrients from dead animals through a variety of morphological, physiological, and/or behavioral adaptations that let them attract, capture, and digest their prey (Schnell 1976, Slack 1980, Knight 1992, Ellison and Gotelli 2001). Lentibulariaceae, one of several carnivorous plant families, includes the genera Genlisea, Pinguicula, Polipompholyx, and Utricularia, which are distributed throughout tropical and temperate regions. The genus Utricularia makes up 42 % of all plants classified as carnivorous (Juniper et al. 1989) with over 214 known species (Taylor 1989). Although occurring worldwide, including the Arctic Circle, the vast majority of species live in tropical and subtropical regions, particularly seasonally wet and with high or very high rain-fall (Taylor 1989).
Utricularia plants present flat bladders or utricles of varied shapes (oval, circular) with bristles or trichomes on the rhizoids, stolons, or foliar segments (Cook 1996), which help capture a wide variety of small aquatic animals (Friday 1989). According to life form, Utricularia plants can be classified as rootless free-submerged, free-floating with specialized floaters or soil-attached through root-like organs (rhizoids), and emergent; and according to their life cycle, as annual or perennial (Cook 1996).
The ecological importance of Utricularia plants resides in its interaction with other elements of the trophic chain, such as insects and crustaceans. Bladders capture prey by suction, a process activated when bristles are touched, apparently guiding the prey into the trap (Pompêo and Bertuga 1996). Lloyd (1942) and Sculthorpe (1967) described the bladders opening and closing mechanism. Some authors have claimed that prey slowly asphyxiates inside the trap and bacteria decompose it later (Sculthorpe 1967), others have suggested the prey is digested with enzymes or other digestive substances (Hegner 1926, Heslop-Harrison 1978).
Although the carnivory of these plants has been known for a long time (Pietropaolo and Pietropaolo 1986), intensive research to discover the ecology of Utricularia has been undertaken only recently. New studies include morphological aspects of the species that conform the genus Utricularia (Rutishauser and Brugger 1992, Sattler and Rutishauser 1992, Rutishauser 1993), the time it takes to capture their prey, the cost of their carnivory, their investment on carnivory and the quantification of number, size and biomass of the bladders (Friday 1989, 1992, Knight and Frost 1991, Knight 1992, Pompêo and Bertuga 1996). It is known that the bladders catch small aquatic animals such as rotifers, copepods, ostracods, cladocerans, ciliates, gastrotriches and chrinomids (Friday 1989, Knight and Frost 1991, Pompêo and Bertuga 1996, Mette et al. 2000) from which the plants absorb N and P (Friday and Quarmby 1994). Botta (1976), Mette et al. (2000) and Richards (2001) also reported the presence of algae, from divisions such as Cyanobacteria, Chrysophyta, Euglenophyta, Chlorophyta and Pyrrophyta. Baumgartner (1987) studied the role of U. vulgaris L. as a predator of late instar of Culex pipiens L., 1758 and its contribution as a potential form of biocontrol. Angerilli and Beirne (1980) studied the influence of the aquatic plants U. minor L, Lemna minor L, and Elodea canadensis Rich. on the colonization of artificial ponds by mosquitoes and their insect predators.
Few studies exist on Utricularia ecology in general and on prey composition in tropical species. In the case of Venezuela, only a taxonomic description of species has been carried out (Velásquez 1994). Our study intended to determine the scarcely known bladder prey content in U. inflata Walter and U. gibba L., and characterize the physicochemical environment where these plants grow.
Materials and methods
Plants were collected in southern Paria Peninsula (Libertador Municipality, Sucre State) in the localities of Bohordal, located 36 masl at 10º3526.8 N, 62º5618.8 W, and Rio de Agua, 18 masl at 10º3426.5 N, 62º5913.5 W. Locations present Agwi type climate, according to the Köeppen classification system, characterized by two well-defined (dry and rainy) seasons (MARN 1985). Total annual precipitation during the study year (2000) was 1 471 mm, with maximum values between June and December. Average temperature ranged between 24 °C and 27 °C.
The small lake at Bohordal had an average depth of 59.8 cm with a maximum depth of 99 cm in January and a minimum of 30-40 cm in May. Arboreal vegetation surrounded the dark-watered lagoon, providing a shady environment. Free-floating Wolfia brasiliensis Wedd. dominates aquatic vegetation with 8090 % cover and submerged U. inflata with 10-20 % cover. Some isolated specimens of Nymphaea alba Salib, Azolla filiculoides Lam., Eichhornia crassipes (Mart.) Solms and L. valdiviana Phil. were also found. In the dry season the whole body of water is covered with W. brasiliensis (Gordon et al. 2003). Vegetation at Río de Agua corresponded to a typical herbaceous wetland, with no tree cover. Average water depth was 29.75 cm, with a maximum of 43 cm in January and a minimum of 14 cm in May, where Typha dominguensis (Pers.) Steud predominates, reaching 70-80 % cover. Other species found were Eleocharis mutata (L.) Roem and Schult., Cyperus articulatus L., C. surinamensis Rottb., Ceratopteris pteridiodes (Hook) Hieron, the free-floating L. valdiviana, and the submerged U. gibba (Gordon et al. 2003), which disappeared at the end of the dry season.
At the time (rainy season, 2000) plants were collected, dominant phytoplankton divisions in sampled water were Bacillariophyta, Chlorophyta, Cyanobacteria,and Euglenophyta. Total density of Bacillariophyta was 36 537 ind/l and 2 447 ind/l in lake and marsh, respectively. Density of Chlorophyta was 3 754 ind/l only in the marsh. Cyanobacteria had densities of 20 650 ind/l in the lake and 3 922 ind/l in the marsh. Likewise, Euglenophyta showed densities of 13 501 ind/l and 4 223 ind/l in lake and marsh, respectively (Delgado, unpub. data). In both locations, zooplankton was composed of copepods, cladocerans, and rotifers. In the Bohordal lake, average densities of copepods, cladocerans, and rotifers were 65, 30, and 26 ind/l, respectively; in Rio de Agua, average densities of copepods, rotifers, and cladocerans were 54, 36, and 22 ind/l, respectively (Zoppi de Roa et al. 2002).
U. inflata and U. gibba are free-submerged aquatic plants characterized by absence of real leaves or roots (Velásquez 1994), was found and collected in the Bohordal Lake and the latter in the T. dominguensis- dominated herbaceous wetland of Rio de Agua.
Water pH, conductivity, dissolved oxygen, and salinity were measured in situ in each of the two locations using a Horiba Walter Quality Checker, Mod. U-10, and water samples taken to analyze N, P, Ca++, Na+, K+, and Mg++ content for the purpose of determining the main physical and chemical characteristics of the environment where the plants grow. Nitrogen was determined by the macro Kjeldahl method and phosphorus by colorimetric analysis (APHA 1992). Metals (Ca++, Na+, K+, and Mg++) were determined by atomic absorption.
Botanical samples were collected, labeled, and preserved in FAA fixing solution prepared with 500 ml alcohol, 100 ml formaldehyde, and 50 ml acetic acid. For each specimen quintuplicate sections of 5 cm were taken from each plant and bladder number recorded for each section. Fifty bladders approximately the same size and all appearing full (smaller bladders were discarded) were set aside from each species, washed with distilled water to eliminate any animal or algae adhered to the outer walls of the bladders so they would not be taken into account when analyzing contents. Bladders were then measured one by one and opened to disclose their contents under a stereoscopic microscope. Contents were analyzed and classified to respective supergroup level according to the available bibliography (Ward and Whipple 1959, Prescott 1970, Bold and Wynne 1985, APHA 1992).
The chi square test was applied to the number of individual algae and animals found in the 50 utricles analyzed for each of the two species, with a significance level of p<0.05. The Spearmans correlation coefficient was calculated to establish the percentage of similitude between the two species based on animal and vegetal morphospecies (taxa) density, also with a significance level of p<0.05. Calculations were made using the Statistics 6.0 software package.
Results
In the Bohordal lake conductivity was 0.06 mS/cm, dissolved oxygen was 2.01 mg/l, and pH was 6.6. N-Kjeldahl and total P were 2.6 mg/l and 0.11 mg/l, respectively; Ca++, Mg++, Na+, and K+ were 4.7 mg/l, 2.4 mg/l, 5 mg/l, and 5.5 mg/l, respectively. In the Rio de Agua herbaceous wetland, conductivity was 2.8 mS/cm, dissolved oxygen 3.4 m/l, pH 7.8, and salinity 1.84o/oo. N-Kjeldahl and total P were 2.81 mg/l and 0.13 mg/l, respectively; Ca++, Mg++, Na+, and K+ were 77 mg/l, 36 mg/l, 177 mg/l, and 12 mg/l, respectively.
Mean bladder length and width were 1 651.81±397.00 µm and 1 219.33±313.75 µm, respectively for U. gibba, and 1 243.46±222.02 µm and 994.78±200.91 µm for U. inflata. Bladder size expressed as length by width was 2.11±0.95 mm2 for U. gibba, and 1.22±0.31 mm2 for U. inflata. Average bladder number in the 5 cm segments cut from the plants was 4 bladders/cm for U. inflata, and 6 bladders/cm for U. gibba. Found inside the utricles were algae and animals, free and/or adhered to walls or debris, partially digested animals, remains from other plants, nematodes, insects, eggs, and other non-identified material.
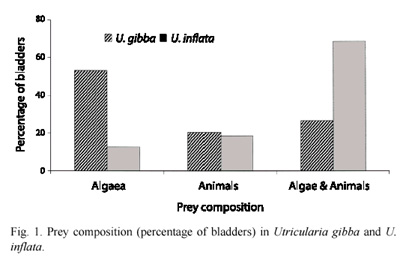
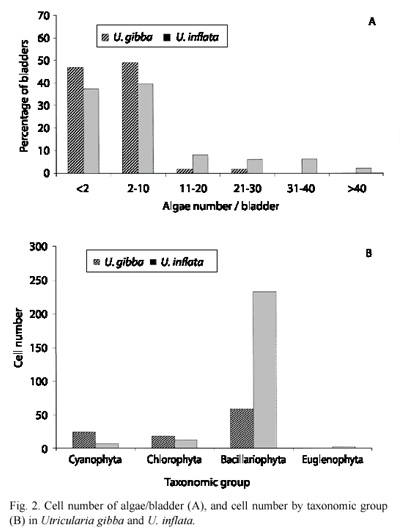
In U. inflata, 69 % of the bladders contained both algae and animals, while in U. gibba, 53 % of the bladders contained only algae (Fig. 1). For both species most bladders contained between two and ten algae (Fig. 2A) with an average of 2 algae/bladder for U. gibba and 5 algae/bladder for U. inflata. A total of 102 algae were identified in U. gibba and 256 in U. inflata. Algae made up 61 % and 67 %, respectively, of all organisms found in U. gibba and U. inflata. Algae were classified into Cyanophyta (Cyanobacteria), Chlorophyta, Bacillariophyta, and Euglenophyta. Abundances of Cyanobacteria and Chlorophyta in both species were relatively similar. Bacillariophyta were more abundant in U. Inflata. Euglenophyta, the least abundant, were found only in U. inflata (Fig. 2B). The chi square test for algal density/division between the two species gave significant differences (X2= 7.81; df= 3; p<0.05). Algal morphotype richness by division was superior in U. inflata, especially in Chlorophyta (Table 1). When prey composition was compared, based on density of vegetal morphospecies between both species, and using Spearmans correlation coefficient, the result was non-significant, rs= -0.26 (p<0.15).
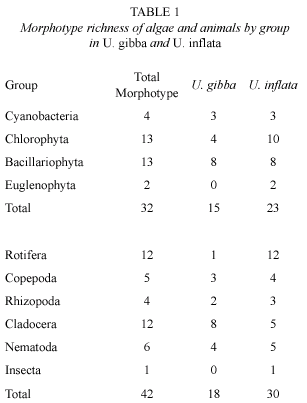
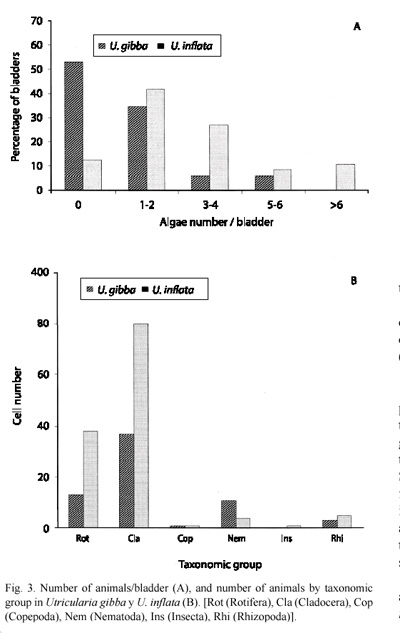
In U. gibba more than 50 % of the bladders contained no animals and 35 % contained 1-2 individuals, and none of the bladders contained more than six. Meanwhile, in U. Inflata a greater proportion of bladders (69 %) contained 1-4 individuals, with as many as 13 in some cases, and only 14 % contained no animals at all (Fig. 3A). Fixty six individual animals were identified in U. gibba with an average density of 1.30 animals/bladder and 129 in U. inflata with an average of 2.58 animals/bladder. Animals represented 39 % and 34 %, respectively, of total of individuals in the bladders. In U. inflata, cladocerans and rotifers were more abundant, while in U. gibba were the nematodes. Copepod and rhizopod densities showed no major differences between the two species. Only one insect was found in U. inflata (Fig. 3B). The chi square test of captured animals density between both species was significant (calculated: X2= 9.601; theoretical: X2= 0.711; df= 4; p<0.05). Morphotype (taxon) richness was greater in U. inflata, especially in rotifers, while in U. gibba, cladocerans were the group with greater richness (Table 1). When prey composition was compared, based on animal morphotype density between both species, and using Spearmans correlation coefficient, the result was non-significant, rs= -0.15 (p<0.44).
Discussion
According to a classification of water bodies by trophic quality, and nitrogen and phosphorus data, Bohordal lake may be classified as a eutrophic ecosystem (OECD 1980, Colmenares 1990, Ryding and Rast 1992). The herbaceous wetland is included in the oligohaline category (Mitsch and Gosselink 2000), from salinity values and a slightly alkaline pH. In this habitat, total nitrogen, total phosphorous, and Ca++, Mg++, Na+, and K+ values are similar to the concentration range reported by Kadlec and Knight (1996) for a eutrophic wetland containing Typha spp. and Scirpus spp., located in North America. So in both localities, in principle, no limitations should be found on nitrogen and phosphorous availability for carnivore plants.
The physicochemical characteristics - such as macronutrients and other ions, salinity and pH - of the water probably influence the composition and abundance of the phytoplankton and, of course, the zooplankton (Delgado et al. unpublished, Zoppi et al. 2002). Since Utricularia catches both phytoplankton and zooplankton (Botta 1976, Pompêo and Bertuga 1996), the prey composition reflects possibly the plankton biodiversity outside the bladders. At the group level a correspondence between prey composition and plankton was observed, however there was a discrepancy regarding Chlorophyta; their presence was not determine in the Bohordal pond, but they were found inside the bladders of U. inflata; also the cladocera density in the water was lower in both environments in comparison with the total number of these organisms in both plant species. Mette et al. (2000) made comparisons between the surrounding plankton and the perifiton and demonstrated that the bladderworts do not trap the animals swimming in the open-water but those organisms staying as perifiton between and on the plants; also indicated that the prey composition from the bladders produces a partly image of environment, because to the presence of partly digestion organisms up to detritus indicate that a lot of organisms could not longer be determined and were therefore not counted. As certain taxa are digested faster than others -eg. ciliates and bdelloid rotifers can after a few hours no longer be taken into account, while crustaceans and armoured rotifers may still be identified after several days - they are not sufficiently represented in the evaluation. Besides there are organisms that live and thrive in the Utricularia spp. trap, feeding on the captured prey without themselves being digested (Pietropaolo and Pietropaolo 1986).
Although both species plants are very similar in what they catch at the level of great group, however according to the data analysis (X2 test, Spearman correlation coefficient) there is no similarity regarding the algae and animal morphotypes caught, that is, the two species consume different morphotypes. U. gibba has more bladders and they are bigger, but U. inflata catches more diverse prey, about three times more than U. gibba. The possible reason for this is that in the Bohordal pond, the diversity and abundance of algae and animals is higher (Delgado et al. unpublished, Zoppi de Roa et al. 2002). The capture potential depends on the quantity as much as on the quality or the available organisms in the microhabitats where the plants grow and it is unquestionable that any variation on plankton composition will have an effect on prey composition (Zamora 1995).
The greater ammount organisms were found inside U. inflata, as much as 13 individuals per utricle. Pompêo and Bertuga (1996) reported on various species of Utricularia, that the number of organisms caught in a 20-hour period varied between 3 and 11 individuals. Apparently, the size of the prey is determinant. U. inflata captures more rotifers, which are of a smaller size, typically 100-500 µm. On the other hand, U. gibba catches a higher diversity of cladocera, which are of higher size, typically 0.2-3mm (Kadlec and Knight 1996).
Results connected to group density and morphospecies diversity allow us to infer that both species of Utricularia capture whatever is around them, without discrimination and they are limited only by bladder size and prey availability and size. Apparently, U. inflata captures prey more efficiently but is limited by prey size, while U. gibba is limited by prey availability. Mette et al. (2000) showed that Utricularia in different European habitats do not capture their prey selectively but are dependent on the existing prey community. Zamora (1995) proposed that success for a carnivorous plant is the result of a complex process that depends on performance of trapping mechanisms as much as on prey abundance, its temporal and spatial distribution, and behavior, which determines the quantity of animal biomass that can be caught.
No animal content in 48 % of bladders in U. gibba, and the high proportion of algae inside the bladders of both species suggest these plants are either inefficient in capturing animal prey or do not require solely animal prey in natural conditions (Richards 2001). Zegarra (pers. comm.) proposes that Utricularia might be showing an evolutionary trend from a mutualistic relationship between bladder plants and plankton organisms, especially the more abundant phytoplankton, towards the utilization of zooplankton or its byproducts, initially by using the debris of those dying in the bladders at the end of their life cycle or by predation from other microfaunal bladder inhabitants, and eventually, by the random emergence and subsequent selection of either metabolic pathways for optimizing nutrient absorption or mechanical adaptations for the development of a more functional carnivory or both. Botta (1976), Mette et al. (2000) reported the presence of autotrophic organisms inside the bladders. Mette et al. (2000) further suggested that these autotrophs are not to be digested but use the bladder as a habitat in which they photosynthesize and reproduce. It seems autotrophic organisms as well as different groups of small animals are part of the food spectrum of Utricularia.
. Since these ecosystems are eutrophic, the presence of U. inflata and U. gibba in them poses a conflict with the conclusions of several authors (Sculthorpe 1967, Heslop-Harrison 1978, Slack 1980, Friday 1989, Knight 1992, Pompêo and Bertuga 1996) about these plants growing in nutrient-poor environments. The majority of aquatic carnivorous plants usually grow in soft or medium-hard, acid or neutral, waters, and its can only use free CO2 (not HCO3-) for photosynthesis (Adamec 1997). Some aquatic carnivorous plants strictly require humic acids for normal growth and development, while other species only grow in a mineral medium with traces of peptone, beef extract, glucose or acetate (Adamec 1997). The growth of some aquatic Utricularia species was promoted considerably by organic substances with or without N (Sculthorpe 1967). In aquatic aquatic carnivorous plants, the role of organic carbon absorbed from prey may be ecologically important, mainly under the conditions of CO2 or light limitation. Adamec (1996) found that the growth of Aldrovanda vesiculosa L. (Droseraceae) - an aquatic carnivorous plant with a way of life and a strategy that resemble those of Utricularia -was greater in waters with a high concentration of CO2 and suggested that carnivory can be a way to obtain additional carbon. These plants possibly confine living organisms not only to absorb nitrogen and phosphorous, also for the CO2 produced through respiratory activity of the community inside the utricles. This would explain why U. gibba and U. inflata lives in environments where nitrogen and phosphorous do not limit growth, and also why a variety of organisms ranging from algae and animals to unidentified plant material are found inside them.
Further research should complement this study by including aspects like seasonal variation in prey composition, variation and percentage of inverted biomass in utricles, and comparison of productivity of these plants with and without bladders to find out how they can be affected by lack of carnivory-supplied nitrogen. Also, a comparison of prey composition in different sites, because, bladderworts of the same species may prey upon different organisms in different habitats (Mette et al. 2000). Also, available and total macronutrient concentration should be evaluated because of the huge difference between them in the water (or soil) (Adamec 1997).
Acknowledgments
Research was supported by a World Bank Grant # 021-044. We deeply appreciate the collaboration of Laura Delgado for the plankton data; to Cruz Salazar, Evelyn Zoppi de Roa and Federico González for their help in identification of algae and animal specimens; to Julio González and Napoleón León for their help in field, Argenis Delfín for water analysis and Jorge Zegarra for his help with the English manuscript.
Resumen
Las plantas carnívoras acuáticas del género Utricularia (Lentibulariaceae), capturan pequeños organismos acuáticos, como rotíferos, copépodos, y cladóceros, por medio de estructuras anatómicas semejantes a vesículas. Este estudio tuvo como objetivo determinar el tamaño y tipo de presas de U. gibba y U. inflata, recolectados en un pequeño lago y en un pastizal inundado, respectivamente, en la localidad de la Península de Paria (Estado Sucre, Venezuela). La acidez, conductividad, cantidad de oxígeno disuelto y la salinidad del agua se midieron in situ para cada sitio de muestreo; también se tomaron muestras de agua para determinar niveles de N-Kjeldahl, P-total, Na+, K+, Ca++, Mg++ y Cl-. Se midieron cincuenta vesículas de cada especie y se analizaron sus contenidos. Los valores de N-Kjeldahl y P-total fueron similares en ambos sitios, y semejantes a los conocidos para ecosistemas eutróficos; sin embargo, las concentraciones de Na+, K+, Ca++, Mg++ y los valores in situ de los parámetros del agua fueron más altos en el pastizal inundado. Las vesículas contenían los siguientes grupos de zooplancton: rotíferos, cladóceros, copépodos, anélidos, rizópodos, e insectos; y las siguientes divisiones de fitoplancton: Bacillariophyta, Chlorophyta, Cyanophyta y Euglenophyta. Las vesículas presentadas por U. inflata fueron menores en tamaño y en cantidad, pero mayores en abundancia y riqueza total de morfoespecies algales y animales en comparación con U. gibba. La similitud en cuanto a composición de presas a nivel taxonómico, entre las dos especies de plantas carnívoras, fue baja.
Palabras clave: Lentibulariaceae, Utricularia, planta carnívora, composición de presas, captura de presas, plancton, Venezuela.
References
Adamec, L. 1996. Photosynthetic characteristic of the aquatic carnivorous plant Aldrovanda vesiculosa. Aquat. Bot. 59: 297-306. [ Links ]
Adamec, L. 1997. Mineral nutrition of carnivorous plants – A review. Bot. Rev. 63: 273-299. [ Links ]
Angerilli, N.P.D & B.P Beirne.1980. Influence of aquatic plants on colonization of artificial ponds by mosquitoes and their insect predators. Can. Ent. 112: 793-796. [ Links ]
APHA. 1992. Standard Methods for the Examination of Water and Wastewater. American Public Health Association, Washington DC, USA. 560 p. [ Links ]
Baumgartner, D.L. 1987. Laboratory evaluation of the bladderwort plant Utricularia vulgaris (Lentibulariaceae), as a predator of late instar Culex pipiens and assessment of its biocontrol potential. J. Am. Mosquito Contr. 3: 50-62. [ Links ]
Bold, H.C. & M.J. Wynne. 1985. Introduction to the Algae. Prentice-Hall, New Jersey, USA. 720 p. [ Links ]
Botta, S.M. 1976. Sobre las trampas y las víctimas o presas de algunas especies argentinas del género Utricularia. Darwiniana 20: 127-154. [ Links ]
Colmenares, A.S. 1990. Evaluación del Estado Trófico del Embalse La Mariposa, Caracas-Venezuela. Tesis de maestría, Instituto Venezolano de Investigaciones Científicas, Caracas, Venezuela. 168 p. [ Links ]
Cook, D.K. 1996. Aquatic Plant Book. S.P.B. Academic, The Hague, Holland. 228 p. [ Links ]
Ellison, A.M. & N.J. Gotelli. 2001. Evolutionary ecology of carnivorous plants. Trends Ecol. Evol. 16: 623-629. [ Links ]
Friday, L.E. 1989. Rapid turnover of traps in Utricularia vulgaris L. Oecologia 5: 272-277. [ Links ]
Friday, L.E. 1992. Measuring investment in carnivory: Seasonal and individual variation in trap number and biomass in Utricularia vulgaris L. New Phytol. 126: 273-281. [ Links ]
Friday, L.E. & C. Quarmby. 1994. Uptake and translocation of prey derived 15N and 32P in Utricularia vulgaris L. New Phytol. 126: 273-281. [ Links ]
Gordon, E., L. Delgado, E. Zoppi de Roa, J. Berti, E. Montiel & S. Ramos. 2003. Composición florística de la vegetación y fisicoquímica del agua en humedales de la Península de Paria (Estado Sucre), 89 p. In Memorias del V Congreso Venezolano de Ecología. Sociedad Venezolana de Ecología, Margarita, Nueva Esparta, Venezuela. [ Links ]
Hegner, R.W. 1926. The interrelations of Protozoa and the utricles of Utricularia. Biol. Bull. 50: 239-270. [ Links ]
Heslop-Harrison, Y. 1978. Carnivorous Plants. Sci. Amer. 104: 156-185. [ Links ]
Juniper, B.E, R.J. Robins & D.M. Joel.1989. The Carnivorous Plants. Academic, London, England. 353 p. [ Links ]
Kadlec, R. & R. Knight. 1996. Treatment Wetlands. Lewis, New York, USA. 893 p. [ Links ]
Knight, S.E. & T.M. Frost. 1991. Bladder control in Utricularia macrorhiza: Lake-specific variation in plant investment in carnivory. Ecology 72: 728-734. [ Links ]
Knight, S.E. 1992. Cost of carnivory in the common bladderwort, Utricularia macrorhiza. Oecologia 89: 348-355. [ Links ]
Lloyd, F.E. 1942. The Carnivorous Plants. Chronica Botanica, New York, USA. 352 p. [ Links ]
Mette, N., N. Wilbert & W. Barthlott. 2000. Food composition of aquatic bladderworts (Utricularia, Lentibulariaceae) in various habitats. Beitr. Biol. Pflanzen 72: 1-13. [ Links ]
Mitsch, W.J. & J.G. Gosselink. 2000. Wetlands. Wiley, New York, USA. 920p. [ Links ]
OECD (Organization for Economic Cooperation and Development). 1980. Cooperative Programme for monitoring of inland waters (Eutrophication Control). Regional project: Shallow lakes and reservoirs. Water Research Center, Paris, France. 297 p. [ Links ]
Prescott, G.W. 1970. The Freshwater Algae. C. Brown, Duque, Iowa, USA. 348p. [ Links ]
Pompêo, M.L & M. Bertuga.1996. Captura de organismos planctônicos pelas plantas carnívoras do gênero Utricularia (Angiospermae, Dicotyledonea). Rev. Brasil. Biol. 56: 697-703. [ Links ]
Pietropaolo, F. & P. Pietropaolo. 1986. Carnivorous Plants of the World. Timber, Oregon, USA. 206 p. [ Links ]
Richards, J.H. 2001. Bladder function in Utricularia purpurea (Lentibulariaceae): Is carnivory important? Amer. J. Bot. 88: 170-176. [ Links ]
Rutishauser, R. & J. Brugger. 1992. Structural and developmental diversity of Utricularia traps. Carnivorous Plants Newsletter 21: 68-74. [ Links ]
Rutishauser, R. 1993. The developmental plasticity of Utricularia aurea (Lentibulariaceae) and its floats. Aquat. Bot. 45:119-143. [ Links ]
Ryding, S.O. & W. Rast. 1992. El Control de la Eutrofización en Lagos y Pantanos. Pirámide, Madrid, Spain. 375 p. [ Links ]
Sattler, R. & R. Rutishauser. 1990. Structural and dynamic descriptions of the development of Utricularia foliosa and Utricularia australis. Can. J. Bot. 68: 1989-2003. [ Links ]
Slack, A. 1980. Carnivorous Plants. MIT, Cambridge MA, USA. 240 p. [ Links ]
Schnell, D.E. 1976. Carnivorous Plants of the United States and Canada. John F. Blair, North Carolina, USA. 125 p. [ Links ]
Sculthorpe, C.D. 1967. The Biology of Aquatic Vascular Plants. Edward Arnold, London, England. 610 p. [ Links ]
Taylor, P. 1989. The Genus Utricularia -A taxonomic monograph. Royal Botanic Gardens, London, England. 724 p. [ Links ]
Ulanowicz, R.E. 1995. Utricularias secret: The advantage of positive feedback in oligotrophic environments. Ecol. Model. 79: 49-57. [ Links ]
Velásquez, J. 1994. Plantas Acuáticas Vasculares de Venezuela. Universidad Central de Venezuela, Caracas, Venezuela. 992 p. [ Links ]
Ward, H.B. & G.C. Whipple. 1959. Freshwater biology. Wiley, New York, USA. 1248p. [ Links ]
Zamora, R. 1995. The trapping success of carnivorous plant, Pinguicula vallisneriifolia: The accumulative effects of availability, attraction, retention and robbery of prey. Oikos 73: 309-322. [ Links ]












 uBio
uBio 

