Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Acta Médica Costarricense
versão On-line ISSN 0001-6002versão impressa ISSN 0001-6012
Acta méd. costarric vol.58 no.2 San José Abr./Jun. 2016
Articles
Pseudothrombocytopenia induced by EDTA and chronic inflammatory demyelinating polyneuropathy
2Laboratorio especializado de Hematología Hospital “Dr. Rafael Ángel Calderón Guardia. CCSS
3Universidad de Iberoamérica UNIBE. jipadillacuadra@gmail.com
Thrombocytopenia is a common finding in critically ill patients. This drive the need to perform laboratory studies to clarify its cause. It should be added that the presence of a low platelet count involves a more complex management of the patient in principle because of the increased risk of bleeding. Besides the real causes of thrombocytopenia, there are conditions or laboratory artifacts that can cause falsely low platelet counts and induce unnecessary studies. One such cause of pseudothrombocytopenia is the anticoagulant Ethylenediaminetetraacetic acid (EDTA) used for handling blood samples.
The pseudothrombocytopenia due to Ethylenediaminetetraacetic acid (PT-EDTA) is a recognized phenomenon that occurs frequently and must be differentiated from true thrombocytopenia, in order to avoid additional studies or even to avoid the patient to receive unnecessary platelet transfusions. A case due to this entity is reported in a patient admitted to the Neurocritical Care Unit with the diagnosis of chronic inflammatory demyelinating polyneuropathy for apheresis therapy.
Case report
A 68 year old patient with personal history of chronic inflammatory demyelinating newly diagnosed, who debuted about four weeks ago with ascending weakness and sensory disturbances is presented. He was treated with a single dose of intravenous gammaglobulin and regained his functional capacity. He is readmitted due to an exacerbation, probably triggered by acute diarrhea.
At the admission to the Neurocritical Unit he was thermodynamically stable, with a vital capacity of 50 cc / kg, and shows distress. He also had a decreased sensitivity and muscular strength in distal extremities, with inability to overcome gravity and horizontal displacement movements were preserved. Since was reported thrombocytopenic with a platelet count of 56,000 / mm3, and a description in the blood smear the read “platelet clumps”. On the second day of admission a central internal jugular line catheter was punctured unsatisfactory, and right carotid was canalized by mistake, it drew attention that although 5000 platelets were documented in the blood analysis, the patient did not develop expansive hematoma or procedurerelated bleeding. A left internal jugular central line was placed with a catheter for apheresis. Therefore, pseudothrombocytopenia was suspected and it was decided to try other anticoagulants and amikacin in the sample, which has been shown to reverse the anticoagulant induced pseudothrombocytopenia.
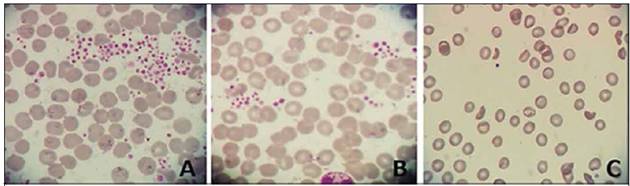
Platelet determination is made with EDTA, citrate and amikacin tubes. An important platelet aggregation is noted in the sample containing EDTA and the dispersion of platelets increases as there is a change to citrate, in which lumps disappear with the use of aminoglycoside (Figure 1). With the same sample it is determined that there are changes in the smear and changes in platelet count (11,000 / mm3 with EDTA to 91,000 / mm3 with amikacin) (Figure 2).
Plasmapheresis with albumin therapy is initiated.
The patient develops a nosocomial lung infection with acute respiratory distress; he requires intubation and management with high-dose vasopressors. HE progresses to refractory septic shock and finally dies.
Discussion
The case illustrates how the incongruity between clinical and laboratory findings (absence of bleeding despite arterial puncture in the presence of a supposed thrombocytopenia), makes the suspicion of an artifact due to laboratory-induced anticoagulant in the sample.
The PT-EDTA was initially described in 1969 by Gowland.1 In 1970, Watkins and Shulman describe a binding factor in the presence of this anticoagulant at low temperaturas.2 There is a widespread use of this anticoagulant for sample handling because of the advantage that it does not distort cell morphology.3 The phenomenon of induced pseudothrombocytopenia is an artifact resulting from the presence of agglutinating anti platelet antibodies.4 The production of platelet clumps causes automatic cell counting systems to provide a falsely low platelet level. In addition, the device misinterprets these lumps as “lymphocytes”. Although it is described primarily in associated to EDTA, it can also occur in blood with heparin or citrate.5
The estimated prevalence is 0.1 to 2% in hospitalized patients and 15-17% in out-patients.6-8
The antibodies described are primarily IgG, although there may be mixtures of IgA, IgM that precipitates at low temperatures.9-13 Although considered an in vitro phenomenon without clinical relevance5 Fukuda Ohashi et al., reported that the presence of the phenomenon could be associated with increased mortality and malignancy.14
It is presumed that the membrane glycoprotein IIB is the site for protein coupling of EDTAdependent antibody on the platelet membrane. Glycoprotein IIb exists alongside IIIa glycoprotein, as a heterodimer dependent of calcium. It is theorized that the dimer dissociates when the concentration of calcium decreases and it re-associates when the availability of the ion increases (Figure 3). The anti-platelet antibody epitope that causes EDTA pseudothrombocytopenia is a cryptoantigen that is only revealed when the IIb glycoprotein dissociates.17
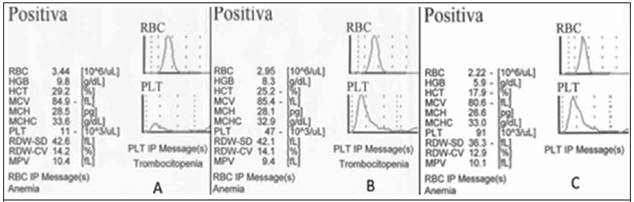
Figure 2 Histograms of peripheral blood. Note the changes in platelet counts with each anticoagulant and the addition of the aminoglycoside: EDTA (A), citrate (B) and amikacin (C).
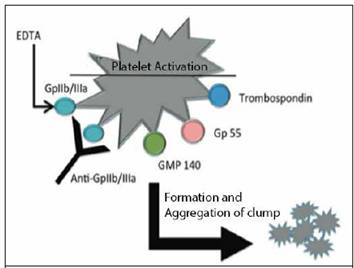
Figure 3 Pathogenesis of thrombocytopenia induced by EDTA. Note the antibody interaction with the heterodimer and expression of GMP 140, gp55 and thrombospondin proteins, with subsequent aggregation and clumping. Adapteb by Lippi et al.18
By this mechanism it is assumed that EDTA, when calcium concentration decreases, it allows the exposure of the binding site of the antibody and the clumping of platelets. In fact, Hyojin et al., achieved to dissociate the platelets upon introduction of calcium chloride in the samples in vitro.17 Recent research have clarified the type of reaction. Lippi et al. describe that autoantibodies react optimally between 0 and 4 degrees Celsius, and after binding with glycoprotein, the expression or activation of antigen the expression of several proteins is stimulated: CD62P, known as protein granulation membrane 140 or GMP140, CD63 known as lysosomal glycoprotein gp55 or type III, and thrombospondin. In the end, this triggers the tyrosine kinase, which brings together groups and platelets, reducing their count.18
Although the presence of an autoimmune disease in the patient could relate to production of antibodies involved in this phenomenon, it was not possible to determine the platelet agglutination due to an specific antibody.
The diagnosis of this condition is established; the first key is the clinical suspicion where the platelet count and the clinic manifestations do not correspond. Suspicion increases if the report includes clumps of platelets, as illustrated in this case. Conditions surrounding the sampling are also important, which is included in the major diagnostic criteria18 (Table 1).
They have recognized several actions to avoid this artifact, including heating the sample to 37 C, neutralization with sodium citrate, or use heparin lithium, ammonium oxalate, theoline, trisodium citrate and addition of aminoglucoside.18 As demonstrated in this case, the disaggregation of the platelet clumps can be achieved by adding amikacin. The mechanism involved in this effect is unknown.19
The differential diagnosis of pseudothrombocytopenia includes causes as defects in the collection process, exposure to drugs (valproate, olanzapine, abciximab) and giant platelet syndrome. The main recommendation to avoid this phenomenon is to analyze the sample almost immediately, or to use other anticoagulants.
It has also tried to reverse it by using substances such as teoline.15 Since aminoglycosides reverse this platelet clumping, it should be considered a tool to distinguish a real thrombocytopenia pseudothrombocytopenia by this mechanism.16
In conclusion, the PT-EDTA should be recognized as an artifact and does not require other diagnostic studies or unnecessary transfusions. Its recognition can be assisted by a simple method, such as addition of amikacin to sample, to reverse platelet clumping. This case shows the importance of the clinical suspicion of the presence of this artifact and to discard true thrombocytopenia.
Affiliation of the authors: 1 Neurocritical Unit, 2 Specialized Laboratory in Hematology Hospital “Dr. Rafael Angel Calderon Guardia. CCSS, 3Iberoamrican University UNIBE.. jipadillacuadra@gmail.com
REFERENCES
1. Gowland E, Kay HE, Spillman JC, Williamson JR. Agglutination of platelet by a serum factor in the presence of EDTA. J Clin Pathol 1969; 22: 460-64. [ Links ]
2. Watkins SP, Shulman NR. Platelet cold agglutinins. Blood 1970; 36: 153-58. [ Links ]
3. Gordan HG, Larson NL. Use of sequestrene as an anticoagulant. Am J Clin Pathol. 1955; 23: 613-18. [ Links ]
4. Berkman N, Michaeli Y, Or R, Eldor A. EDTA-dependent pseudothrombocytopenia: A clinical study of 1 8 patients and a review of the literature. Am J Hematol 1991; 36:195. [ Links ]
5. Bizarro N. EDTA-dependent pseudothrombocytopenia: a clinical and epidemiological study of 112 cases, with 10-year follow-up. Am J Hematol 1995; 50: 103-109. [ Links ]
6. Bartels PC, Schoorl M, Lombarts AJ. Screening for EDTA-dependent deviations in platelet counts and abnormalities in platelet distribution histograms in pseudothrombocytopenia. Scand J Clin Lab Invest 1997; 57: 629-636. [ Links ]
7. Vicari A, Banfi G, Bonini PA: EDTA-dependent pseudothrombocytopaenia: a 12-month epidemiological study. Scand J Clin Lab Invest 1988; 48: 537-542. [ Links ]
8. Zandecki M, Genevieve F, Gerard J, Godon A: Spurious counts and spurious results on haematology analysers: a review. Part I: platelets. Int J Lab Hematol 2007; 29: 4-20. [ Links ]
9. Onder O, Weinstein A, Hoyer LW. Pseudothrombocytopenia caused by platelet agglutinins that are reactive in blood anticoagulated with chelating agents. Blood 1980; 56: 177-182. [ Links ]
10. Pegels JG, Bruynes ECE, Engelfriet CP, von dem Borne AEG. Pseudothrombocytopenia: Animmunologic study on platelet antibodies dependent on ethylene diamine tetraacetate. Blood 1982; 59: 157-161. [ Links ]
11. Hoyt RH, Durie BGM. Pseudothrombocytopenia induced by a monoclonal IgM kappa platelet agglutinin. Am J Hematol 1989; 31: 50-52. [ Links ]
12. Ryo R, Sugano W, Goto M, et al. Platelet release reaction during EDTA induced platelet agglutinations and inhibition of EDTA-induced platelet agglutination by anti-glycoprotein Ilb/IIIa complex monoclonal antibody. Thromb Res 1994; 74: 265-272 [ Links ]
13. Imai H, Nakamoto Y, Miki K, Miyakuni T, Miura AB. Pseudothrombocytopenia and IgA-related platelet agglutinin in a patient with IgA nephritis. Nephron 1983; 34: 154-158. [ Links ]
14. Ohashi-Fukuda N, Inokuchi R, Sato H, Nakamura K, Iwagami M, Wada T, et al. Poorer prognosis with ethylenediaminetetraacetic acid-dependent pseudothrombocytopenia: a single-center case-control study. Medicine (Baltimore). 2015; 94:e674. [ Links ]
15. Ohnuma O, Shirata Y, Miyazawa K. Use of theophylline in the investigation of Pseudo-Thrombocytopenia induced by EDTA. J Clin Pathol. 1988;41:915-17. [ Links ]
16. Sakurai S, Shiojima I, Tanigawa T, Nakahara K. Aminoglycosides prevent and dissociate the aggregation of platelets in patients with EDTA-dependent pseudothrombocytopenia. Br J Haematol 1997; 99: 817-823. [ Links ]
17. Hyojin Chae, Myungshin Kim, Jihyang Lim, Eun-Jee Oh, Yonggoo Kim and Kyungja Han. Novel method to dissociate platelet clumps in EDTA-dependent pseudothrombocytopenia based on the pathophysiological mechanism. Clin Chem Lab Med 2012;50: 1387-1391. [ Links ]
18. Lippi G, Plebani M. EDTA-dependent pseudothrombocytopenia: further insights and recommendations for prevention of a clinically threatening artifact. Clin Chem Lab Med 2012; 50:1281-1285. [ Links ]
19. Zhou X, Wu X, Deng W, Li J, Luo W. Amikacin Can Be Added to Blood to Reduce the Fall in Platelet Count. Am J Clin Pathol 2011; 136: 646-652 [ Links ]
Received: January 04, 2016; Accepted: February 18, 2016











 texto em
texto em