Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.49 no.2 San José jun. 2001
of Missionary Bay, North Queensland, Australia
Received 21-III-2000. Corrected 10-X-2000. Accepted 10-X-2000
Abstract
To assess the occurrence, spatial distribution and species composition of small infauna on a mangrove shore, core samples were taken along a transect in Missionary Bay at Hinchinbrook Island, north-east Australia. Three sites were arranged within the mangrove forest and one site was located in an adjacent mudflat. The sites were surveyed four times between November 1988 and October 1989. Based on the records from all samples and sites, 39 taxa were identified. Diversity (H') ranged from 1.18 to 2.38. Overall, total abundances of small infauna (retained on a 0.25 mm sieve) reached a mean value of 5 477 ind. m-², with little variation throughout the transect or over time. Species numbers and diversity were higher in the mudflat than at the mangrove sites. The taxonomic composition changed between the mangrove forest and the mudflat: Oligochaeta were more abundant in mangrove sediments, whereas Polychaeta dominated in the mudflat. Of the polychaetes, Capitellidae were almost restricted to the mangrove sites, Sabellidae were recorded frequently at all sites, and Sigambra parva and Myriochele sp. were confined to the mudflat and the mangrove fringe. These species accounted also for dissimilarities between sites. Multivariate analyses showed a distinct assemblage at the mudflat compared to the mangrove sites. This survey showed that small infauna is an abundant component of mangrove sediments, which has been previously underestimated.
Key words
Mangroves, benthic community, infauna, macrobenthos.
Traditionally, the fauna of marine sediments is classified into meio- and macrofauna by the use of defined sieve sizes, whereby organisms retained on a 0.5 or 1 mm mesh sieve are refered to as macrofauna (Rees 1940, Mare 1942, McIntyre 1969, Rees 1984, Bachelet 1990, Schlacher and Woolridge 1996). Schwinghamer (1981, 1983) has shown that the distribution of biomass among different size classes of benthic organisms is a conservative structural feature and relates to pre-dominant life-styles of the size groups for microbes, meio- and macrofauna. In tropical tidal flats, however, the size range of benthic fauna deviates in two ways from the scheme developed by Schwinghamer. First, the abundance of megabenthos, which can not be quantified by the use of sieves, and second by the abundance and diversity of infauna intermediate in size between meio- and macrofauna, the so-called "mesofauna" (Dittmann 1995). Alongi (1989) also put forward a concept of a high proportion of opportunistic, small-sized species in tropical infaunal assemblages.
In mangroves, epibenthic species dominate the macrofauna. Crustaceans and gastropods are especially abundant and rich in species (Macnae and Kalk 1962, Macnae 1967, 1968, Sasekumar 1974, Frith et al. 1976, Alongi and Sasekumar 1992, Guerreiro et al. 1996) and mangrove crabs play an essential role for leaf litter degradation in these systems (Robertson 1986, Micheli, 1993). Meiofauna, on the other hand, occurs in low abundances in mangrove sediments (Alongi 1987a, b, c). Further infaunal components of mangrove sediments were often recorded in a qualitative way only or with the use of a 1mm sieve size. Small-sized infauna had not yet been studied in mangrove sediments. These organisms could play an important role in the food web of mangrove ecosystems, as small macrofauna can constitute a major food source for juvenile fish and prawns, which use mangroves as nursery sites (Robertson 1988, Daniel and Robertson 1990). Therefore, abundance and distribution of small-sized infauna were surveyed in mangrove and adjacent tidal flat sediments.
The study sites were arranged along a transect, as earlier studies on mangrove fauna have shown clear zonation patterns in relation to tidal elevation, forest type and sediment properties (Macnae and Kalk 1962, Sasekumar 1974, Frith et al. 1976, Wells 1983). These surveys crossed through mangroves into ambient sand- and mudflats and generally showed lower densities and species numbers in mangroves than tidal flat sediments (see Alongi 1989 and Alongi and Sasekumar 1992 for further references). Only Frith et al. (1976) found more species in a mangrove than mudflat and Schrijvers et al. (1995a) recorded higher macrobenthic densities in mangrove than sandflat sediments. This study presents the first quantitative records of small-sized infauna from mangrove sediments.
Materials and Methods
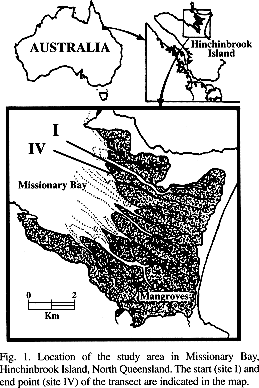
The study was carried out in the mangroves of Missionary Bay at Hinchinbrook Island (18°13'S; 146°11'E), North Queensland, Australia. Four transect sites were arranged along the boardwalk of the Australian Institute of Marine Science at Coral Creek (Fig. 1), covering a distance of about 500 m. Site I was ca. 10 m inside the forest from Coral Creek. This site was frequently inundated and the sediment black and solid mud. The second and third sites lay further within the mangrove forest. Site II was on a more elevated part of the mangrove peninsula, about halfway along the boardwalk. The mangrove canopy was thin here so that more light reached the sediment, which was a dry and firm mud rich in mangrove detritus. Site III was ca. 10 m beyond the end of the boardwalk. Here, the sediment was rather soft mud. Site IV was located in a mudflat seaward of the mangrove fringe. At this site, the sediment was knee-deep mud which also contained shell fragments. Mudwhelks (mainly Cerithidea cingulata) and Theodoxus oualaniensis were abundant on the sediment surface of the mudflat. The macrofauna in the mangrove forest was dominated by sesarmid crabs (Robertson 1986, Micheli 1993). The forest consisted of several Rhizophora spp. and Brugiera gymnorrhiza, Ceriops tagal co-occurred at the more elevated site and Avicennia marina grew on the mangrove fringe towards the mudflat. Detailed accounts on this mangrove area and environmental parameters of the studied sites are available from Alongi (1987a, 1988), Boto and Wellington (1984) and Boto et al. (1989).
Samples were taken on 17 November 1988 and in 1989 on 3 May, 2 August and 13 October. The mudflat site IV could not be sampled in November 1988 due to the incoming tide. At every sampling occasion, six replicate samples were taken at each site. To extract sediment samples from the mud, a cut off syringe (50ml) with piston was used as a corer. This corer had a surface area of 6.61 cm² and sediment was extracted to 5 cm depth. Usually several trials were necessary to get an entire sediment core, as crab burrows were often hit with the corer. The samples were sieved over 0.25 mm mesh size to assess the small sized infaunal organisms (mesofauna sensu Dittmann 1995), which would be lost by the use of larger mesh sizes traditionally taken for macrobenthic samples (see also Bachelet 1990 and Schlacher and Woolridge 1996 for adequate choice of sieve size). The animals were sorted alive and counted under a dissecting microscope. The sediment contained many small particles of mangrove detritus, which made the sorting very cumbersome. Distinguishable species were always recorded separately and determined to the lowest possible taxonomic level. For polychaetes, genus or species could be identified, while most gastropods were treated on family level with few species named. Oligochaeta and nemertines could not be determined further.
Species similarity between sites was calculated by the Sørensen Index QS (Sørensen 1948). To test for significant differences in abundances between the sites and sampling months, Kruskal-Wallis H-test and the U-test by Wilcoxon, Mann and Whitney were applied (Sachs 1992). Multivariate analyses were carried out using the PRIMER software package by the Plymouth Marine Laboratory. Cluster analysis (Bray-Curtis similarity, group-average linkage) were made on double square root transformed data in search of species assemblages. Differences in assemblages between sites were tested with ANOSIM and species discriminating sample groupings were analysed with SIMPER (Clarke 1993).
Results
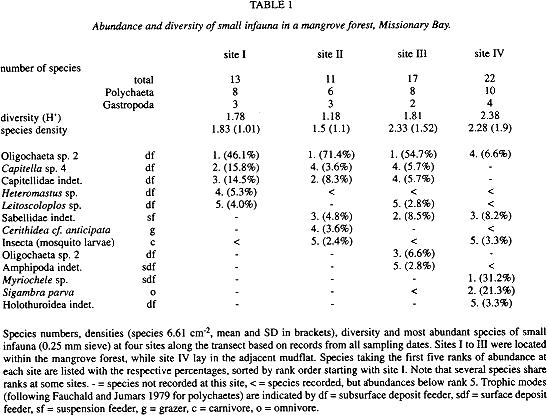
Species: Based on the records from all samples and sites, 39 taxa were distinguished during this survey, with polychaetes richest in species (19), followed by gastropods (9). Species numbers differed between transect sites and most species were recorded in the mudflat (Table 1). Diversity was higher at the mudflat site than in the mangroves, with lowest diversity at site II. On average, only two species were found per sample and species densities were low at all sites (Table 1). Species similarity was higher between the mangrove sites I, II and III (QS values of 0.43 to 0.5) than between the mudflat and mangrove sites I and II (QS = 0.29 and 0.3 resp.). However, the mudflat and the adjacent mangrove site III were similar in their species composition (QS = 0.46).
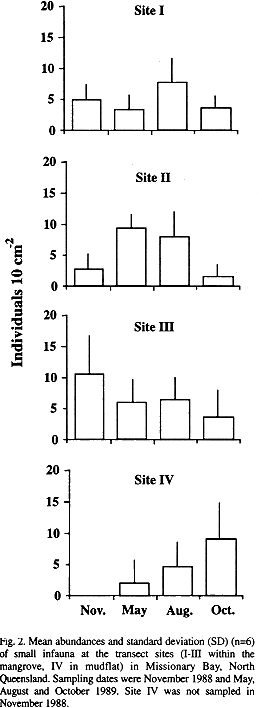
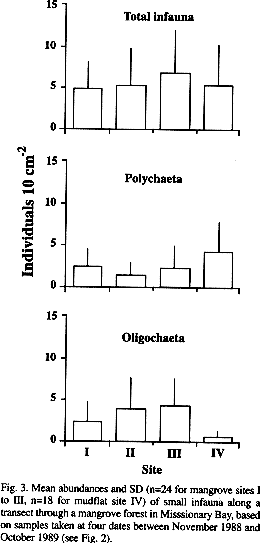
Abundances and distribution: Abundances of small infauna rarely exceeded 10 ind. 10 cm-² along the studied transect (Fig. 2). Extrapolating individual densities to m² (beware of high variations and unknown spatial distributions), the overall mean amounts to 5 477 ind m-², varying from 4 796 (site I) to 6 687 ind m-² (site III).
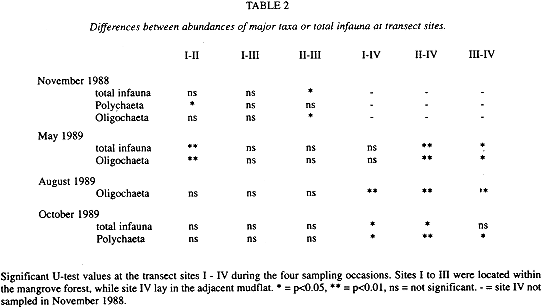
There was little variation in abundances over time at the single transect sites (Fig. 2). Only at site II abundances were significantly higher (H-test, p<0.05) in May and August than in October, while at site IV abundances were significantly higher (H-test, p<0.05) in October than in May.
Comparing abundances of total infauna or major taxa at the single months revealed mainly significant differences between the mangrove sites and the mudflat site, while few significant differences occurred between single mangrove sites (Table 2). Here, the only differences were recorded in relation to site II. Most of the differences between mangrove and mudflat sites were due to abundances of oligochaetes and polychaetes.
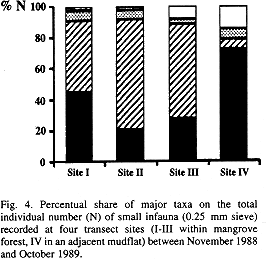
Based on the entire survey, abundances of total infauna did not vary between sites. Polychaeta occurred throughout the transect with highest abundances in the mudflat. Oligochaeta were mainly confined to the mangrove sites (Fig. 3). This is also reflected in the share of these taxa on the total individual numbers per site. Polychaeta accounted for most of the individuals at the mudflat site, while Oligochaeta made up 46 to 71 % of the individuals at the mangrove sites (Fig. 4). Gastropoda recorded in the infaunal samples occurred in almost the same abundance throughout the transect. Mosquito larvae were recorded in the samples of all sites. Further taxa (nemertines, bivalves, amphipods, ophiurids, holothurids, pycnogonids, acari) were restricted to the mangrove fringe (site III) and the adjacent mudflat.
Looking at single species, an oligochaete species was most abundant at the mangrove sites, while the first three ranks of abundance at the mudflat site were taken by polychaetes (Table 1). Rank orders of abundance of species differed at each site. The species taking the first five ranks listed in Table 1 comprised 94 % of the individuals at site II, about 86 % at site I and III and 74 % at site IV, where the remaining 16 species shared rank six. The first five ranks comprised only deposit feeders at site I, but encompassed five different trophic modes at site IV (Table 1).
Within the polychaetes, distinct distribution patterns occurred along the transect. Capitellidae were almost restricted to the mangrove sites, where they accounted for 45 (site III) to 82 % (site I) of polychaete abundances, compared to 2 % in the mudflat. Sabellidae were recorded frequently at all sites. Sigambra parva (Pilargidae) occurred almost exclusively in the mudflat, where it accounted for 30 % of the polychaetes, and at site III nearby (7 %). Myriochele sp. (Oweniidae) was confined to the mudflat and accounted for 43 % of the polychaetes recorded here.
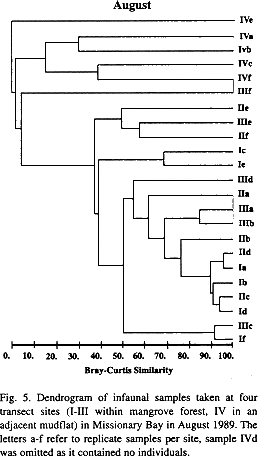
Multivariate analyses detected distinct infaunal assemblages of mangrove and mudflat sites. The mangrove sites were similar to each other, only site III formed a defined cluster in November as did site I in October. The mudflat site IV gave distinct clusters in the dendrograms for May, August (Fig. 5) and October. With the greater similarities between mangrove sites, the ANOSIM results (May: R=0.203, p=0.006, August: R=0.319, p=0.000; October: R=0.305, p=0.006) show only slightly greater similarities within than between sites. S. parva, Myriochele sp., indetermined oligochaetes and polychaetes of the families Sabellidae and Capitellidae contributed most to the dissimilarities between the mudflat and mangrove sites.
Discussion
The survey presented here identified small-sized infauna as an abundant component of benthic fauna in mangrove sediments. The differences in the spatial distribution of small-sized infauna further confirmed previous studies showing a distinct macrofauna of mangroves and mudflats.
Species numbers recorded at the transect sites were low, but within the range of records from other mangroves and tidal flats compiled by Alongi and Sasekumar (1992). Nandi and Choudhury (1983) also found low species numbers of macrofauna in mudflats near mangroves of the Sunderbans. Other tidal flats in the tropics, however, have very high species numbers (Macnae and Kalk 1962, Day 1974, Reise 1991, Dittmann 1995). The number of species and species diversity tends to increase towards the lower intertidal (Vohra 1971, Guerreiro et al. 1996, Dittmann 2000). This trend is supported by the distribution of species along the transect through the mangroves presented here. The richness of polychaete species and their high species number in sand- and mudflats adjacent to mangroves are in agreement with records from other parts of the world (Day 1974, Frith et al. 1976, Wells 1983, Schrijvers et al. 1995a, Guerreiro et al. 1996).
The abundances of small infauna recorded here are 10 to over 60 times higher than previous records of macrobenthos from mangrove sediments (see Alongi and Sasekumar 1992) and only comparable to macroinfaunal densities from East-African mangroves (Schrijvers et al. 1995a). They are, however, lower than abundances of small infauna in other tidal flats of the North Queensland coast (Dittmann 1995, 2000), where mesofaunal abundances even exceed the highest macrobenthic abundances previously known for tropical tidal flats (Vargas 1987). Thus, while small infauna is more abundant than larger macrofauna in mangrove sediments, it does not reach abundances recorded from tidal flats.
The samples were taken at the beginning (November 1988) and end (May 1989) of the wet season and during the dry season (August and October 1989), but no temporal variations relating to seasons were recorded. Abundances were lower in October at the elevated mangrove site II, but higher at the mudflat site in the same month compared to the other sampling dates. Without further studies it will not be possible to relate this finding to any seasonality in abundances or population dynamics of species occurring at the respective sites. Temporal variations in infaunal numbers rarely showed consistent patterns with monsoon climate (Nandi and Choudhury 1983, Alongi 1989, Alongi and Sasekumar 1992), although species-specific responses to dry and wet seasons were reported by Vargas (1989, 1996) and Broom (1982). Daniel and Robertson (1990) related temporal differences in epibenthos of mangrove waterways to hydrographic changes with the wet- and dry season. Alongi (1988) reported seasonal differences of microbial biomass, chlorophyll-a and further sediment factors in the low- and mid intertidal region of the study site in Missionary Bay. Thus, the availability of food for small-sized infauna could have varied over the year at the different transect sites.
Several factors have been proposed to regulate infaunal densities in mangrove sediments: physical forces, competition with epifauna, predation by epifauna, poor quality of food and chemical defense by mangroves (Alongi 1989, Alongi and Sasekumar 1992). These factors could also account for the differences in spatial distribution and species composition found along the transect and are discussed in the following.
Sediment properties vary with tidal elevation and forest type and thus affect the distribution of meio- and macrofauna in mangroves (Alongi 1987a, Guerreiro et al. 1996). The oxic layer, judged by the colour of the sediment, was slightly thicker in the mudflat than at the mangrove sites. Bioturbation and oxygen production by a microphytobentic layer could have contributed to the higher diversity and higher abundances of polychaetes in the mudflat. Alongi (1987a) recorded little variation in sediment properties (% silt, grain size, water content) at a mid- and low-intertidal site in Missionary Bay, which relate to my transect sites I and IV. However, he measured higher values of total organic matter as well as organic C and N at the mid-intertidal site, which would correspond with the distribution of oligochaetes and capitellids. Oligochaetes and capitellid polychaetes were most abundant at the mangrove sites and their occurrence is often related to muddy sediments rich in organic matter (Pearson and Rosenberg 1978). These findings from Missionary Bay are therefore in accordance with records of Schrijvers et al. (1995a), who found a correlation of macrobenthic oligochaetes with % mud and organic matter in mangrove sediments.
Interactions between endo- and epifauna have rarely been studied in mangrove forests, therefore their effect on the abundance and distribution of small infauna cannot be fully assessed. Most mangrove crabs feed on leaves, microbes, detritus and carrion, while few species are classified as carnivore (see Alongi and Sasekumar 1992, Schrijvers et al. 1996). Juvenile penaeids prey upon small macrofauna (Robertson 1988). Small infauna could indeed provide a rich food source for juvenile fish and prawns, as they are easily accessible at the sediment surface where they occur in great numbers. Given the higher abundance of small than larger infauna, trophic interactions and food webs of mangrove ecosystems have to be further studied.
The uptake of bacteria and microalgae by fiddler crabs and gastropods (Dye and Lasiak 1987) could imply a food competition with infauna and indeed, Schrijvers et al. (1995b, 1996) found a density increase of meiobenthic oligochaetes in exclusion cages as well as a resource competition between epibenthos and amphipods. In my study, amphipods occurred only at the mangrove fringe and mudflat, but it is not known whether this distribution results from competitive interactions or is due to sediment properties. Along the transect, a greater diversity of trophic modes at the mudflat than the mangrove sites could reflect a wider range of available resources outside of the mangroves. In fact, low abundance of protozoa, microalgae and meiofauna recorded in mangrove sediments provide a poor quality of food there (Alongi 1987a, 1988, 1989), contrasting with a high density of meiofauna and an abundance and productivity of benthic diatoms in tropical tidal flats (Swennen et al. 1982, Guerreiro et al. 1996, Dittmann 1995, 2000).
Alongi and Sasekumar (1992) discuss that tannins leaching from mangrove detritus could regulate infaunal densities as it was shown for meiofauna (Alongi 1987c). However, tannin concentrations increase with depth (Boto et al. 1989) and it is unlikely that the lower values near the sediment surface could inhibit the small infauna occurring there.
While the infauna of the mangrove sites at the studied transect were more similar to each other, the mudflat site differed in abundances and species composition and was distinguished from the other sites by multivariate analysis. These distinct faunal compositions corroborate findings from macrobenthic surveys in other Indo-Pacific mangroves, which showed a defined fauna for sand- and mudflats and a fauna peculiar to mangroves (Day 1974, Sasekumar 1974, Frith et al. 1976, Schrijvers et al. , 1995a, Guerreiro et al. 1996). The agreement between the cited macrobenthic surveys and the records of small infauna presented here substantiates that mangroves and tidal flats constitute two different habitats.
Acknowledgements
This study was carried out during a postdoctoral fellowship funded by the German Science Foundation (DFG grant III 02-Di 396/1-2). I am grateful to the Australian Institute of Marine Science for its hospitality and providing excellent research facilities. My thanks also go to the crew of the "Harry Messel" and D. Alongi, K. Boto, F. Micheli and A. Robertson for introducing me to the mangrove environment and help in the field. J.A. Vargas and two anonymous reviewers gave helpful comments to the manuscript.
Resumen
Para evaluar la presencia, distribución espacial y composición de especies de la infauna pequeña en una costa de manglar, se tomaron muestras con barrenos a lo largo de un transecto en la bahía Missionary, isla Hinchinbrook, noreste de Australia. Se arreglaron tres sitios es el bosque de manglar y un sitio se localizó en un banco de lodo adyacente. Los sitios se muestrearon cuatro veces entre noviembre de 1998 y octubre de 1989. Basados en los registros de todas las muestras y sitios, se identificaron 39 táxones. La diversidad (H') varió de 1.18 a 2.38. Entre todo, las abundancias totales de la infauna pequeña (retenida en un tamiz de 0.25 mm) alcanzaron un valor promedio de 5477 ind. m -2,con poca variación por todo el transecto o por tiempo. Los números de especies y la diversidad fueron más altos en el banco de lodo que en los sitios de manglar. La composición taxonómica cambio entre el bosque de manglar y el banco de lodo: Oligochaetos fue má abundante en los sedimentos de manglar, mientras que Polychaeta dominó en el banco de lodo. De los poliquetos, Capitellidae estuvo casi restringida a los sitios de manglar, Sabellidae se registró con frecuencia en todos los sitios y Sigambra parva y Myriochele sp. se confirmaron al banco de lodo y al borde del manglar. Estas especies contaron también para las diferencias entre los sitios. Los análisis multivariados mostraron un conjunto diferente en el banco de lodo en comparación con los sitios del manglar. Este estudio mostró que la infauna pequeña es un componente abundante de sitios de manglar, y que ha sido previamente subestimado.
References
Alongi, D.M. 1987a. Intertidal zonation and seasonality of meiobenthos in tropical mangrove estuaries. Mar. Biol. 95: 447-458. [ Links ]
Alongi, D.M. 1987b. Inter-estuary variation and intertidal zonation of free-living nematode communities in tropical mangrove systems. Mar. Ecol. Prog. Ser. 40: 103-114. [ Links ]
Alongi, D.M. 1987c. The influence of mangrove-derived tannins on intertidal meiobenthos in tropical estuaries. Oecologia 71: 537-540. [ Links ]
Alongi, D.M. 1988. Bacterial productivity and microbial biomass in tropical mangrove sediments. Microb. Ecol. 15: 59-79. [ Links ]
Alongi, D.M. 1989. Ecology of tropical soft-bottom benthos: a review with emphasis on emerging concepts. Rev. Biol. Trop. 37: 85-100. [ Links ]
Alongi, D.M. & A. Sasekumar. 1992. Benthic Communities, p. 137-171. In A.I. Robertson & D.M. Alongi (eds.). Tropical Mangrove Ecosystems. Coastal and Estuarine Studies 41. American Geophysical Union, Washington, D.C. [ Links ]
Bachelet, G. 1990. The choice of a sieving mesh size in the quantitative assessment of marine macrobenthos: a necessary compromise between aims and constraints. Mar. Environm. Res. 30: 21-35. [ Links ]
Boto, K.G. & J.T. Wellington. 1984. Soil characteristics and nutrient status in a northern Australian mangrove forest. Estuaries 7: 61-69. [ Links ]
Boto, K.G., D.M. Alongi & A.L.J. Nott. 1989. Dissolved organic carbon-bacteria interactions at sediment-water interface in a tropical mangrove system. Mar. Ecol. Prog. Ser. 51. 243-251. [ Links ]
Broom, M.J. 1982. Structure and seasonality in a Malaysian mudflat community. Estuarine, Coastal Shelf Sci. 15: 135-150. [ Links ]
Clarke, K.R. 1993. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 18: 117-143. [ Links ]
Daniel, P. & A.I. Robertson. 1990. Epibenthos of mangrove waterways and open embayments: Community structure and the relationship between exported mangrove detritus and epifaunal standing stocks. Estuarine, Coastal Shelf Sci. 31: 599-619. [ Links ]
Day, J.H. 1974. The ecology of Morrumbene estuary, Moçambique. Trans. roy. Soc. S. Afr. 41: 43-97. [ Links ]
Dittmann, S. 1995. Benthos structure on tropical tidal flats of Australia. Helgoländer Meeresunters. 49: 539-551. [ Links ]
Dittmann, S. 2000. Zonation of benthic communities in a tropical tidal flat of north-east Australia. J. Sea Res. 43: 33-51. [ Links ]
Dye, A.H. & T.A. Lasiak. 1987. Assimilation efficiencies of fiddler crabs and deposit feeding gastropods from tropical mangrove sediments. Comp. Biochem. Physiol. 87: 341-344. [ Links ]
Fauchald, K. & P.A. Jumars. 1979. The diet of worms: A study of polychaete feeding guilds. Oceanogr. Mar. Biol. Ann. Rev. 17: 193-284. [ Links ]
Frith, D.W., R. Tantanasiriwong & O. Bhatia. 1976. Zonation of macrofauna on a mangrove shore, Phuket Island. Phuket Mar. Biol. Cent., Res. Bull. 10: 1-37. [ Links ]
Guerreiro, J., S. Freitas, P. Pereira, J. Paula & A. Macia. 1996. Sediment macrobenthos of mangrove flats at Inhaca Island, Mozambique. Cah. Biol. Mar. 37: 309-327. [ Links ]
Macnae, W. 1967. Zonation within mangroves associated with estuaries in North Queensland. Estuaries 83: 432-441. [ Links ]
Macnae, W. 1968. A general account of the fauna and flora of mangrove swamps and forests in the Indo-West-Pacific region. Adv. mar. Biol. 6: 73-270. [ Links ]
Macnae, W. & M. Kalk. 1962. The fauna and flora of sand flats at Inhaca Island, Moçambique. J. Animal Ecol. 31: 93-128. [ Links ]
Mare, M.F. 1942. A study of a marine benthic community with special reference to the micro-organisms. J. mar. biol. Ass. U.K. 25: 517-554. [ Links ]
McIntyre, A.D. 1969. Ecology of marine meiobenthos. Biol. Rev. 44: 245-290. [ Links ]
Micheli, F. 1993. Feeding ecology of mangrove crabs in north eastern Australia: mangrove litter consumption by Sesarma messa and Sesarma smithii. J. Exp. Mar. Biol. Ecol. 171: 165-186. [ Links ]
Nandi, S. & A. Choudhury. 1983. Quantitative studies on the benthic macrofauna of Sagar Island, intertidal zones, Sunderbans, India. Mahasagar-Bull. Nat. Inst.Oceanogr. 16: 409-414. [ Links ]
Pearson, T.H. & R. Rosenberg. 1978. Macrobenthic succession in relation to organic enrichment and pollution of the marine environment. Oceanogr. Mar. Biol. Ann. Rev. 16: 229-311. [ Links ]
Rees, C.B. 1940. A preliminiary study of the ecology of a mud-flat. J. mar. biol. Ass. U.K. 24: 185-199. [ Links ]
Rees, H.L. 1984. A note on mesh selection and sampling efficiency in benthic studies. Mar. Poll. Bull. 15: 225-229. [ Links ]
Reise, K. 1991. Macrofauna in mud and sand of tropical and temperate tidal flats, p. 211-216. In M. Elliott and J.-P. Ducrotoy (eds.). Estuaries and Coasts: Spatial and Temporal Intercomparisons. Olsen and Olsen, Fredensborg. [ Links ]
Robertson, A.I. 1986. Leaf-burying crabs: their influence on energy flow and export from mixed mangrove forests (Rhizophora spp.) in northeastern Australia. J. Exp. Mar. Biol. Ecol. 102: 237-248. [ Links ]
Robertson, A.I. 1988. Abundance, diet and predators of juvenile banana prawns Penaeus merguiensis in a tropical mangrove estuary. Aust. J. Mar. Freshw. Res. 39: 467-478. [ Links ]
Sachs, L. 1992. Angewandte Statistik. Springer , Heidelberg. [ Links ]
Sasekumar, A. 1974. Distribution of macrofauna on a Malayan mangrove shore. J. Animal. Ecol. 43: 51-69. [ Links ]
Schlacher, T.A. & T.H. Woolridge 1996. How sieve mesh size affects sample estimates of estuarine benthic macrofauna. J. Exp. Mar. Biol. Ecol. 201: 159-171. [ Links ]
Schwinghamer, P. 1981. Characteristic size distribution of integral benthic communities. Can. J. Fish. Aquat. Sci. 38: 1255-1263. [ Links ]
Schwinghamer, P. 1983. Generating ecological hypotheses from biomass spectra using causal analysis: a benthic example. Mar. Ecol. Prog. Ser. 13: 151-166. [ Links ]
Schrijvers, J., D. Van Gansbeke & M. Vincx. 1995a. Macrobenthic infauna of mangroves and surrounding beaches at Gazi Bay, Kenya. Hydrobiologia 306: 53-66. [ Links ]
Schrijvers, J., J. Okondo, M. Steyaert & M. Vincx. 1995b. Influence of epibenthos on meiobenthos of the Ceriops tagal mangrove sediment at Gazi Bay, Kenya. Mar. Ecol. Prog. Ser. 128: 247-259. [ Links ]
Schrijvers, J., H. Fermon & M. Vincx. 1996. Resource competition between macrobenthic epifauna and infauna in a Kenyan Avicennia marina mangrove forest. Mar. Ecol. Prog. Ser. 136: 123-135. [ Links ]
Sørensen, T. 1948. A method of establishing groups of equal amplitude in plant sociology based on similarity of species content. K. Danske Vidensk. Selsk. 5: 1-34. [ Links ]
Swennen, C., P. Duiven & A.L. Spaans. 1982. Numerical density and biomass of macrobenthic animals living in the intertidal zone of Surinam, South America. Neth. J. Sea Res. 15: 406-418. [ Links ]
Vargas, J.A. 1987. The benthic community of an intertidal mud flat in the Gulf of Nicoya, Costa Rica. Description of the community. Rev. Biol. Trop. 35: 299-316. [ Links ]
Vargas, J.A. 1989. Seasonal abundance of Coricuma nicoyensis Watling and Breedy, 1988 (Crustacea: Cumacea) on a tropical mud flat. Rev. Biol. Trop. 31: 207-211. [ Links ]
Vargas, J.A. 1996. Ecological dynamics of a tropical intertidal mudflat community, p. 355-371. In K.F. Nordstrom and C.T. Roman (eds.). Estuarine Shores: Evolution, Environments and Human Alterations. Wiley, Chichester. [ Links ]
Vohra, F.C. 1971. Zonation on a tropical sandy shore. J. Animal. Ecol. 40: 679-709. [ Links ]
Wells, F.E. 1983. An analysis of marine invertebrate distributions in a mangrove swamp in northwestern Australia. Bull. Mar. Sci. 33: 736-744. [ Links ]
1 Zentrum für Marine Tropenökologie, Fahrenheitstr. 1, 28359 Bremen, Germany. Fax +49-421-2208 330; sdittmann@uni-bremen.de












 uBio
uBio