The venom of Bothrops asper induces severe coagulation disturbances in accidentally envenomed humans. However, only few studies have been conducted to identify components that interact with the hemostatic system in this venom. In the present work, we fractionated B. asper venom in order to investigate the possible presence of inhibitors of platelet aggregation. Using a combination of gel filtration, anion-exchange chromatography, and reverse-phase high performance liquid chromatography, we isolated an acidic protein which shows a single chain composition, with a molecular mass of ~8 kDa, estimated by SDS-polyacrylamide gel electrophoresis. Its N-terminal sequence has high similarity to disintegrins isolated from different snake venoms, which are known to bind to cellular integrins such as the GPIIb/IIIa fibrinogen receptor on platelets. The purified protein exerted potent aggregation inhibitory activity on ADP-stimulated human platelets in vitro, with an estimated IC50 of 50 nM. This biological activity, together with the biochemical characteristics observed, demonstrate that the protein isolated from B. asper venom is a disintegrin, hereby named "bothrasperin". This is the first disintegrin isolated from Central American viperid snake species.
Keywords: Snake venom, disintegrin, Bothrops, platelet aggregation.
Snake venoms constitute rich sources of proteins that interact with the hemostatic system of vertebrates, either by promoting or by inhibiting particular steps of the coagulation cascade (Markland 1998, Kamiguti et al. 1998, Braud et al. 2000, Matsui et al . 2000). Such proteins may be of central relevance during the clinical course of accidental human envenomations by various snake species, particularly from the families Viperidae and Crotalidae, by inducing striking alterations in the coagulation system that might lead to fatal hemorrhages. In addition, the remarkable specificity of such venom proteins towards defined components of the hemostatic system, in many instances, has opened the possibility to exploit them as molecular tools for the study of coagulation reactions, for the development of in vitro diagnostic assays, and for potential therapeutic applications (Niewiarowski et al. 1994, Markland 1998, Braud et al. 2000). ]]>
In Central America, the majority of human envenomations due to snakebites are inflicted by Bothrops asper, commonly known as "ter-ciopelo" or "barba amarilla" (Gutiérrez 1995, 2002). As typical of many viperid/crotalid snakes, the venom of B. asper induces severe coagulation disturbances (Barrantes et al. 1985). Only few studies have been conducted to identify toxins affecting hemostasis in B. asper venom, despite their potential relevance for its lethal action. A thrombin-like enzyme has been described (Aragón and Gubensek 1978), and more recently, an inducer of platelet aggregation (aspercetin) from this venom was reported (Rucavado et al. 2001). In the present study, the possible presence of inhibitors of platelet aggregation in the venom of B. asper was investigated, resulting in the purification and characterization of a disintegrin, hereby named "bothrasperin".Materials and methods
Venom. The venom of Bothrops asper was a pool obtained from more than 30 specimens collected in the Atlantic region of Costa Rica, and kept at the serpentarium of the Instituto Clodomiro Picado, Universidad de Costa Rica. Immediately after extraction, the venom was centrifuged to remove insoluble debris, lyophilized, and stored at -20°C.
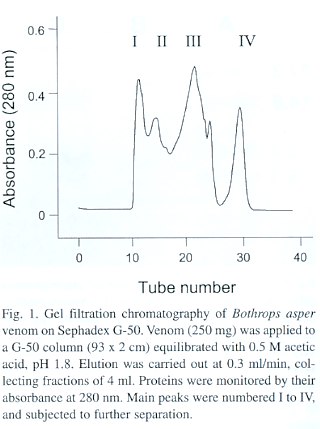
Gel filtration chromatography. Venom samples of 250 mg were dissolved in 0.5 M acetic acid, pH 1.8, and applied to a column of Sephadex G-50 (93 x 2 cm; Pharmacia, Sweden) equilibrated with the same solvent. Elution was carried out at 0.3 ml/min, monitoring proteins with an absorbance detector at 280 nm (Bio-Rad, USA). Fractions of 4 ml were collected, dried in a vacuum centrifuge (Savant SpeedVac System, AES 1010), and stored at -20°C.
Anion-exchange fast protein liquid chromatography (FPLC). Aliquots of 2 mg of fraction III from the gel filtration step were dissolved in 0.05 M ammonium acetate, pH 7.5, and applied to a Mono-Q anion-exchange column using an FPLC instrument (Pharmacia, Sweden). A linear gradient from 0.05 M to 0.24 M ammonium acetate, pH 7.5, was developed in 30 min. Fractions were collected manually, vacuum dried, and finally dissolved in 100 µl of 0.12 M NaCl, 0.04 M sodium phosphate, pH 7.2 (PBS) , in order to screen for platelet aggregation inhibitory activity.
High performance liquid chromatography (HPLC). Subfraction III-3 from the anion-exchange chromatography step was applied to a C18 reverse-phase column (15 x 4.6 mm; Vydac, USA) using an HPLC instrument (Waters model 600E, USA). Elution was carried out with a linear gradient from 0 to 60% acetonitrile in 0.1% trifluoroacetic acid, at 1 ml/min during 60 min.
Gel electrophoresis. A triphasic discontinuous polyacrylamide gel electrophoresis system in the presence of sodium dodecylsulphate (SDS-PAGE; Hames 1981) was utilized to monitor the protein composition of venom fractions during purification, as well as to estimate the molecular mass of the isolated disintegrin. Final monomer concentrations of the gel layers were 4% (stacking gel), 10% (upper resolving gel), and 15% (lower resolving gel). A set of very low molecular weight markers (VLMW; Pharmacia, Sweden) was run in parallel to samples, at a constant voltage of 150 V, and proteins were finally visualized by R-250 Coomassie blue staining.
Assay for inhibition of platelet aggregation. Fresh platelet-rich human plasma was prepared by centrifugation of blood from healthy volunteers, at 100 G during 15 min. Aliquots of 450 µl of this preparation were incubated with 10-20 µl of venom fractions for 5 min at 37°C. Then, platelet aggregation was initiated by the addition of 50 µl of 0.1 mM ADP, and monitored by the increase in light transmittance signal using a model 530-VS aggregometer (Chrono-Log Corporation, 530-VS) interfaced to a chart recorder, during 5-8 min. Platelet-poor plasma (450 µl) alone was utilized as a blank, whereas platelet-rich plasma (450 µl) incubated only with ADP (50 µl) was utilized as a positive control for aggregation. All samples were analyzed in duplicate assays. Protein concentration of the samples was estimated by the Bradford colorimetric microassay (Bradford 1976).
N-terminal sequence. The native purified protein was subjected to direct N-terminal sequencing by automated Edman degradation, using a model LF 3000 Beckman sequencer (Beckman, USA).
Results ]]>
The separation of B. asper venom components by gel filtration on Sephadex G-50 is shown in Fig. 1. All four major peaks obtained (I-IV) caused a direct coagulation of human plasma in the assay for platelet aggregation inhibitory activity, and therefore could not be screened for the presence of aggregation inhibitors. SDS-PAGE analysis revealed that peak I contained the high molecular weight venom components, whereas peak IV had very low amounts of protein, its absorbance being probably due to non-proteinaceous small compounds. Peaks II and III, on the basis of the molecular weight of its components, were selected for further fractionation by anion-exchange FPLC on Mono-Q.
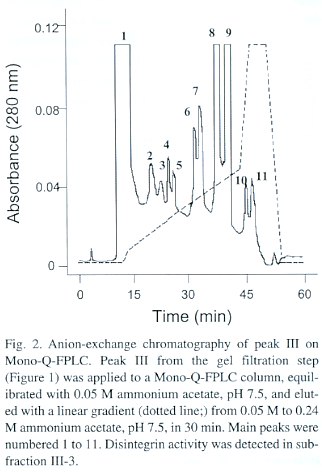
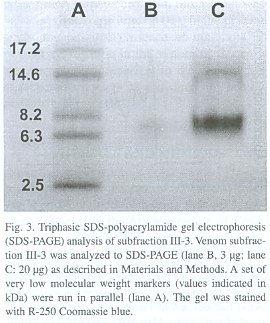
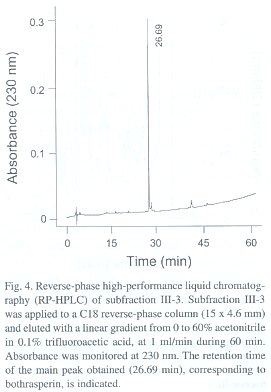
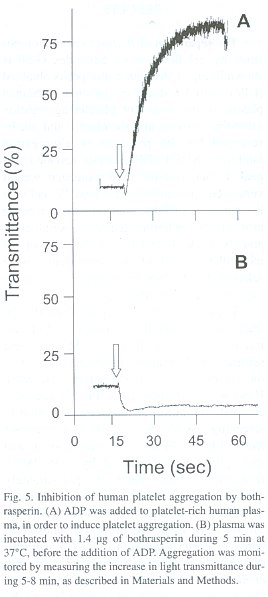
By testing a series of different concentrations of the purified disintegrin, an inhibitory concentration 50% (IC50 ) of 50 nM was calculated as its platelet aggregation inhibitory potency. The final protein preparation was subjected to N-terminal sequence analysis, in which the first 11 amino acid residues were identified as NH 2 -EAGEEXDXGTE (where X is a possible cysteine residue). This partial sequence is compared to that of other disintegrins in Table 1.

Discussion
Snake venoms may contain a variety of components with the ability to inhibit platelet aggregation, which, to date, correspond to one of the following types: disintegrins, -fibrinogenases, 5-nucleotidases, and phospholipases A2 (Markland 1998). This study demonstrates that the venom of B. asper, the most relevant venomous snake in Central America from the medical point of view, contains at least one component with potent anti-aggregating activity upon human platelets. This protein, here named bothrasperin, was isolated by a combination of gel filtration, ion-exchange, and RP-HPLC chromatographies. It was identified as a disintegrin on the basis of its molecular weight (8 kDa), acidic nature, N-terminal amino acid sequence, and biological activity. Its potency to inhibit human platelet aggregation in vitro (IC50 ) was estimated at approximately 50 nM.
Disintegrins from snake venoms have attracted interest in recent years, due to their clinical potential as platelet aggregation inhibitors in the thrombotic events that are frequently involved in cardiovascular and cerebrovascular disease (Markland 1998). Another relevant area of interest focuses on their anti-angiogenic and anti-metastasic activities in animal cancer models, due to their ability to interact with cellular integrins that participate in the mechanisms of neovascularization and tumor cell invasion (Danen et al. 1998, Yeh et al. 1998). On the other hand, the precise role of disintegrins in the toxic actions of snake venoms has not been established yet. Some venom hemorrhagic metalloproteinases contain a disintegrin-like domain, which might be released after partial proteolysis (Bjarnason and Fox 1994). Some of these disintegrin-like domains have been shown to interact with integrins (Kamiguti et al. 1996, Jia et al. 1997). It has been hypothesized that the disintegrin-like domain, by targeting particular cell surface integrins, may have a role in directing the large, multidomain hemorrhagic toxins towards specific locations, therefore enhancing their toxic efficiency (Matsui et al. 2000). In addition, if inhibition of platelet aggregation by disintegrins occurs also in viv o, hemorrhage induced by venom metalloproteinases might be potentiated, a possibility that awaits to be experimentally evaluated.
The disintegrins usually contain a recognition motif composed by the sequence Arg-Gly-Asp (RGD motif), which confers to them an affinity towards cellular integrins such as the GPIIb/IIIa fibrinogen receptor on platelets (Zhou et al. 1996, Markland 1998), explaining their anti-aggregating activity. In the case of bothrasperin, its N-terminal amino acid sequence showed a high similarity with jararacin, a disintegrin of 73 amino acids (7,739 Da) isolated from the venom of the South American species Bothrops jararaca (Scarborough et al. 1993). Further characterization of the structural and functional properties of bothrasperin will be of importance to determine its specificity and its role in the pathophysiological alterations occurring in snakebite envenomations by B. asper, as well as to explore potentially useful activities related to its ability to inhibit platelet aggregation.
Acknowledgements ]]>
The valuable collaboration of Timoteo Olamendi, Fernando Zamudio, and Lourival Possani (Instituto de Biotecnología, Universidad Nacional Autónoma de México at Cuernavaca, México) in the N-terminal sequence determination of bothrasperin is gratefully acknowledged. Financial support for these studies was obtained from Vicerrectoría de Investigación, Universidad de Costa Rica (VI-741-AO-085) and the Embassy of Japan in Costa Rica. This work was performed in partial fulfillment of the Licenciatura degree of A. Pinto at the Faculty of Microbiology, University of Costa Rica.Resumen
El veneno de la serpiente Bothrops asper induce graves alteraciones de la coagulación en los humanos accidentalmente envenenados. Sin embargo, se han realizado pocos estudios para identificar los componentes del veneno que interactúan con el sistema hemostático. En el presente trabajo, fraccionamos el veneno de B. asper para investigar la posible presencia de inhibidores de la agregación plaquetaria. Empleando una combinación de técnicas cromatográficas (filtración en gel, intercambio aniónico y cromatografía líquida de alto desempeño en fase reversa), aislamos una proteína acídica de cadena simple, con una masa molecular de ~8 kDa, estimada mediante electroforesis en gel de poliacrilamida con SDS. Su secuencia de aminoácidos N-terminal muestra una alta similitud con la de disintegrinas aisladas de diferentes venenos de serpiente, las cuales se unen a integrinas celulares como el receptor de fibrinógeno GPIIb/IIIa de las plaquetas. La proteína purificada ejerce una potente acción inhibitoria sobre la agregación in vitro de plaquetas humanas estimuladas con ADP, con una IC 50 estimada en 50 nM. Esta actividad biológica, sumada a las características bioquímicas observadas, demuestran que la proteína aislada del veneno de B. asper es una disintegrina, a la cual denominamos "both-rasperina". Esta es la primera disintegrina aislada de una especie de vipéridos de Centroamérica.
References
Aragón, F. & F. Gubens¢ek. 1978. Characterization of thrombin-like proteinase from Bothrops asper venom. pp. 107-111. In P. Rosenberg (ed.). Toxins: Animal, Plant and Microbial. Pergamon, Oxford.
Barrantes, A., V. Solís & R. Bolaños. 1985. Alteración de los mecanismos de la coagulación en el envenenamiento por Bothrops asper (terciopelo). Toxicon 23: 399-408. [ Links ]
Bjarnason, J.B. & J.W. Fox. 1994. Hemorrhagic metallo-proteinases from snake venoms. Pharmac. Ther. 62: 325-372. [ Links ]
Bradford, M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248-254.
Braud, S., C. Bon & A. Wisner. 2000. Snake venom proteins acting on hemostasis. Biochimie 82: 851-859. [ Links ]
Danen, E.H., C. Marcinkiewicz, I.M. Cornelissen, A.A. van Kraats, J.A. Pachter, D.J. Ruiter, S. ]]>
Niewiarowski & G.N. van Muijen. 1998. The disintegrin eritostatin interferes with integrin a4b1 function and with experimental metastasis of human melanoma cells. Exp. Cell. Res. 238: 188-196.Gutiérrez, J.M. 1995. Clinical toxicology of snake bites in Central America, p. 646663. In J. Meier & J. White (eds.). Handbook of Clinical Toxicology of Animal Venomous and Poisons. CRC, Boca Raton.
Gutiérrez, J.M. 2002. Comprendiendo los venenos de serpientes: 50 años de investigaciones en America Latina. Rev. Biol. Trop. 50: 377-394.
Gutiérrez, J.M., C. Avila, E. Rojas & L. Cerdas. 1988. An alternative in vitro method for testing the potency of the polyvalent antivenom produced in Costa Rica. Toxicon 26: 411-413. [ Links ]
Hames, B.D. 1981. Peptide mapping by limited proteolysis using SDS-polyacrylamide gel electrophoresis, pp. 219-229. In B.D. Hames & D. Rickwood (eds.). Gel Electrophoresis of Proteins, a Practical Approach. IRL Press, Oxford.
Jia, L.G., Wang X.M., J.D. Shannon, J.B. Bjarnason & J.W. Fox. 1997. Function of disintegrin-like/cys-teine-rich domain of atrolysin A: inhibition of platelet aggregation by recombinant protein and peptide antagonists. J. Biol. Chem. 272: 13094-13102.
Kamiguti, A.S., C.R.M. Hay, R.D.G. Theakston & M. Zuzel. 1996. Insights into the mechanism of hemorrhage caused by snake venom metalloproteinases. Toxicon 34: 627-642.
Kamiguti, A.S., M. Zuzel & R.D.G. Theakston. 1998. Snake venom metalloproteinases and disintegrins: interactions with cells. Braz. J. Med. Biol. Res. 31:853-862. [ Links ]
Markland, F.S. 1998. Snake venoms and the hemostatic system. Toxicon 36: 1749-1800. [ Links ]
Matsui, T., Y. Fujimura & K. Titani. 2000. Snake venom proteases affecting hemostasis and thrombosis. Biochim. Biophys. Acta 1477: 146-156. [ Links ]
Niewiarowski, S., M.A. McLane, M. Kloczewiak & G.J. Stewart. 1994. Disintegrins and other naturally occurring antagonists of platelet fibrinogen receptors. Seminars in Haematology. 31: 289-300.
Rucavado, A., M. Soto, A.S. Kamiguti, R.D.G. Theakston, J.W. Fox, T. Escalante & J.M. Gutiérrez. 2001. Characterization of aspercetin, a platelet aggregating component from the venom of the snake Bothrops asper which induces thrombocytopenia and potentiates metalloproteinase-induced hemorrhage. Thromb. Haemost. 85: 710-715.
Scarborough, R.M., J.W. Rose, M.A. Naughton, D.R. Phillips, L. Nannizzi, A. Arfsten, A.M. Campbell & I.F. Charo. 1993. Characterization of the integrin specificities of disintegrins isolated from American pit viper venoms. J. Biol. Chem. 268: 1058-1065.
Yeh, C.H., H.C. Peng & T.F. Huang. 1998 Accutin, a new disintegrin, inhibits angiogenesis in vitro and in vivo by acting as integrin aVb3 antagonist and inducing apoptosis. Blood 92: 3 268-3 276. [ Links ]
Zhou, Q., C. Dangelmaier & J.B. Smith. 1996. The hemorrhagin catrocollastatin inhibits collagen-induced platelet aggregation by binding to collagen via its disintegrin-like domain. Biochem. Biophys. Res. Commun. 219: 720-726. [ Links ]
1 Instituto Clodomiro Picado, Facultad de Microbiología,
2 Departamento de Bioquímica, Escuela de Medicina, Universidad de Costa Rica, San José, Costa Rica;
3 Laboratorio de Investigación, Hospital Nacional de Niños, Caja Costarricense de Seguro Social, San José, Costa Rica.
4 Correspondence to: Bruno Lomonte, PhD, Instituto Clodomiro Picado, Facultad de Microbiología, Universidad de Costa Rica, San José, Costa Rica, fax (+506) 292-0485, e-mail: blomonte@cariari.ucr.ac.cr ]]>