Introduction
Tricalcium silicate-based materials such as mineral trioxide aggregate (MTA) have biological, physicochemical, and mechanical properties that are well suited to clinical applications. These types of cement are radio-opaque and form a calcium silicate gel when mixed with water. The first formulation of this type of material was patented in 1995 and was the grey MTA, which consisted of Portland cement and bismuth oxide (1,2).
MTA can modulate tissue repair and hard tissue deposition due to its ability to promote the precipitation of carbonate apatite through the release of calcium ions and their interaction with phosphate in the dentin matrix or tissue (3). However, it is difficult to handle and can be challenging to insert and condense clinically (4,5).
An endodontic cement based on MTA has useful properties such as: the sealing capacity (6,7) is related to the possibility of expansion (8); can set in the presence of fluids; and can release hydroxyl and calcium ions. Additionally, such cement can form a mineralized barrier in the adjacent tissue (9,10). As they are clinically efficacious, new tricalcium silicate materials are increasingly being introduced to the market, one of which is MTA Flow®(Ultradent Products Inc, South Jordan, UT) .
MTA Flow® is a new MTA-based cement composed of a di- and tri-calcium silicate gray powder that is set with a water-based gel and is intended for use in canal sealing, pulpotomies, pulp capping, apexification, root resorption, and other endodontic procedures. According to the manufacturer, it can be manipulated in various powder/gel ratios, resulting in a smooth consistency that is easy to use, and adaptable (11).
It is known that the marginal adaptation and bond strength of a material to dentin is a fundamental factor in the success of endodontic procedures and is related to the biomineralization capacity of the material. Therefore, the constant evaluation of different dental materials is essential. This research aimed to evaluate the biomineralization process and bond strength of MTA Flow® in its three different consistencies: Putty, Thick, and Thin, using the push-out test in an ex vivo model.
Materials and methods
Using an SBT diamond blade (Bay Technology South, San Clemente, CA, USA) on a high precision cutting equipment (Buehler Ltda., USA), 2mm thick discs were made from middle third radicular dentin of 61 human teeth. Two dentin discs were obtained from each tooth, and the canal was enlarged with Gates Glidden #6 rotary instruments to obtain a standardized cavity of 1.5mm in diameter. Subsequently, the root sections were immersed in a 17% ethylenediaminetetraacetic acid (EDTA) solution for 3 min, followed by 1 min in distilled water. Then, they were dried.
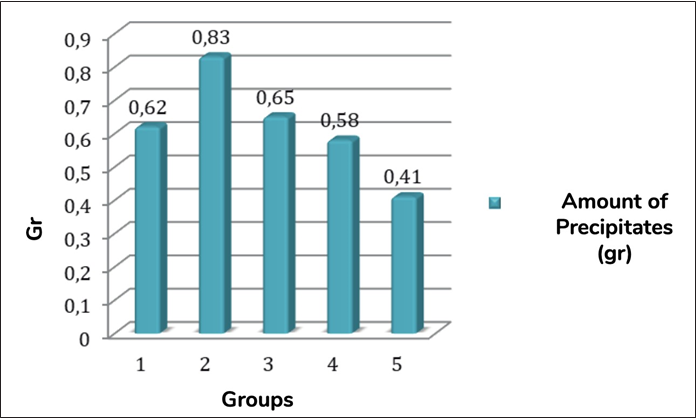
The dentin discs were randomly divided into 5 groups (n=30) and filled with different cement, as described below: G 1: MTA Angelus (Angelus Dental, Paraná, Brazil), G 2: Biodentine (Septodont, Paris, France), G 3: MTA Flow® Putty, G 4: MTA Flow® Thick and G 5: MTA Flow® Thin. Subsequently, the samples were divided into 3 subgroups (SG): SG1 (n=5): analysis of the biomineralization process; SG2 (n=20): evaluation of the push-out strength; and SG3 (n=5): evaluation of the cement/dentin interface on SEM.
Analysis if the biomineralization process (SG1)
Immediately after being filled with their respective cement, 5 samples were placed individually in plastic containers, with 30mL of phosphate- buffered saline (PBS; pH=7.2) at 37°C for 15 days. The buffer solution was replaced after 1, 3, 5,10, and 15 days, and measured using a digital pH meter (pH Pen, PH-50 EXTECH, USA). The readings were taken 2 min after the electrode was immersed in the solution. Between each reading, the electrode was washed with deionized water and dried with absorbent paper. Afterward, the solution was processed to determine the calcium ion release at the Laboratory of Analytical Services of the School of Chemistry in the University of Costa Rica. The sample was measured with a Varian Atomic Absorption Spectrophotometer (SpectrA A220 Fast Sequential, USA). Five unsealed samples were immersed in 30mL of PBS for the same period of time as a negative control. The values obtained were recorded, grouped, and descriptively analyzed.
At the same time as the solution was changed, the surface of the samples was gently scraped with a spatula. The collected precipitates were dried in an incubator at 37°C and weighed on a digital analytical balance (Mettler PE, 160, USA). The weights of the precipitates were recorded and statistically evaluated using t-tests with the GraphPad Prism statistical program. A P-value <0.05 was considered statistically significant.
Subsequently, 2 samples of precipitates were dried at 37°C for 48 hours and placed in a cylindrical metal (stubs), which was covered with a 300 Å gold-palladium layer. The specimens were examined with a scanning electron microscope (SEM; HITACHI, S-570, Japan), operating at 15 Kv.
The chemical composition of the precipitates was determined using an SEM micro-energy dispersion analysis (EDAX) (HITACHI, S-570). To analyze the ultrastructural aspect, the precipitates were photographed at variable magnifications (500-2500X). The data were recorded and analyzed descriptively.
Evaluation of the bonding force and push- out resistance (SG2)
Twenty samples were randomly divided into two groups (n=10). In group A (negative control), a wet cotton ball was placed on the cement and stored for 72 hours at 37°C. The samples from group B were individually suspended in sterile plastic vials containing 30 mL of PBS (pH=7.2) for 15 days at 37°C. After the experimental periods, the push-out test was performed using a universal testing machine that applied a constant compressive force at 0.5 mm/min. The push-out force value in MPa was calculated from the force kgf (maximum force applied to the cement before the displacement was recorded) converted to Newton (N) divided by the area in 2mm. A Student’s t-test was performed to evaluate the same type of cement, with and without PBS immersion.
Results
In all groups, it was possible to observe the formation of white precipitates after the first 20 min post-immersion in PBS. In general, the samples filled with Biodentine had a higher precipitation. However, this was not statistically different from the other groups (G1, G3, and G4 (p= 0.0536)), except when compared with G5 (P < 0.05) (Figure 1).
Regardless of the group, the peak pH was recorded on the fifth day (11.3 to 10.3). On day 15, there was a slight drop in all samples (from 10.9 to 10). G2 had the highest pH at all intervals and was the group that reached alkalization the fastest. However, MTA Flow®, regardless of the consistency used, showed a similar behavior throughout the observation period. The pH of the control group remained at 7.2 throughout the experimental period (Figure 2). The G2 samples showed the highest amount of calcium ion release. G1 showed the highest amount of calcium ion on day 1, while for the remaining groups, the ion release peak occurred on day 3. From day 5, the release decreased in all groups (Table 1).
The results obtained from the push-out resistance test are summarized in Table 2. The push-out resistance 72 hours after filling was significantly different (p=0.0090) between groups G2 and G5. However, after immersion in PBS for 15 days, there was a significant difference between all groups (G1,G2,G3,G4) in relation to G5 (p=0.0147). All samples immersed in PBS for 15 days showed significantly greater resistance to displacement than that observed for samples in contact with a wet cotton ball for 72 hours (p= 0.0097).
The ultra-structural evaluation in SEM revealed the presence of precipitates with different morphologies (Figure 3, Figure 4 and Figure 5). In general, all cement promoted the formation of precipitates, and no ultra-structural or chemical composition differences between them occurred. On average, the presence of calcium and phosphorus in a molar Ca/P concentration of 1.43 to 1.82 was indicated from three punctual evaluations of each sample (Figure 4 and Figure 5). Depending on the area analyzed, different morphologies were found. It was possible to observe a greater quantity of precipitates with acicular prolongations, typical of amorphous calcium phosphate (Figure 3 and Figure 4). More compact and spherical precipitates were also observed (Figure 3 and Figure 5). The images of the precipitates denote the presence of areas with higher calcification (arrows).
Regarding the evaluation of the cement/ dentin interface using observational análisis in SEM, the formation of the intermediate layer of carbonate apatite was partially detected in all groups (Figure 6, Figure 7, Figure 8). In general, zones of biomineralization were observed at the interface, but these were not continuous. The G2 group showed the best marginal adaptation to dentin and the greatest formation in the biomineralization process (Figure 6) even though the apatite interface was not continuous along the entire circumference. Regarding MTA Flow® Thin (G5), regions of detachment gaps and peripheral sealing defects were apparent (Figure 8 and Figure 9).
However, fluorescence microscopy showed that, in general, all cement had adequate marginal adaptation. Samples filled with Biodentine displayed dentin infiltration (Figure 9). MTA Angelus also demonstrated continuous marginal adaptation along the surface. MTA Flow® Putty and Thick displayed adequate marginal adaptation. However, the samples filled with MTA Flow® Thin, once the material was hardened and immersed in PBS for 15 days, showed the presence of peripheral gaps and loss of mass (Figure 9).
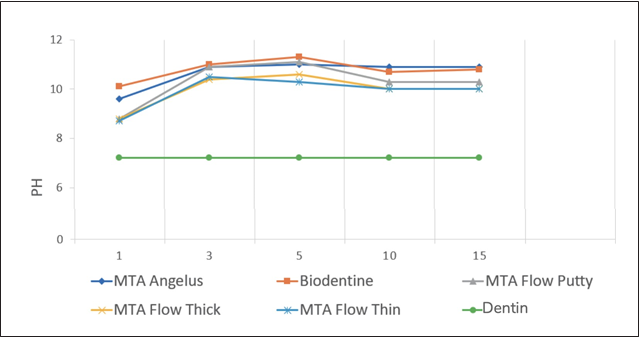
Figure 2 Average pH values of the solutions in contact with the cement at different experimental periods.
Table 1 Total values of calcium ion release.
| Experimental Groups | Total (mg/L) |
|---|---|
| Group 1 | 21.02 |
| Group 2 | 36.11 |
| Group 3 | 23.22 |
| Group 4 | 23.85 |
| Group 5 | 15.98 |
| Dentin Disc | 1.5 |
Table 2 Mean bonding force (MPa) and standard deviation (SD) values of all groups.
| Groups | G1 | G2 | G3 | G4 | G5 |
|---|---|---|---|---|---|
| After 72 h | 3.84 (1.32) | 4.23 (0.92) | 3.94 (1.12) | 3.32 (1.10) | 2.58 (0.86) |
| After PBS Inmersion 15 dias | 6.60 (0.63) | 7.18 (0.42) | 6.22 (0.40) | 5.97 (0.25) | 4.12 (0.37) |

Figure 3 A. Areas with more compact precipitates and mineralization (arrows). B. Spherical precipitates with acicular extensions.
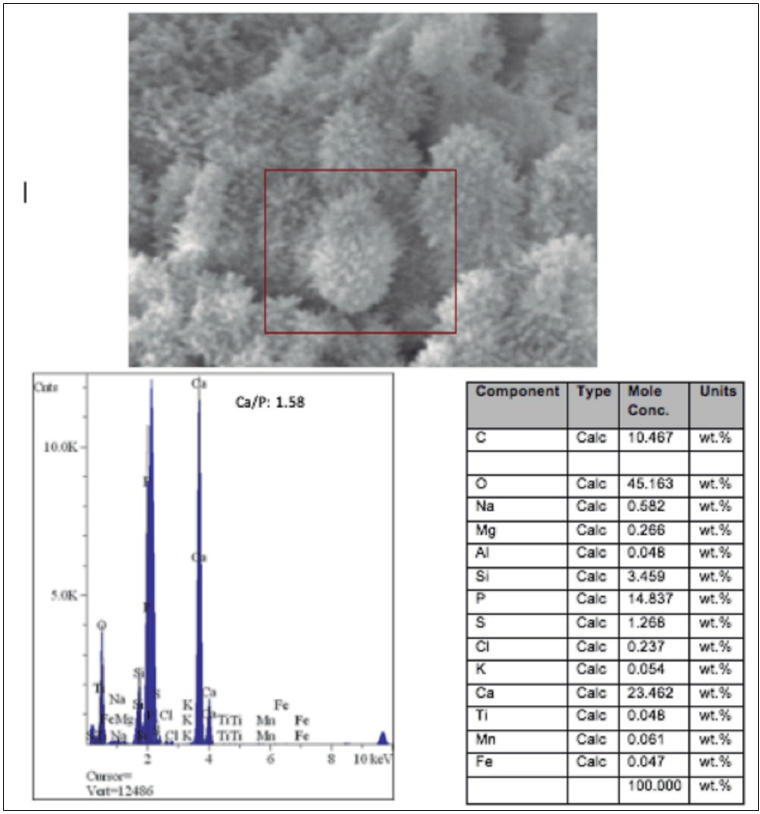
Figure 4 A. Scanning electron microscope (SEM) photomicrograph of the acicular precipitates analyzed by micro-energy dispersion analysis (EDAX; HITACHI, S-570). B. EDAX spectrum showing the presence of calcium and phosphorus at a Ca/P ratio of 1.58. Table with the molecular concentrations of the elements provided by EDAX-SEM.
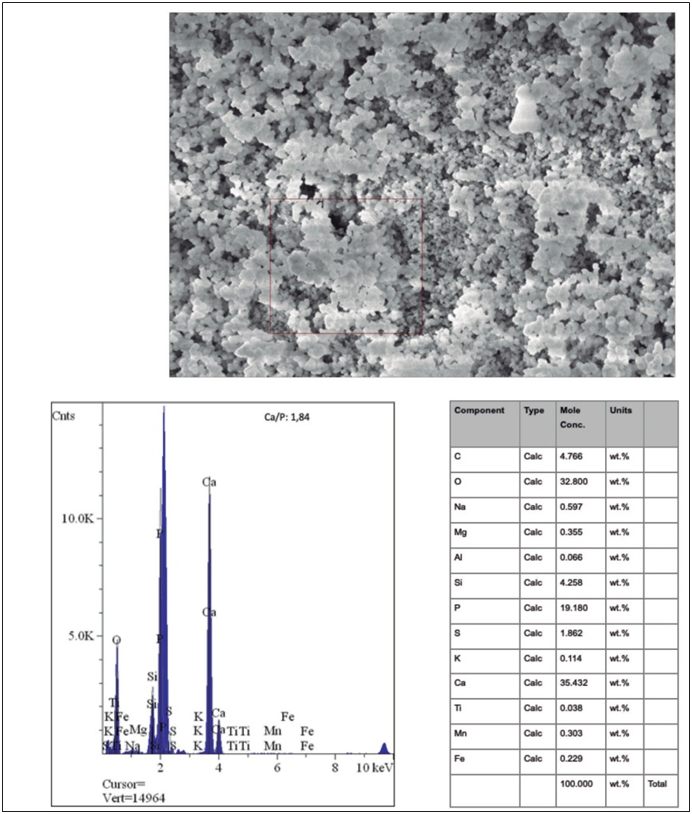
Figure 5 A. Scanning electron microscope (SEM) photomicrograph of the compact precipitates analyzed by micro- energy dispersion analysis (EDAX; HITACHI, S-570). B. EDAX spectrum showing the presence of calcium and phosphorus in a Ca/P ratio of 1.84. Table with the molecular concentration of the elements provided by EDAX-SEM.
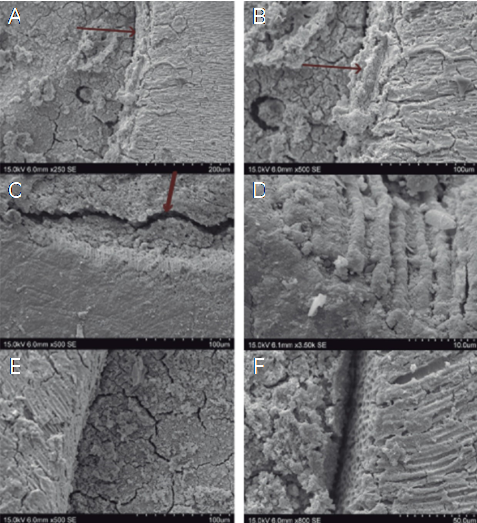
Figure 6 Representative micrographs of the MTA Angelus group. A, B. The formation of the apatite interface is observed (arrow). C, D. SEM Artifact - separation of cement can be observed (arrow), however the apatite interlayer remains attached to the dentin and tubular mineralization is demonstrated. E, F. It is shown that the formation of the apatite interlayer was not found in all regions circumferentially.

Figure 7 Representative microphotographs of G2 - Biodentine. A, B, C. The formation of the apatite interface can be observed (arrow). D. Apatite interface formation was not found in all circumferential regions.
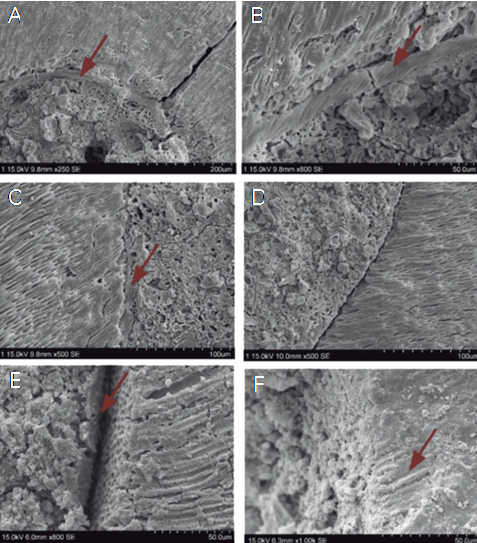
Figure 8 Representative photomicrographs of the samples filled with MTA Flow®. A,B. MTA Flow® Putty samples showed formation of the apatite interface more homogeneously. C,D. MTA Flow® Thick samples demonstrated formation of a discontinuous apatite interface (arrow). However, the material maintains an adequate marginal adaptation. E,F. MTA Flow® Thin showed fewer, discontinuous and disordered biomineralization nuclei in different regions.
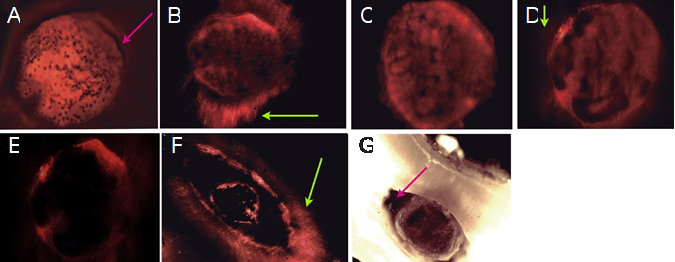
Figure 9 Representative fluorescence microphotographs of the different groups. A. MTA ANGELUS showed presence of few areas with marginal gaps (Arrow). Most of the periphery has good marginal adaptation. B. The dentin infiltration of the material, tag-like formation (green arrow) and marginal adaptation around all Biodentine/dentin interface can be observed. C. MTA Flow® Putty showed good marginal adaptation to dentin. D. MTA Flow® Thick demonstrated some intratubular infiltration (green arrow). E and F MTA Flow® Thin showed open areas and discontinuous marginal adaptation. Also denoted areas within the material with big voids demonstrating loss of mass during PBS immersion. However is possible to observed intratubular infiltration (green arrow). G. MTA Flow® Thin Photomicrograph showed loss of mass and/or contraction of the cement (arrow). After 15 days of immersion in PBS, the cement do not demonstrated mass stability nor marginal adaptation.
Discussion
MTA Flow® was developed to easily acquire a suitable consistency for a variety of clinical applications, such as pulp capping, pulpotomies, perforation, resorption sealing, and apexification (11). Our study was designed to evaluate the biomineralization process and displacement resistance of MTA Flow® at different consistencies when compared to standard gold tricalcium silicate formulations (MTA Angelus and Biodentine).
This study has shown that MTA Angelus, Biodentine, and MTA Flow® promote the formation of the same type of precipitate. After the first 20 min in PBS, the formation of white precipitates was observed; this increased proportionally with time. Scientific research has shown that these deposits provide clinical advantages by creating a suitable environment for the differentiation of stem cells and osteoblasts that promote the formation of new bone (12).
The largest amount of precipitates was in the Biodentine group. However, this was not significantly different from MTA Angelus, MTA Flow® Putty, and MTA Flow® Thick. MTA Flow® Thin produced the least amount of precipitates.
Analysis of the calcium ion release from the precipitates obtained in each group determined that Biodentine released the most calcium ions, while MTA Flow® Thin released the least.
SEM showed the formation of a greater amount of acicular precipitates with a molar ratio Ca/P of 1.58, typical of amorphous calcium phosphate, and a smaller amount of compact precipitates with a Ca/P ratio of 1.84. As demonstrated, acicular precipitates are characteristic of pH-dependent autocatalytic transformation from the metastable phase of amorphous calcium phosphate to the apatite phase (13). Acicular precipitates are a consequence of the surface activity of apatites. They incorporate elements into their structure and substitute certain elements which results in morphological alterations (14).
The pH of the solutions in contact with the cement showed the highest peak on the fifth day (from 11.3 to 10.3). On the tenth day, there was a slight drop in all the samples (10.9 to 10). Biodentine showed the highest pH at each interval and reached alkalinization faster. MTA Flow® showed similar behavior during the experimental period, regardless of the consistency used (Figure 2). These findings are consistent with the theory of biomineralization, which describes that at a physiological pH, the formation of carbonate apatite occurs in a two-phase process. The first occurs in the initial 5 days, where the Ca/P ratio is lower (1.58) and is associated with an increase in pH (from 11.3 to 10.3), favoring the formation of amorphous calcium phosphate. During the second phase, an increase in the Ca/P ratio (1.84) is observed, and a slight drop in pH (from 10.9 to 10). In this study, the decrease occurred from day 10 in all cases, which can be attributed to the incorporation of hydroxyl ions (OH-) released by the cement, to the OH- sites of the apatite, promoting the conversion of carbonate apatite (15). Therefore, it is clear that from the day of exposure, the process of biomineralization begins.
Biodentine samples had a higher precipitate formation and higher resistance to dislodgement in the push-out test. However, this was not significantly different from the other groups except for MTA Flow® Thin, which had significantly lower bond strength and resistance to dislodgement. This may be related to several issues, but considering the biomineralization process can be attributed to the poor precipitate formation and, therefore, lower mineralization. Likewise, this finding is related to the analysis of the MTA Flow® Thin microphotographs, in which marginal mismatches and gaps were found. These could have occurred because this cement has a more watery consistency and less powder content.
All the samples immersed in PBS showed significantly higher resistance to displacement than the samples that were in contact with a wet cotton ball for 72 hours. When interacting with PBS, the five types of cement underwent a biomineralization process, which initially leads to greater retention of the cement with the dentin, through a micromechanical bonding system. These findings are in agreement with Reyes-Carmona et al., who demonstrated that PBS solution in contact with MTA promotes biomineralization and improves the resistance to displacement (13,15).
In all the groups, it was possible to observe the formation of an intermediate layer of partially carbonated apatite. Such a layer is to be expected because, from day 10, the pH began to drop, thus meaning that the remineralization process initiated. Although complete remineralization of dentin was not observed, in areas with apatite interface, MTA Angelus appeared to be in close contact with the dentin. Biodentine showed a thicker apatite interface. However, compared to MTA Angelus, it did not have the same level of intratubular remineralization.
MTA Flow® Putty and Thick presented intratubular mineralization, plus good adaptation to dentin, similar to MTA Angelus. MTA Flow® Thin has a more diluted consistency and shows good intratubular infiltration. However, due to its consistency, there is a possibility of hydrolysis.
Clinically it is important for a cement to display acceptable push-out strength to dentin values, which can be achieved with adequate marginal adaptation, chemistry bond, infiltration and intratubular remineralization. Moreover, Martini et al. asserted that the sealing capacity of a material depends on the expansion it generates (16). This may be why Biodentine obtained excellent results in the push-out test. However, from a biological and clinical point of view, in addition to good sealing, efficient remineralization is required.
MTA Flow® consistencies showed thinner interfaces when compared to Biodentine and similar to MTA Angelus. The areas that showed a greater marginal adaptation under fluorescence microscopy are those with a higher percentage of mineralization. Biodentine has a more fluid consistency than MTA Angelus. Therefore, it tends to generate greater dentin infiltration. The MTA Flow® Putty, possibly because of its consistency, did not achieve intratubular infiltration to the same level as Biodentine. MTA Flow® Thick achieves a higher intratubular infiltration compared to MTA Flow® Putty.
The lower bioactivity of MTA Flow® Thin may be due to its consistency. As it presents a higher aqueous phase and a lower amount of material, it will likely hydrolyze. Therefore, although it achieved an excellent intratubular infiltration, it did not manage to biomineralize as much as the other samples. This is in contrast to the study by Martini et al., which indicated that MTA Flow® has low solubility, similar to MTA Angelus. This finding was probably because, in this study, MTA Flow® was only evaluated using the putty consistency (16).
Despite the dentin infiltration capacity of MTA Flow® Thin, it presents the lowest values regarding marginal adjustment, push-out and remineralizes less than the other cements evaluated. Clinically, it can be assumed that it does not provide the necessary conditions for optimal restoration, despite providing dentin infiltration. Dentin infiltration should not be the only feature to consider when selecting a bioactive material. The ability to remineralize both at the dentin interface and intratubularly, plus acceptable physical, chemical and bonding characteristics must be considered.
MTA Flow® represents a new bioactive calcium silicate material. It appears to be an alternative to conventional MTA cement due to its alkalization capacity and ability to form calcium phosphate deposits, similar to MTA Angelus and Biodentine. However, MTA Flow® Thin is not recommended in the treatment of apical plugs and other procedures, as indicated by the manufacturer, because the loss of volume of an endodontic cement is undesirable as this reduces its sealing capacity and therefore creates optimal conditions for bacterial and fluid microfiltration (16).
In conclusion, MTA Flow®, in Putty or Thick consistencies, presents a biomineralization process and push-out strength similar to MTA Angelus and Biodentine, however, this decreases considerably in the Thin consistency.