Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.49 no.3-4 San José dic. 2001
Reproductive activity and condition index of Holacanthus passer (Teleostei: Pomacanthidae)
in the Gulf of California, Mexico
Marcial Arellano-Martínez and Bertha Patricia Ceballos- Vázquez
Centro Interdisciplinario de Ciencias Marinas, Apdo. Postal 592. 23000, La Paz. B. C. S., México. Fax: 51(112) 25322; e-mail: marellam@redipn.ipn.mx
Received 24-IV-2000. Corrected 30-XI-2000. Accepted 30-1-2001.
Abstract
Annual variation and relationship between the gonadosomatic index (lG) and the condition index (K) of Holacanthus passer were evaluated in 194 specimens from Cueva de León, Gulf of California (24°02' N,110º 24' W). There was a significant negative correlation between IG and K in both sexes: indices exhibit an oposite seasonal pattern. The reproductive activity of both sexes is inversely related with nutritional status. There was a significant difference in IG but not in K values between males and females. The condition index may be used to determine reproduction time of the species in other localities without sacrificing the organisms. This could be a valuable tool to develop monitoring programs for fisheries and culture of this species.Key word: Holacanthus passer, Pomacanthidae, gonadosomatic index, condition index, Gulf of California, Mexico.
The gonadosomatic index has been used as an indicator of reproductive activity (Elorduy-Garay and Ramírez-Luna 1994, Merayo 1996. Rajasilta et al. 1997). Its utility to detect hydrated ovaries and therefore detect reproductive period from increase in weight has been established by Hunter and Macewicz (1985), Thus the gonadosomatic index provides a useful estimation of spawning duration (Ceballos-Vázquez and Elorduy-Garay 1998). When the gonadosomatic index has been histogically validated, it provides useful information about the annual trend in the reproductive activity of the population. In Holacanthus passer Valenciennes, 1846, the annual trend in the mean monthly values of the gonadosoinatic index is consistent with the reproductive activity and histological data. This indicates that the period of reproductive activity is from April to November (Arellano-Martínez et al. 1999).
In fisheries ecology, the study of condition is a standard practice and assumes that a heavier fish of a given length is in better condition (Bolger and Connolly 1989). Condition has been most effectively used to compare monospecific populations living under similar or apparently different environmental conditions, in determining the timing and duration of gonad maturation, and as an indication of changing gross nutritional balance during chronic alterations in feeding activity or food supply (Bolger and Connolly 1989).
The condition index has been used as a simple indicator of the physical and physiological status of the fish (e.g. it works as a relative measure of the nutritional status) (Encina and Granado-Lorencio 1997). It is also known that a close relationship exists between physiological status, condition, and reproductive stage. The purpose of this study is to evaluate the relationship between the gonadosomatic index and the condition index of a population of king angelfish H. passer V. from Gulf of California, Mexico.
Materials and methods
From June 1992 to May 1993, adult specimens of the king angelfish H. passer were collected monthly with Hawaiian speargun at 2 to 10 m depth, at Cueva de León (24°02' N, 110°24' W) in the Gulf of California, Mexico. Organisms were measured (total length, mm) and weighed (total weight, g). Gonads were dissected and weighed (0.01 g accuracy).
The size of the ovary in the fish increases as it develops; therefore the ratio between the size of the ovary and the size of the body is an objective indicator of ovarian development (West 1990). Thus the gonadosomatic index (IG) was calculated as follows:
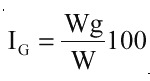
where Wg = gonad weight, and W = total weight.
The ratio between weight and length was used to calculate the condition index (K) as follows:
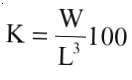
where W = total weight, and L = total length (Maddock and Burton 1999). The condition index as sumes that growth is isometric, but the exponent 3 can be considered simply as a method of transforming the linear dimensions of length to the cubic dimensions of weight (Bolger and Connolly 1989).
As IG and K are a percentage value, the arcsine transformation (Sokal and Rohlf 1995) was used to attain normality and homoscedasticity of data for statistical analysis.
Results
A total of 194 specimens were collected: 112 females and 82 males. Females ranged in total length from 90 to 222 mm (161.9 + 22.3 mm) and males ranged from 81 to 253 mm (201.2 + 40.5 mm).
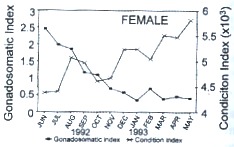
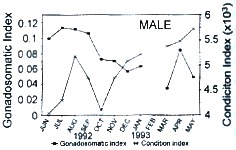
The mean values of IG (Fig. 1a, b) were low (< 1.25 for females and < 0.08 for males) from mid-autumn (October-November) to mid-spring (May), except in April for males when there was an increase in the IG due to a larger proportion of developing and ripe individuals in the sample (Fig 1b). At the beginning of summer (June), the female index reached the highest value (2.45), and tended to decrease over the next months (Fig. 1a). On the other hand, the highest values of K for both sexes were observed from late autumn (December) through mid-spring (January to May), whereas the lowest values occurred from late spring (June) to mid-ailtumn (November), in coincidence with reproductive activity (Fig. 1a, b).
Fig. 1. Monthly variation of the mean values of gonado-somatic index and condition
index of Holacanthus passer from Cueva de León, B.C.S., México. a) Females, b) Males.
The values of IG and K had an opposite seasonal pattern (Fig. 1): There was a significant negative linear correlation (P < 0.05) between IG and K for females (r = -0.76) and males (r = -0.63).
There was a significant difference in IG (One way analysis of variance ANOVA, F1,192 = 282.48; P < 0.05) but not in K values (One way ANOVA, F1.192 = 0.06; P > 0.05) between males and females. One way ANOVA analysis, computed independently for each sex, detected seasonal differences of IG and K: female IG (F11,100 = 21.05; P < 0.05), male IG (F10.70 = 4.80; P < 0.05), female K (F11,100 = 12.41; P < 0.05), and male K (F10,70 = 14; P < 0.05). Tukey's test for IG detected three groups: The first group includes female samples from June to August 1992, showing IG values > 1.8 %; and male samples from June to September 1992 with values > 0.09 %. The second group consists of female samples from September to December 1992, and February 1993, with IG values between 0.58 % and 1.17 % respectively, and male samples from October 1992 to January, and April 1993, with IG values between 0.05 % and 0.7 %. The third group contains female samples from January to May except February 1993, with IG values < 0.34 %; and male samples from March and May 1993 with values < 0.05 %.
Discussion
The presence of ripe males started in April (as indicated by the considerable rise of IG values) whereas the presence of ripe females was detected until June. These results are supported by a histological analysis performed on the same organisms (Arellano-Martínez el al. 1999). Consistent with our results, the authors
found that 16.6 % of males were ripe in April, whereas ripe females were not found until June (66.6 %). This suggests that males are potentially reproductive more time than females. The male gonad is smaller than the female gonad, therefore the male fish requires less stored energy for gonadal development and it may start to mature earlier than females. In freshwater species, other authors (based on IG cycles) determined that maximum testis development occurred before that of ovaries (Mann 1974, Philippart 1981, Poncin el al. 1987, Cowx 1990, Encina and Granado-Lorencio 1997), our results suggest that this phenomenon also occurs in marine species.
The IG monthly variation in H. passer is closely related to the reproductive activity on the basis of histological validation made by Arellano-Martínez et al. (1999). On the other hand, K usually has been used as a relative measure of the nutritional status of fish (Encina and Granado-Lorencio 1997). In H. passer, the reproductive activity is inversely related to the nutritional status. It is more evident for females because of their higher energy requirement for gonad development (Encina and Granado-Lorencio 1997).
In H. passer, the higher K values found during the inactive reproductive period (smaller IG values, winter-spring) could be produced by accumulation of reserve substances, to be used during ripening, The beginning of gonadal development (rising IG values) coincided with the highest K values, whereas their depletion occurred in both sexes just before the maximum gonadal development (highest IG values) and during spawning (summer-autumn).
There are some explanations for the depletion of K values. One could be a change in feeding patterns, which could in turn be a behavioral response to certain stressors such as water contamination with chemicals (Brown et al. 1987). Another could be an increase in metabolic rate in response to stress factors such as physical disturbance, which force the fish to struggle (Barton and Schreck 1987). According to our data, in H. passer, the decrease in K values is interpreted as depletion of energy reserves such as liver glycogen or body fat, which later is transferred from somatic tissues to the gonad to be used in oocyte maturation. The same decline in condition during the spawning period is recorded for other species (Bagenal 1957, Ceballos-Vázquez 1993, Rajasilta et al. 1997, Maddock and Burton 1999). Nevertheless, in order to get a precise conclusion on the interaction between condition and reproductive biology in H. passer, a more detailed study must be accomplished, including calorimetric, biochemical and histochemical studies in gonad, liver and muscle in arder to know the energetic transference between organs in relation to the reproductive cycle.
Notwithstanding, our results reveal that the condition index may be used to determine the time of reproduction of the species in other localities without sacrificing the organisms. This could be a valuable tool to develop monitoring programs for tisheries and to culture this species.
Acknowledgments
We are grateful to Dirección de Estudios de Postgrado e Investigación del Instituto Politécnico Nacional for funding this work. Thanks to Alejandra García for her right suggestions that improve the paper, and to Ellis Glazier for editing this English-language text.
Resumen
Se evaluó la variación anual y la relación entre el índice gonadosomático (lG) y el índice de condición (K) de Holacanthus passer de un total de 194 especímenes recolectados en Cueva de León, Golfo de California, México (24°02' N, 110°24' W). Se encontró una correlación negativa significativa entre IG y K para ambos sexos: los índices presentan un patrón estacional opuesto. La actividad reproductiva de ambos sexos está inversamente relacionada con el estado nutricional del pez. Se encontró una diferencia significativa en los valores de IG pero no en los valores de K entre machos y hembras. El índice de condición puede ser usado para determinar la época de reproducción de la especie en otras localidades sin sacrificar al organismo y puede ser una herramienta valiosa para el desarrollo de programas de monitoreo para la pesquería y cultivo de la especie.
References
Arellano-Martínez M., B.P. Ceballos-Vázquez, F. García Domínguez & F. Galván-Magaña. 1999. Reproductive biology of king angelfish Holacantus passer, Valenciennes, 1846 in the Gulf of California, Mexico. Bull. Mar. Sci. 65: 677-685. [ Links ]
Bagenal, T.B. 1957. The breeding and fecundity oí tbe long rough dab Hippoglossoides platessoídes (Fabr.) and the associated cyde in condition. J. Mar. Biol. Ass. U.K. 36: 339-375. [ Links ]
Barton, B.A. & C.B. Schreck. 1987. Metabolic cost oí acote physical stress in juvenile steelhead. Trans. Amer. Fish. Soco 116: 257-263. [ Links ]
Bolger, T. & P.L. Connolly. 1989. The selection of suitable indices for the measurement and analysis of fish condition. J. Fish Biol. 34: 171-182. [ Links ]
Brown, J.A, A. Johansen, P.H., Colgan, P.W. & R.A. Mathers. 1987. Impairment of early feeding behavior oílargemouth bass by pentachlorophenol exposure: a preliminary assessment. Trans. Amer. Fish. Soco 116: 71-78. [ Links ]
Ceballos-Vázquez, B.P. 1993. Biología reproductiva del conejo Caulolatilus affinis, Gill 1865 (Pisces: Branchiostegidae), en el Canal Cerralvo, Baja California Sur, México. Master in Science Thesis, Centro Interdisciplinario de Ciencias Marinas-Instituto Politécnico Nacional, La Paz, B.C.S. México. [ Links ]
Ceballos-Vázquez, B.P. & J. Elorduy-Garay. 1998. Gonadal development and spawning of the Golden-eyed tilefish Caulolatilus affinis (Pisces: Branchiostegidae) in the Gulf of California, México. Bull. Mar. Sci. 63: 469-479. [ Links ]
Cowx, I.G. 1990. The reproductive tactics of roach, Rutílus rutilus (L.) and dace, Leuciscus leuciscus (L.) populations in the river Exe and Culm, England. Pol. Arch. Hydrobiol. 37: 193-208. [ Links ]
Elorduy-Garay, J. & S. Ramírez-Luna. 1994. Gonadal development and spawning of female ocean whitefish, Caulolatilus princeps (Pisces: Branchiostegidae) in the Bay ofLa Paz, B.C.S., Mexico. J. Fish Biol. 44: 553-566. [ Links ]
Encina, L. & C. Granado-Lorencio. 1997. Seasonal variations in the physiological status and energy content of somatic and reproductive tissues of chub. J. Fish Biol. 50: 511-522. [ Links ]
Hunter, J.R. & B.J. Macewicz. 1985. Measurement oí spawning frequency in multiple spawning fishes, p. 79-94. In R. Lasker (ed.). An egg production method for estimating spawning biomass of pelagic fish: application to the northern anchovy, Engraulis mordax. NOAA Tech. Rep. NMFS-36. [ Links ]
Maddock, D.M. & M.P. Burton. 1999. Gross and histological of ovarian development and related condition changes in American plaice. J. Fish Biol. 53: 928944. [ Links ]
Mann, R.H.K. 1974. Observations on the age, growth, reproduction and foot of the dace, Leuciscus leuciscus (L.) in two rivers in southern England. J. Fish Biol. 6: 237-253. [ Links ]
Merayo, C.R. 1996. Seasonal change in the biochemical composition of the muscle and liver of bib (Trisopterus luscus L.) (Pisces, Gadidae) from the Cantabrian Sea (N Spain). Sci. Mar. 60: 489-495. [ Links ]
Philippart, J.C. 1981. Ecologie d' une population de vandoises, Leuciscus leuciscus (L.) dans la Riviere Ourthe (Bassien de la Meuse, Belgique). Ann. Limnol. 17: 41-62. [ Links ]
Poncin, P., Ch. Melard & J.C. Philippart. 1987. Utilisation de la température et de la photopériode pour controler la rnaturation sexuelle en captivité de trois espéces de poissons cyprinidés européens: Barbus barbus (L.), Leuciscus cephalus (L.) et Tinca tinca (L.). Résultats préliminaires. Bull. Franc. Péche Piscicult. 304: 1-12. [ Links ]
Rajasilta, M., J. Paranko & T. Laine. 1997. Reproductive characteristics of the male herring in the northern Baltic Sea. J. Fish Biol. 51: 978-988. [ Links ]
Sokal, R. & J.H. Rohlf. 1995. Biometry. Freeman, New York. 887 p. [ Links ]
West, G. 1990. Methods of assessing ovarian development in fishes: A review. Aust. J. Mar. Freshwater Res. 41: 199-222. [ Links ]












 uBio
uBio