Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.48 n.1 San José Mar. 2000
in the Opa reservoir, Ile-Ife, Nigeria
*O.O. Fawole1and G.A.O. Arawomo2
Received 11-I-1999. Corrected 11-XI-1999. Accepted 18-XI-1999.
Abstract
The fish Sarotherodon galilaeus was collected in Opa reservoir, Obafemi Awolowo University, Ile-Ife, Nigeria. Sampling began in January, 1992 and extended until March, 1994. The fishing methods employed for collecting the 853 specimens were gillnetting and castnetting. Sex ratio was approximately 1:1 (male: female). Length at maturity was 12.5 cm (males) and 11.7 cm (females). The fecundity was 1 048 ± 785 (604-2173, n= 117) with an egg diameter of 2.49 ± 1.81 (1.0 - 4.5, n = 117) mm. The gonado-somatic index was 0.33 ± 0.21 (0.09 - 1.34, n = 275) (males) and 1.88 ± 1.01 (0.33 - 6.19, n = 282) (females). The species breeds year-round (mature specimens occurred in samples collected throughout the 27-month study period). This fish is a biparental mouth booder.
Key words
Reproductive biology, Sarotherodon galilaeus, fecundity, gonado-somatic index, biparental.
Sarotherodon galilaeus (Artedi) is one of the dominant and endemic cichlids of Nigerian inland water bodies (Petr. 1974).
Cichlids are perhaps the most economically important fishes of tropical African freshwaters (Fryer and Iles 1972). The fecundity and in most cases, some other aspects of the reproductive biology of cichlids have been documented by Akintunde (1976), Fagade and Adebisi (1979), Fagade (1983), Adebisi (1987) and many others. The knowledge of fish fecundity is needed in establishing its production potential and consequently its exploitation and management rationale.
This work aims at providing further information on the fecundity of S. galilaeus especially in the Opa reservoir.
Materials and Methods
Study area: The study area was the Opa reservoir which was impounded in 1978. The major tributaries are rivers Opa, Obudu and Esinmirin. The reservoir has a catchment area of about 116 km2 (4o 31'E to 4o 39'E, 7o 21' N to 7o 35'N) in Osun State, Nigeria. The minimum depth is 0.95m while the maximum depth is 6.4m.
During the period of study, sampling for S. galilaeus began in January, 1992 and extended until March, 1994. The two fishing methods employed were gillnetting and cast netting. The mouth of fishes caught by cast netting were examined for egg incubation and fish frys. In the laboratory, each specimen was measured, weighed and split open. The sex and the stages of development of the gonad were determined by visual inspection and graded according to Nikolsky's (1963) scale. The gonads were removed and weighed. The gonad weight expressed as a percentage of the fish somatic weight (Sturm 1978) was used as the gonado-somatic index (GSI). The GSI was used to follow the seasonal changes in the gonads of S. galilaeus in Opa reservoir. The mature ovaries (stages III and IV) were preserved in Gilson's fluid and agitated at intervals. The surrounding ovarian tissues were removed and the number of eggs in each pair of ovaries were determined by direct enumeration. The egg diameters were measured using ocular micrometre in a binocular microscope.
Results
Sex ratio: The result shows that, of the 853 specimens that had observable gonads, 413 were males while 440 were females, that is, a ratio of 1:1.07 (male: female).
The sex ratios of the 1992, 1993 and 1994 populations, 1:1.04, 1:1.11 and 1:0.97 were similar and they followed the same pattern. These deviations from the expected 1:1 ratio were not statistically significant.
The sex ratios for offshore and inshore catches also followed the same pattern as the deviations were not statistically significant.
Maturity size: In this study, the minimum sizes of the mature male and female specimens were taken as the maturity sizes. The smallest male with a mature gonad had a total length of 12.5cm, a standard length of 9.4cm and a weight of 36g while the smallest female with a mature gonad had a total length of 11.7cm, a standard length of 8.7cm and a weight of 34g.
Fecundity: Only the largest eggs were used in estimating the fecundity of the species. The total lengths of the specimen examined ranged from 11.7cm to 31.0cm, the standard lengths ranged from 8.7cm to 24.2cm while the weights ranged from 34g to 578g. The total fecundity in the ovary ranged from 604 eggs in a fish of total length 15.6cm, standard length 10.9cm and weight 115g to 2173 eggs in a fish of total length 31.0cm, standard length 24.2cm and a weight of 578g.
The highest fecundity was observed in the biggest specimen while lowest fecundity of 604 eggs was not observed in the smallest specimen. The smallest specimen had a fecundity of 611 eggs.
The mean relative fecundity was 5.17 eggs per gram body weight and this ranged from 2.88 eggs to 17.97 eggs per gram body weight.
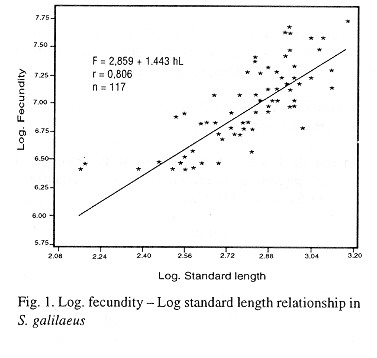
The equation describing the relationship between fecundity and standard length is given as:
F = aLb (Bagenal, 1967)
where F = Fecundity
L = Standard length in cm
b = Slope of the regression line (regression constant).
a = Intercept of the regression with the y - axis (regression coefficient).
Through a logarithm transformation, the equation becomes
lnF = b lnL + a
Thus, the relationship between fecundity and standard length (Fig.1) was described by the regression equation.
The correlation coefficient (r) was 0.806 (p = 0.001, n = 117). This indicates a high correlation between fecundity and standard length.
Egg sizes: In each mature ovary, the eggs were of varying sizes, i.e. the egg size was not uniform. The mature eggs were brown in colour. The diameter of the eggs varied in fish of the same length or weight. The egg diameters ranged from 1.0mm to 4.5mm.
Gonad Reproductive Stages: Six stages of gonad development were observed in the specimens of S. galilaeus examined. The stages are:
Stage I Immature, inactive
Stage II Immature, developing
State III Maturing
Stage IV Mature (Ripe)
Stage V Ripe running
Stage VI Spent.
This suggests that ripe and unripe specimens occurred throughout the study period.
The gonado-somatic indices (GSI) ranged from 0.09 to 1.34 in males and 0.33 to 6.19 in females.
Breeding habits: During the study period, thirteen specimens comprising five males and eight females, were caught with eggs in their mouth. These ranged from a fish of total length 16.7, standard length 12.8cm and a body weight of 98g to a fish of total length 24.3cm, standard length 18.4cm and body weight of 267g. The total number of eggs found in the mouth ranged from 7 to 208. The results showed that the eggs found in the mouth were relatively few in number suggesting that some of them might have been lost during capture.
Fish frys were however not found in the mouth throughout the study period.
Discussion
The sex ratio for S. galilaeus in Opa reservoir is approximately one male to one female as the deviation from the expected 1:1 (male: female) was not significant. In Iita lake in Ibadan, Fagade et al. (1984) observed an overall sex ratio of 6 males to 10 females indicating a preponderance of females of S. galilaeus. Fryer and Iles (1972) reported a sex ratio of 2 males to 1 female of S. galilaeus in lake Rudolf. The results obtained in this study indicates that for every female, there is a male specimen.
The smallest mature male had a total length of 12.5cm while the smallest mature female had a total length of 11.7cm. Fagade et al. (1984) reported that in S. galilaeus of Iita lake, the total lengths of the smallest mature male and female were 15.6cm and 10.6cm respectively. Fagade (1979) also reported that the total lengths of the smallest mature male and female were 87mm and 99mm respectively in Tilapia guineensis. The male specimen of S. galilaeus in Opa reservoir matures at about the same size as the female.
In the fecundity studies, the average number of eggs obtained per female was 1,048 while the number of eggs in each mature ovary varied from 604 to 2 173. The result obtained in this study is lower than that observed by other workers for S. galilaeus.Adebisi (1987) obtained a fecundity of 1 452 for a female specimen with total length of 28.4cm. Fagade et al. (1984) reported a fecundity range of 598 and 3 960 for S. galilaeus whose body length ranged from 13.4cm and 24.1cm in IITA lake in Ibadan. Ben-Tuvia (1960) reported Tilapia galilaea whose body length was 32cm as having a fecundity of 5 010.
In this study, fish specimens of the same length or weight had variable fecundities. Bagenal (1957) asserted that fish species exhibit wide fluctuations in fecundity among fish of the same species, size and age. Fagade et al. (1984) suggested that variation in fecundity may be due to differential abundance of food. The wide fluctuations observed in the fecundity of S. galilaeus from Opa reservoir may be attributed to differential feeding success within the members of the population.
The occurrence of eggs of varying sizes is also an indication of multiple spawning by this species. The egg diameter varied in fish of the same length or weight. Fryer and Iles (1972) reported that S. galilaeus had a maximum egg diameter of 2.2mm while Fagade et al. (1984) obtained a range of 0.7mm to 3.6mm for S. galilaeus in Iita lake. The mean egg diameter of 2.49mm obtained in this study is an indication that S. galilaeus produces bigger eggs that those of the same species from the great lakes of East Africa.
Mature ovaries were available all the year round and this is an indication that the fish breeds throughout the year. The GSI for the males were always lower than those of the females. This is associated with the relatively heavier female gonads. In this study, both sexes of S. galilaeus are involved in mouth brooding activities hence the fish species is a biparental mouth brooder. The mouth brooding habit and the ability to reproduce many times a year might be responsible for its relative success in the reservoir.
Acknowledgements
The authors thank the Opa Dam Authority of the Obafemi Awolowo University, Ile-Ife for granting the permission to carry out the research in Opa reservoir.
References
Adebisi, A.A. 1987. The relationships between the fecundities, gonado-somatic indices and egg sizes of some fishes of Ogun River, Nigeria. Arch. Hydrobiol, III: 151 - 156. [ Links ]
Akintunde, E.A. 1976. The biology of Tilapia and Sarotherodon species in Lake Kainji with special reference to S. galilaeus M. Phil. Thesis. University of Ife, Ile-Ife Nigeria. [ Links ]
Bagenal, T.B. 1957. Annual variation in fish fecundity J. Mar, Biol. Ass. U.K. 36: 377 - 382. [ Links ]
Bagenal, T.B. 1967. A short review of fish fecundity in The biological basis of freshwater fish production. Proceedings of IBP Symposium, Oxford, Blackwells, 89 - 111. [ Links ]
Ben-Tuvia, A. 1960. The biology of the Cichlid fishes of Lakes Tiberias and Hulet. Bull. Res. Coun. Israel: 86: 153 - 188. [ Links ]
Fagade, S.O. 1979. Observation on the biology of two species of Tilapia from the Lagos Lagoon, Nigeria. Bull. Inst. Fr. Afr Noire 41: 627 - 653. [ Links ]
Fagade, S.O. 1983. The biology of Chromidotilapia guntheri from a small lake. Arch. Hydrobiol 97: 60 - 72. [ Links ]
Fagade, S.O. & A.A. Adebisi 1979. On the fecundity of Chrysichthys nigrodigitatus (Lacepede) of Asejire dam, Oyo State, Nigeria Nig. J. Nat. Sci. 1: 127 - 131. [ Links ]
Fagade, S.O.; A.A. Adebisi & A.N. Atanda 1984. The breeding cycle of Sarotherodon galilaeus in the IITA lake, Ibadan Nigeria. Arch. Hydrobiol. 100: 493 - 500. [ Links ]
Fryer, G. & T.D. Iles 1972. The Cichlid Fishes of the Great Lakes of Africa. Their Biology and Evolution: Oliver and Boyd, Edinburgh, Scotland 641 pp. [ Links ]
Nikolsky, G.V. 1963. The Ecology of Fishes. Academic Press, London; 352 pp. [ Links ]
Sturm, de L.M.G. 1978. Aspects of the Biology of Scombermorus maculatus (Mitchill) in Trinidad J. Fish Biol. 13: 155-172. [ Links ]
1Department of Pure and Applied Biology, Ladoke Akintola University of Technology, P.M.B. 4000, Ogbomoso, Nigeria.
Fax: 038 - 720750, e-mail: lautech-dvc@informatics.com.ng
2 Department of Zoology, Obafemi Awolowo University, Ile-Ife, Nigeria.