Resumen
Palabras clave
alprazolam, farmacocinética, diferencias por sexo, sujeto sano.
Abstract
Alprazolam, sex-kinetic behavior, healthy subjects.
Introducción
En el caso particular de los psicotrópicos, la información disponible tiende a enfatizar el análisis de los aspectos dinámicos debido a las implicaciones que tienen para los pacientes sus efectos farmacológicos sobre el sistema nervioso central (3,4). Por tanto, el presente ensayo se llevó a cabo para describir el perfil cinético tras la administración de una dosis única de alprazolam (ALZ) y evaluar posibles diferencias cinéticas debidas al factor sexo.
Sujetos, procedimientos y métodos
El protocolo y los procedimientos experimentales se ajustaron a las directrices éticas internacionales (5), el primero fue conocido y aprobado por el Comité Etico de Investigación Clínica del Hospital de la Santa Cruz y San Pablo (Barcelona) y por las autoridades sanitarias españolas. Los sujetos fueron gratificados en términos económicos por su colaboración.
Sujetos : Ocho varones (V) jóvenes y sanos (edad: 22,25±1,48 años, peso: 78,50±10,76 kg y talla: 177,12±8,18 cm) y ocho mujeres (M) jóvenes y sanas (edad: 22,37±1,41 años, peso: 56,64± 4,83 kg y talla: 162,75 ± 3,88 cm). Se incluyeron en este ensayo, tras obtener su consentimiento informado y resultados normales a partir de la exploración física y neuropsiquiátrica, así como de las evaluaciones bioquímicas y hematológicas. En caso de mujeres, se verificó que todas aplicaran medidas eficaces para la anticoncepción (únicamente, sujeto # 09 tomaba gestágenos orales) y que no estuvieran lactando ni embarazadas. Ningún sujeto superó un 28% de índice de masa corporal (6). Adicionalmente, se verificó que ninguno presentaba alguna de las siguientes condiciones : historia previa de alcoholismo o drogodependencia, consumidor importante de bebidas estimulantes (más de 5 porciones diarias de café, té, cola, chocolate, etc.), antecedentes de alergia, idiosincracia o hipersensibilidad a fármacos, serología positiva para la hepatitis B o por virus HIV ; antecedentes o presencia de patología renal, hepática, gastrointestinal, psiquiátrica u otras enfermedades crónicas que comprometan el estado general, antecedentes de cirugía mayor o enfermedad grave en los 3 meses anteriores al estudio, haber participado en el desarrollo de otro ensayo clínico durante el mes precedente al inicio de este estudio, ingesta de medicamentos psicoactivos u otros fármacos que pueden producir interacción con benzodiazepinas (teofilina y derivados, preparados vitamínicos con complejo B, anti-H-1 nootrópicos y otros estimulantes cerebrales) durante las dos semanas previas, así como incapacidad para comunicarse o cooperar con los investigadores. No se aceptó el uso de medicación concomitante a lo largo del ensayo. ]]>
Condiciones Experimentales : Tras ayuno de 10 horas para sólidos y 6 horas para líquidos, a cada sujeto se le administró una dosis oral de 1 mg de alprazolam (ALZ). A través de una bránula insertada en la vena antecubital hubo extracción sanguínea (8mL) a los siguientes tiempos 0, 0, 5h, 1h, 1,5h, 2h, 3h, 4h, 6h, 7h, 9h, 10h, 12h, 24h y 48h postmedicación. Las muestras se depositaron y centrifugaron en tubos heparinizados, se separó el plasma, se depositó en tubos plásticos y se congeló a -20 °C para su análisis posterior. Todos los participantes recibieron una merienda estandarizada a 2h, 4h, 6h, 8h y 10h, se mantuvieron en reposo relativo y la ingesta de líquidos (agua) fue ad libitum a partir de 2h. Se mantuvo una supervisión constante y se procedió con el control incruento de las constantes vitales en forma periódica. Puesto que los sujetos volvían al Servicio a 24h y 48h, se hicieron pruebas de laboratorio y gabinete tanto al iniciar como al finalizar cada sesión.Cuantificación de Fármacos en Plasma : Alícuotas de plasma se descongelaron para proceder con extracción en fase sólida. Procesamiento de la muestra : Extracción: para acondicionamiento 3 ml de metanol/dietilamina 166/1, 3 mL de carbonato de potasio 0.01 g/mL en acetonirilo/H2O (9/1) ; plasma 1 mL+ lavado : 3mL de H2O y 300 mL de acetonitrilo ; elución : 1 mL cloroformo. El eluato de secó por vacío de N2 durante 30 min en baño de +25°C. Condiciones cromatográficas : Fase móvil = solución de tampón hipofosfato de sodio 1mM/metanol 40/60 pH 7. Velocidad de flujo = 1 mL/min. Bomba Waters-Millipore 501®, columna Spherisorb® C18 5m 120x4,6 mm, detector Kontron HPLC 332® para absorción UV con sensibilidad = 1,0 con lectura en 221 nm, registrador Data Module Waters® para lectura cromatográfica y cuantificación del analito.
Validación: Límite de cuantificación= 1.25 ng de ALZ /mL de plasma. La linealidad se valoró con 2,5,5,10,15 y 25ng/ml ; el coeficiente r= 0,9991; la exactitud y precisión se comprobó con concentraciones crecientes de 5 a 25 ng/ml, una recuperación creciente de 83,96 a 96,40% y un coeficiente de variación que osciló entre 1,71 y 4,30%.
Procesamiento de Datos y Análisis : Con las concentraciones plasmáticas (Cp) a cada tiempo de lectura se graficaron los perfiles para cada grupo según el sexo. De cada sujeto se identificó la concentración máxima Cmax y el tiempo de la C max (T max ), y se calculó el tiempo de vida media de eliminación (T1/2 ) y el área Bajo la Curva de 0-48 h (ABC 0-48 ) aplicando el programa PK-calc (7). Los datos se agruparon por sexo, se comparó la Cp a cada tiempo de lectura y los parámetros cinéticos aplicando test del t (prueba de Mann-Whitney para Tmax) ; y cualquier valor p<0,05 se consideró como significativo estadísticamente.
Resultados
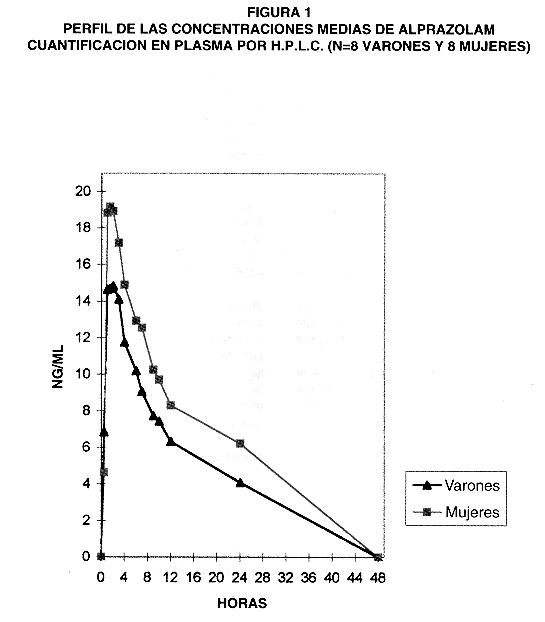
El fármaco se detectó inmediatamente en plasma, excepto por 1 sujeto que se pudo cuantificar hasta 1h. Como se muestra en el perfil (Figura 1), la concentración media más elevada se obtuvo a 1,5h y fue de 10,57±2,32 ng/nL (V =10,57±2,62 ng/mL ; M= 10,73± 2,14 ng/mL). A 24h se detectó el fármaco en 13 sujetos (81% ; V=7, M=6), y a +48h aún persistían en n= 5 de los sujetos (31% ; V=,3, M= 2).
Los valores individuales se describen en la Tabla 1. La Cmax global fue de 11,73±2,55 ng/mL, que se alcanzó a un Tmax entre 1h y 1,5h en el 68% de los sujetos (n= 11 ; V=5, M=6). .El descenso progresivo de los niveles sanguíneos se correspondió con un t1/2 global de 14,21±5,72 h. El ABC0-48 tuvo una magnitud global de 135,16±62,89 ng/mL· h.
No hubo diferencias estadísticamente significativas atribuibles al factor sexo en el perfil cinético global ni en las Cp a los diferentes tiempos. Tampoco fueron estadísticamente significativas las diferencias en los descriptores cinéticos al agruparlos por sexo.
Hubo una buena tolerabilidad sistémica al ALZ ; no se registraron cambios relevantes a nivel cardiovascular y hematológico, ni en las pruebas bioquímicas de control. ]]>
Agradecimientos
Por su colaboración durante la etapa experimental, a la Dra. MC Bayes Genís y al personal del área de Investigación Farmacológica, Instituto de Investigación- HSCSP, Barcelona. ]]>
2. Rosser SV, : Gender bias in clinical research and the diference it makes. Appl Clin Trials 1993 ;2 : 44-50. [ Links ]
3. Yonkers KA, Harrison W. : The inclusion of women in psychopharmacologic trials. J Clin Psychopharmacol 1993; 13 :380-382. [ Links ]
4. Yonkers KA, Kando J, Cole JO, Blumenthal S. : Gender differences in human pharmacokinetics and pharmacodynamics of psychotropic medication. Am J Psychiatry 1992 ; 149: 587-595. [ Links ]
5. Gallant D, Krinsky SL. : Ethical and legal considerations in drug trials. En : Prien RF, Robinson DS (Eds.):Clinical evaluation of psychotropic drugs : principles and guidelines. New York : Raven Press, 1994: 261 - 280. [ Links ]
6. Lentner C. (Ed.) : Body mass and height of adults. En : Geigy Scientific Tables, Somatometric data, vol 3. Basle : International Medical and Pharmaceutical Information Ciba-Geigy Limited, 1984: 324 - 329. [ Links ]
7. Shumaker RC. : PKcalc a basic interactive computer program for statistical and pharmacokinetics analysis of data. Drug Metab Rev 1986 ; 17 : 331-348. [ Links ]
8. Garfield FM. : Quality assurance principles for analytical laboratories. Virginia : AOAC Press,1984. [ Links ]
9. Directiva 88/320/CEE del Consejo de la Comunidad Económica Europea (CCEE) del 7 de junio de 1988 relativa a la Inspección y verificación de las buenas prácticas de laboratorio (BPL) : 13 pp. [ Links ]
10. Maldener G. : Requeriments and test for HPLC apparatus and methods in pharmaceutical quality control. Chromatographia 1989 ; 28: 85-88. [ Links ]
11. Greenblatt DJ, Divoll M, Abernethy DR, Ochs HR, Shader RI : Clinical pharmacokinetics of the newer benzodiazepines. Clin Pharmacokinet 1983; 8: 233 - 252. [ Links ]
12. Abernethy DR, Greenblatt DJ, Divoll M, Smith RB, Shader RI.: The influence of obesity on the pharmacokinetics of oral alprazolam and triazolam. Clin Pharmacokinet 1984: 9: 177 -183. [ Links ]
13. Bourin M , : Pharmacocinétique des benzodiazépines. En : Les benzodiacépines, de la pharmacocinétique á la dependance. 2éme édition. París : Elipses, 1989 : 31 - 98.
14. Ciarulo DA, Antal EJ, Smith RB, Olson DR, Goldberg DA, Rand EH, Raskin RB, Philips JP, Shader RI, Greenblatt DJ,.: The realtionship of alprazolam dose to steady-state plasma concentrations. J Clin Psychopharmacol 1990 ; 10 :27 - 32. [ Links ]
15. EllerMG, Della-Coletta AA.: Absence of effect of food on alprazolam absortion from sustained release tablets. Biopharmaceut Drug Disp 1990 ;11 :31 - 37. [ Links ]
16. Norman TR, Burrows GD, Mcintyre MI. : Pharmacokinetic and pharmcodynamic effects of a single nocturnal dose of alprazolam. Int Clin Psychopharmacol 1990 ; 5 :111 - 117. [ Links ]
17. Greenblatt DJ, Wright CE. : Clinical pharmacokinetics of alprazolam. Therapeutics implications. Clin Pharmacokinet 1993 ;24 :453 - 471. [ Links ]
18. Lader M. : Clincial Pharmacology of benzodiazepines. Ann Rev Medicine 1987 ; 38 :19 - 28. [ Links ]
19. Shader RI, Greenblatt DJ. : Use of benzodiazepines in anxiety disorders. N Eng J Med 1993 ;328: 1398 - 1405. [ Links ]
20. Flórez J, Hurlé MA. : Fármacos ansiolíticos y sedantes. En : Flórez J, Armijo JA, Mediavilla A (Eds.) : Farmacología Humana 2TM edición. Barcelona : Masson-Salvat Medicina,1991: 383 - 397. [ Links ]
21. Kirkwood C, Moore A, Hayes P, DeVane L, Pelonero A. : Influence of menstrual cycle and gender on alprazolam pharmacokinetics. Clin Pharmacol Ther 1991 ; 50 : 404 - 409. [ Links ]
* Depto. Farmacología y Toxicología Clínica, Escuela de Medicina, U.C.R; Depto. Farmacoterapia, CCSS. Médico becaria de la
Agencia Española de Cooperación Internacional en el Ärea de Investigación Farmacológica, Servicio de Farmacología Clínica. Hospital de la Santa Cruz y San Pable (HSCSP), Barcelona.
** Lab. De Cromatografía, Area de Investigación Farmacológica, Instituto de Investigación- HSCSP), Barcelona.
*** Area de Investigación Farmacológica, Instituto de Investigación-HSCSP, Barcelona. ]]>
| | | ||||||||
| Vol | ]]>
Cmax | | | | | | | | |
| 01 | | | | ]]> 13,14 | | | | | |
| 02 | | | | | ]]> 04 | | | | |
| 07 | | | | | | ]]> 13,52 | | | |
| 08 | | | | | | | ]]> 90 | | |
| 10 | | | | | | | | ]]> 186,70 | |
| 11 | | | | | | | | | ]]> 30,52 |
| 13 | | | | | | | | | |
| 16 | ]]> 12,18 | | | | | | | | |
| 0 | | ]]> | | | | | | | |
| DE | | | ]]> 28,09 | | | | | | |
 ]]>
]]>