Introduction
Herbivorous insects become pests of cultivated systems (Mazzi & Dorn, 2012), because of the increasing nutritional value of plants via the addition of nitrogen and homogenization of plant communities in the agricultural system (Matson, Parton, Power, & Swift, 1996; Altieri, 1999; Rusch, Bommarco, & Ekbom, 2017). Aphids (Hemiptera: Aphididae) are among the leading insect pests worldwide due to the physiological stress their feeding generates in their host plants and their ability to transmit phytopathogenic viruses (Goggin, 2007; Brault, Uzest, Monsion, Jacquot, & Blanc, 2010).
The identification of aphid species is complex due to their evolutionary tendency of losing taxonomically useful morphological characters and their environmental plasticity (Foottit, 1997; Foottit, Maw, Von Dohlen, & Hebert, 2008). Classical morphological and taxonomical identification involves micrometric measurements of certain characteristics at specific developmental stages, which are usually during the adult stage of the female (Holman, 1974), thus requiring the rearing of insects (Blackman & Eastop, 2007). The presence of different morphological variants in the same species increases the difficulty of identifying these insects and subsequently identifying and characterizing species-specific relationships at different levels, such as herbivore-host, parasitoid-host and virus-vector relationships (Foottit et al., 2008; Miller & Foottit, 2009; Nie, Pelletier, Mason, Dilworth, & Giguère, 2011; Pelletier et al., 2012).
A useful complement in the identification of species of the family Aphididae has been the use of the mitochondrial region called the “DNA barcode”, which corresponds to a fragment of approximately 700 base pairs at the 5 ‘end of the cytochrome c oxidase I (COI) gene (Foottit et al., 2008; Miller & Foottit, 2009; Chen, Jiang, & Qiao, 2012). This technique proposed by Hebert, Cywinska and Ball (2003), has allowed for the rapid identification of aphid species by considering genetic distance thresholds specific to this group and comparing the sequences with those available in public reference databases (Coeur d’acier et al., 2014). DNA barcoding has also contributed to the discrimination of cryptic aphid species (Piffaretti et al., 2012), the association of morphological variants with different developmental stages in the same species (Miller & Foottit, 2009) and the establishment of different morphotypes that a species can present in different host plants (Lokeshwari, Kumar, & Manjunatha, 2014).
Another advantage of molecular techniques is the identification of species at any developmental stage of the pest (Armstrong & Ball, 2005). This type of reliable and early detection is of great phytosanitary value because it allows for a timely and adequate response to a potential impact (Armstrong & Ball, 2005; Mazzi & Dorn, 2012). At the same time, proper identification of the species is also essential for establishing integrated pest management (IPM) and clarifying different ecological and trophic interactions that may affect the biological control of pests (Foottit, Maw, Havill, Ahern, & Montgomery, 2009; Pelletier et al., 2012).
Among the different ecological interactions that aphids establish with other organisms, the mutualistic relationship with ants (Hymenoptera: Formicidae) is noteworthy. Although this interaction is facultative and not species-specific (Buckley, 1987; Collins & Leather, 2002), it contributes to the population growth of aphids, which provide sugar secretions to ants while the ants offer protection from generalist and specialist aphidophagous predators (Styrsky & Eubanks, 2007; Powell & Silverman, 2010a). Negative aspects of this interaction may include reduced size and reproductive fitness of aphids and its predation by ants (Billick, Hammer, Reithel, & Abbot, 2007; Yao, 2014; Hosseini, Hosseini, Katayama, & Mehrparvar, 2017). This mutualist association can interfere with integrated pest management and this interference is why its description in agricultural systems is relevant.
Aphids are the main pest of the pepper Capsicum spp. in Colombia because they can transmit phytopathogenic viruses of the genera Potyvirus and Cucumovirus (Van Emden & Harrington, 2003), which affect these crops (Pardey & García, 2011), and depending on the level of incidence of the disease, and the age of the crop plants at the time of the infection, losses in the crop may reach 100 % (Kenyon, Kumar, Tsai, & Hughes, 2014). However, the morphological identification of these aphids is complex, aphid species identity and diversity are unknown, therefore, the ecological interactions with other insects in this agricultural system are not well identified. For this reason, this study sought to identify both the aphid species present in the pepper crops, cayenne pepper Capsicum annuum L. and tabasco pepper Capsicum frutescens L. (Solanaceae), in Southwestern Colombia, as well as mutualistic ant species associated with the care of aphid colonies and to evaluate the effect of ant care on the size of the aphid colonies. Finally, the present study aimed to explore and illustrate the plant-aphid-ant interaction networks in Capsicum spp. commercial farming systems.
Materials and methods
Obtaining insects: Commercial crops of Capsicum spp. were visited in seven locations in Valle del Cauca, Southwestern Colombia, Bolivar (04°17’60.0” N & 76°12’28.8” W), Roldanillo (4°24’14.4” N & 76°08’42.0” W; 04°28’30.0” N & 76°07’04.8” W), Toro (04°36’21.6” N & 76°04’58.8° W; 04°36’21.6” N & 76°05’24.0” W), Dagua (03°45’10.8° N & 76°39’21.6” W), Guacari (03°45’00.0” N & 76°19’30.0” W), Rozo (03°37’15.2” N & 76°25’14.5” W; 03°37’10.6” N & 76°23’22.9” W), Vijes (03°42’18.0” N & 76°25’48.0” W); and they included four cayenne pepper (C. annuum) and six tabasco pepper (C. frutescens) sampling sites (Fig. 1). Each location was visited at two phenological stages between January (before flowering) and August (flowering and fruiting stage) of 2017. The area analysed corresponds to the inter-Andean valley of the Cauca river, located between the Western and central mountain ranges. In this regional scale, the surface covered is an inter-Andean tectonic depression between 900 m to 1 000 m of elevation, it presents 23.3 ± 6 ° C and 79 ± 24 % RH (Instituto Geográfico Agustín Codazzi, 1998; Cueto, 2006; Montoya-Colonia, 2010).
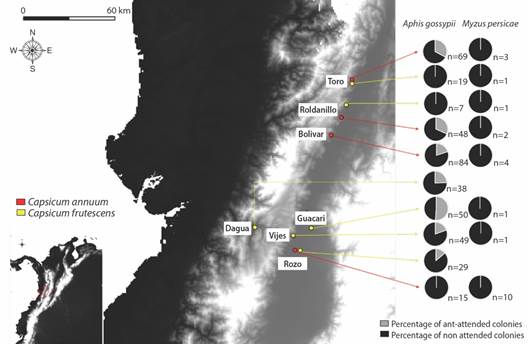
Fig. 1 Geographical location of the sampled sites. Yellow indicates the localities with tabasco pepper (C. frutescens) crops and red indicates the localities with cayenne pepper (C. annuum) crops.
Because aphids enter the fields of tabasco pepper crops in Southwestern Colombia from the edges and present an aggregate distribution pattern (Tálaga et al., 2017), a manual sampling of aphids was performed, covering the perimeter of the cultivated area, in plants separated by 20 m to avoid the collection of aphids belonging to the same colony and to obtain a good representation of the diversity of these insects. The individual colonies of aphids as well as the associated ants that were observed attending the aphid colonies were manually collected in 90 % ethanol, separated in the laboratory, and stored at -20 °C until analysis.
Identification of aphids and mutualistic ants: To identify the aphids present in the crops, the apterous adult insects collected were grouped according to the macroscopic morphological characteristics described by Holman (1974). For the DNA extraction and COI sequencing procedures, specimens of each morphological variant were randomly selected in each crop and sampled locality.
DNA extraction was performed using the complete body of each aphid and the commercial animal tissue kit from QIAGEN following the manufacturer’s recommendations. Amplification of the COI fragment (658 base pairs) was performed using primers LCO1490 (5’ ggtcaacaaatcataaagatattgg-3’) and HCO2198 (5’-taaacttcagggtgaccaaaaaatca-3’) (Folmer, Black, Hoeh, Lutz, & Vrijenhoek, 1994) in 25 μL of amplification cocktail with 1X PCR buffer solution, 2 mM MgCl2, 0.05 mM dNTPs, 0.25 μM of each primer, 1U of Taq DNA polymerase, and 5-10 ng of DNA. The temperature profile for the reaction included an initial denaturation step for 5 min at 92 °C, which was followed by 35 cycles of 30 s at 94 °C, 1 min at 52 °C, and 1 min at 72 °C; and a final extension for 5 min at 72 °C. Sequencing was performed via the specialized supplier Macrogen USA, and the sequences obtained were edited and aligned using Mega 7 (Kumar, Stecher, Tamura, & Dudley, 2016).
The COI identification sequence was mainly based on three aspects: the neighbour joining (NJ) analysis based on the Kimura two-parameter (K2P) nucleotide substitution model (Kumar et al., 2016), the estimation of genetic distances between COI haplotypes estimated with the K2P model using Mega 7 (Kumar et al., 2016) and a comparison of the similarity and sequence homology of the unique haplotypes obtained for aphids with those available in public databases, such as the NCBI GenBank (Hebert et al., 2003; Ratnasingham & Hebert, 2007). Sequences representing each haplotype per species on the different host plants were deposited in GenBank under the unique accession codes MK766466-MK766469.
To estimate the genetic distancing thresholds expected for the species found, the haplotypes reported in the NCBI database and their intraspecific and interspecific differentiation values were compared with those obtained by analysing the distance values in the collected aphids via the Automatic Barcode Gap Discovery ABGD programme (Puillandre, Lambert, Brouillet, & Achaz, 2012).
The identification of ants was performed at the Laboratory of Ant Biology and Ecology, Universidad del Valle (Cali, Colombia) using specialized taxonomic keys (Fernández, Guerrero, & Delsinne, 2019), the AntWeb v8.41 digital tool (California Academy of Sciences, 2020), and by direct comparison with specimens deposited at the Entomological Museum of Universidad del Valle (MUSENUV).
Mutual association between aphids and ants: To visualize the structural patterns of the host plant-aphid-ant trophic network, the programme Food Web Designer 3.0 was used (Sint & Traugott, 2016). To evaluate the effect of ant care on the size and structure of aphid colonies, generalized linear models were calculated using the programme R V3.4.4 (R Core Team, 2018). The response variables were the total number of aphids for each colony and the proportion of nymphs, with the latter used as a measure of the reproductive success of the colony.
Results
Identification of pest aphids: The 1 504 apterous adult insects collected were grouped according to the macroscopic morphological characteristics described by Holman (1974) in two groups corresponding to the genera Aphis (N= 1 460) and Myzus (N= 45). Aphis presented 4 colour variants, while Myzus presented only one.
Among the 144 specimens selected for the COI characterization, a total of 124 aphids of the genus Aphis were obtained, and they included four colour variants, yellow (21 specimens from tabasco pepper and 18 from cayenne pepper), beige (13 sampled in tabasco pepper and 15 from cayenne pepper), green (22 specimens from tabasco pepper and 19 from cayenne pepper), brown (5 specimens collected in tabasco pepper and 11 from cayenne pepper), while a total of 20 aphids of the genus Myzus were obtained, and they included a single colour pattern in both hosts. The NJ analysis based on the K2P nucleotide substitution model (Fig. 2) indicated that the different morphological groups corresponded to two genetic groups, with each genetic group only presenting one haplotype. The genetic distance estimated with the K2P model among the COI haplotypes was 9.3 %. A comparison of the haplotypes obtained with those available in the NCBI database indicated a similarity and homology of sequences of 100 %, with the collected specimens in group 1 corresponding to the species Aphis gossypii (Glover, 1877) and those in group 2 corresponding to the species Myzus persicae (Zulser, 1776).

Fig. 2 Neighbour joining (NJ) analysis based on the K2P model with 1 000 bootstrap samples using 144 COI sequences of aphids collected in cayenne (C. annuum) and tabasco (C. frutescens) pepper crops in Southwestern Colombia.
The haplotype sequences of A. gossypii (69 haplotypes, 276 sequences) and M. persicae (24 haplotypes, 101 sequences) available in the NCBI were analysed with ABGD software, and variation of less than 2 % was expected at the intraspecific level and the differentiation between these two species ranged between 9 and 11 %. These findings indicate that the distance value observed for this marker (9.3 %) between the two genetic groups of aphids collected in the cultivation of Capsicum spp. (Fig. 2) corresponded to interspecific differentiation.
Mutualistic association between aphids and ants: We found 23 colonies of M. persicae, with 19 in cayenne pepper crops and 4 in tabasco pepper; however, none of the colonies showed an association with ants (Fig. 1). We also found a total of 408 colonies of A. gossypii, with 192 in C. frutescens and 216 in C. annuum; and of these colonies, 46 C. frutescens colonies and 48 C. annuum colonies were attended by ants, for an incidence of ant care of 23.9 and 22.2 %, respectively (Fig. 1, Table 1). A total of 19 ant species were recorded from colonies of A. gossypii, and they represented four subfamilies and eight tribes, which are listed in Table 1. Of the 19 species, 11 and 12 attended colonies of A. gossypii in cayenne and tabasco pepper crops, respectively (Fig. 3). Of these ant species, Brachymyrmex heeri (Forel, 1874), Dorymyrmex brunneus (Forel, 1908), Monomorium floricola (Jerdon, 1851) and Paratrechina longicornis (Latreille, 1802) were found in common between the two crops. B. heeri represented 21 % of the interactions with aphids, as summarised in Table 1. The locality with the largest number of ant species attending to aphids was Toro, with a total of 10 species recorded. Finally, the species with the greatest distribution was M. floricola, which was recorded in five locations.
Table 1 Record of ant species that attend colonies of A. gossypii in crops of Capsicum spp. in Southwestern Colombia
| Ant species | Cayenne pepper C. annuum | Tabasco pepper C. frutescens | ||||||
| Bolívar | Roldanillo | Toro | Dagua | Guacarí | Rozo | Vijes | ||
| Azteca instabilis (Smith, 1862) | 0 | 0 | 1 | 0 | 0 | 0 | 0 | |
| Brachymyrmex heeri (Forel, 1874) | 2 | 7 | 5 | 0 | 0 | 0 | 6 | |
| Brachymyrmex longicornis (Forel, 1907) | 0 | 2 | 2 | 0 | 0 | 0 | 0 | |
| Brachymyrmex obscurior (Forel, 1893) | 0 | 0 | 1 | 0 | 0 | 0 | 0 | |
| Camponotus brevis (Forel, 1899) | 0 | 0 | 3 | 0 | 0 | 0 | 0 | |
| Camponotus lindigi (Mayr, 1870) | 0 | 0 | 0 | 4 | 4 | 0 | 0 | |
| Cardiocondyla obscurior (Wheeler, 1929) | 0 | 0 | 0 | 0 | 0 | 0 | 1 | |
| Crematogaster abstinens (Forel, 1899) | 0 | 0 | 0 | 0 | 1 | 0 | 0 | |
| Dorymyrmex brunneus (Forel, 1908) | 0 | 0 | 1 | 1 | 2 | 1 | 1 | |
| Linepithema neotropicum (Wild, 2007) | 0 | 0 | 0 | 0 | 1 | 0 | 0 | |
| Monomorium floricola (Jerdon, 1851) | 2 | 1 | 1 | 0 | 0 | 3 | 1 | |
| Nylanderia steinheili (Forel, 1893) | 0 | 0 | 1 | 0 | 0 | 0 | 0 | |
| Paratrechina longicornis (Latreille, 1802) | 1 | 2 | 1 | 0 | 6 | 0 | 0 | |
| Pheidole sp.1 | 0 | 0 | 0 | 2 | 9 | 0 | 0 | |
| Pseudomyrmex simplex (Smith, 1877) | 0 | 0 | 0 | 0 | 1 | 0 | 0 | |
| Solenopsis geminata (Fabricius, 1807) | 0 | 0 | 0 | 1 | 0 | 0 | 0 | |
| Tapinoma melanocephalum (Fabricius, 1793) | 1 | 2 | 3 | 0 | 0 | 0 | 0 | |
| Tetramorium bicarinatum (Nylander, 1846) | 9 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Tetramorium simillimum (Smith, 1851) | 0 | 0 | 0 | 1 | 0 | 0 | 0 | |
| Number of mutualistic events recorded | 15 | 14 | 19 | 9 | 24 | 4 | 9 | |
| Number of species involved in mutualistic interaction | 5 | 5 | 10 | 5 | 7 | 2 | 4 | |
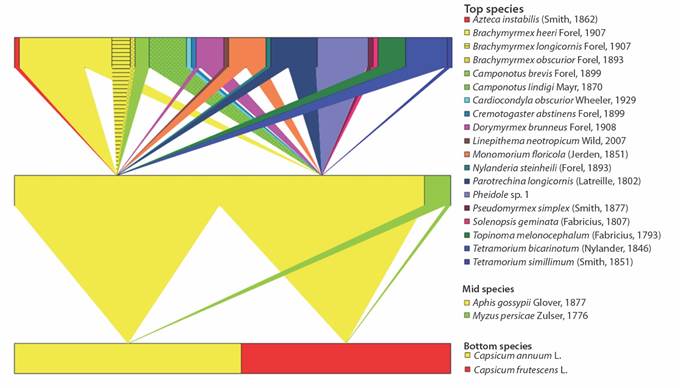
Fig. 3 Network illustrating the mutualistic relationship between ants and aphids that attack cayenne pepper C. annuum and tabasco pepper C. frutescens crops in Southwestern Colombia.
The colonies collected from M. persicae had an average colony size of 3 aphids, while A. gossypii had an average colony size of 10.5 aphids. Because only A. gossypii showed an interaction with ants, generalized linear models were estimated using the data of this species. This analysis indicates that A. gossypii colonies had a greater number of individuals when they were attended by ants for both crops (P < 0.05), while the proportion of nymphs per colony was higher in colonies attended by ants for tabasco pepper (P < 0.001) (Fig. 4, Table 2).
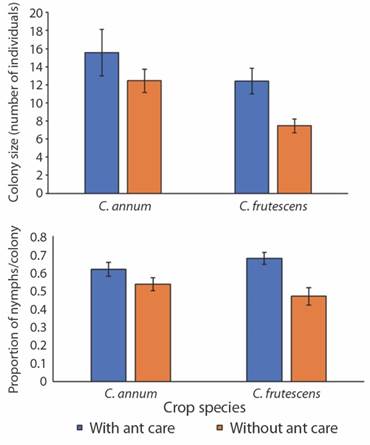
Fig. 4 Effect of ant care on the size of colonies and proportion of nymphs in colonies of A. gossypii established in cayenne and tabasco crops. The error bars indicate 95 % confidence intervals.
Table 2 Generalized linear models for colony size (number of individuals) and proportion of nymphs in colonies of A. gossypii established in cayenne pepper C. annuum and tabasco pepper C. frutescens
| Crop | Response var. | Distr. error | P (Chi) | DF resid. | Dev. Resid | Effect of ants on aphid colonies? |
| C. annuum | Colony size | Negative binomial | 0.0321 | 214 | 4.59 | Yes |
| Nymphs/colony | Binomial | 0.0619 | 214 | 3.49 | No | |
| C. frutescens | Colony size | Negative binomial | < 0.001 | 190 | 29.99 | Yes |
| Nymphs/colony | Binomial | < 0.001 | 190 | 12.28 | Yes |
Discussion
The biological identification of insect pests whose taxonomy is complex, such as aphids, is fundamental for establishing trophic interactions that may affect integrated pest management. Our results support the implementation of DNA barcoding as a cost-efficient aphid identification tool for non-specialists (Mazzi & Dorn, 2012) because aphid species could be differentiated with certainty on a regional spatial scale using 9.5 % of the adult specimens collected based on this tool and the availability of reference sequences in the NCBI. Other researchers have concluded that this tool is particularly helpful in identifying aphids, such as A. gossypii, which has different colour patterns in different hosts, e.g., Malvaceae, Cucurbitaceae and Solanaceae (Lokeshwari et al., 2014).
Although aphids in warm areas are anholocyclic and reproduce only by parthenogenesis (Blackman, 1974; Dixon, Kindlmann, Leps, & Holman, 1987), different phenotypes are presented depending on the stage of development of the insect, the host plant and environmental stress (Lokeshwari et al., 2014). Tálaga et al., (2017) reported that in C. annuum, M. persicae has a conserved phenotype during its different stages of development; however, on this same host, A. gossypii is polymorphic for body colour, and has a total of eleven phenotypes throughout its development. Our results are consistent with those reported by Tálaga et al., (2017) because we showed that in C. annuum and C. frutescens, the aphid M. persicae is monomorphic while A. gossypii shows polychromy. This pattern was consistent in the different locations of the inter-Andean valley of Southwestern Colombia. The molecular marker used here efficiently complemented the identification based on macroscopic morphological characteristics.
Although the DNA barcode has reported as a tool for conducting studies of genetic diversity and population genetics (Hebert & Gregory, 2005), in the case of Aphididae, intraspecific variation at the mitochondrial level is low (Xu, Chen, Cheng, Liu, & Francis, 2011). Moreover, the genetic diversity of these insects is much lower in areas where their introduction is more recent (Figueroa et al., 2005), which may explain the absence of intraspecific haplotype variation of A. gossypii and M. persicae at the sampled spatial scale because although the geographic origin of A. gossypii and M. persicae is not clear, Aphidinae is presumably native to temperate regions, especially of the Northern Hemisphere (Kim, Lee, & Jang, 2011). The polyphagous species A. gossypii, for example, presents an intraspecific differentiation of 0.62 % according to Foottit et al., (2008), whose study included specimens from locations that span the geographic range of the species.
Many species of the family Aphididae are important pests in agriculture worldwide because in addition to direct damage to plants caused by their feeding, they transmit phytopathogenic viruses (Blanc, Uzest, & Drucker, 2011; Fereres & Raccah, 2015). Capsicum spp. crops worldwide are affected by viral diseases belonging to the families Bromoviridae and Potyviridae, and both A. gossypii and M. persicae are vectors of these phytopathogens (Kenyon et al., 2014). A. gossypii is a competent vector of cucumber mosaic virus (CMV) (Bromoviridae) and of chilli veinal mottle virus (CVMV) (Potyviridae) (Hooks & Fereres, 2006; Kenyon et al., 2014), while M. persicae has been reported as a vector of CMV, pepper veinal mottle virus (PVMV) (Potyviridae) and potato virus Y (PVY) (Potyviridae) (Palukaitis, Roossinck, Dietzgen, & Francki, 1992; Martín, Collar, Tjallingii, & Fereres, 1997). In Southwestern Colombia, the pepper deforming mosaic virus (PepDMV) (Potyviridae: Potyvirus) has been reported as the predominant viral disease agent in Capsicum spp. (Pardey, Posso-Terranova, & García, 2005); however, the persistence, distribution, and insect vectors of these diseases in these crop systems are unknown. A. gossypii and M. persicae have not been reported as PepDMV vectors; however, it is necessary to evaluate their role in the transmission of this disease because insects of Aphididae are strongly associated with viruses of the family Potyviridae.
The colonies of M. persicae were found in lower proportions and densities in comparison to the colonies of A. gossypii on cayenne and tabasco peppers, although both aphid species present similar values in terms of life cycle duration, longevity and fertility when grown on cayenne pepper under laboratory conditions (Tálaga et al., 2017). The low density in the colonies of M. persicae compared to those of A. gossypii on the same hosts can be explained by the regional context of the agricultural system of peppers in Southwestern Colombia. In Colombia, peppers are intensively planted in large-scale mechanized agricultural systems (Corporación Colombia Internacional, 2006), although on a small scale, they are also planted in home gardens. These crops grow as a sun-exposed monocrop in the inter-Andean valley of the Cauca River, with an average temperature of 23.3 ± 6 °C and relative humidity of 79 ± 2.4 %. These conditions limit the development of M. persicae, which has lower reproductive performance and slower development time compared to A. gossypii when the daily temperature is above 20 °C (Satar, Kersting, & Uygun, 2008). A. gossypii shows optimal reproductive development above 17 °C on plants of Capsicum spp. (Satar et al., 2008), explaining why the regional conditions of the inter-Andean valley of the Cauca River possibly constitute an advantage for the colonization of pepper plants by this aphid.
The ants present in the pepper crops did not attend to colonies of M. persicae, and the number and density of their colonies were lower than those of A. gossypii. Previous studies have reported that A. gossypii frequently establishes interactions with ants (Kaplan & Eubanks, 2002; Kaplan & Eubanks, 2005; Powell & Silverman, 2010a) while M. persicae, which is considered a non-mutualistic species of aphid, does not (Powell & Silverman, 2010b). Although M. persicae has the same ability to produce sugar exudates with carbohydrate quality similar to that generated by other aphid species (Hogervorst, Wäckers, & Romeis, 2007), the ants do not interact with the colonies of this insect, and when both species are present, M. persicae is preferably consumed by the ants as prey (Powell & Silverman, 2010b). The response behaviour of M. persicae to potential predators explains why it does not establish a relationship with ants because this species of aphid rapidly disperses from the colony when disturbed by natural enemies and ants (Nault, Montgomery, & Bowers, 1976; Belliure, Amorós-Jiménez, Fereres, & Marcos-García, 2011). Attendance by ants is more likely when the aphid species presents swarming behaviour because the area attended by ants is smaller (Stadler & Dixon, 2005). In myrmecophiles (e.g., different species of the genus Aphis), the aphids of the colony are less easily dispersed or do not disperse in their interaction with ants because the alarm response is suppressed, which promotes the swarming behaviour of the colony (Nault et al., 1976).
Not all ant species attend to aphid colonies, and those that are involved in this interaction are opportunistic species that frequent extrafloral nectaries as a source of energetic resources (Delabie, 2001). In this study, species of Formicinae, Dolichoderinae and Myrmicinae were reported, and the genera reported in these subfamilies have been previously involved in this type of interaction with different species of Aphididae (Katayama & Suzuki, 2003; Stadler & Dixon, 2005; Buffa, Jaureguiberry, & Delfino, 2009; Silva & Perfecto, 2013; Campos & Camacho, 2014). The species of these subfamilies are considered omnivorous, opportunistic ants that are typical of agroecosystems and intervened habitats (Fontenla & Alfonso-Simonetti, 2018). Some species of Pseudomyrmex have been reported in this interaction with scale insects (Delabie, 2001; Ramírez, de Ulloa, Armbrecht, & Calle, 2001; Buffa et al., 2009) and aphids (Delfino, 2005; Silva & Perfecto, 2013). Species of this genus are considered to be general foragers and opportunistic predators (Fontenla & Alfonso-Simonetti, 2018).
A. gossypii colonies attended by ants had a greater number of individuals, which may promote infestations of Capsicum spp. in Southwestern Colombia. Ant care is influenced by the quality (composition) and quantity of the honeydew (Völkl, Woodring, Fischer, Lorenz, & Hoffmann, 1999; Katayama & Suzuki, 2002), which indicates that a larger colony may be more tractive to be attended by ants. However, studies carried out by Hosseini et al., (2017), indicate that regardless of the initial size of the colony, this interaction generates an increase in the density of the colony and the growth rate is higher compared to the unattended colonies. On the other hand, aphids in small colonies invest more energy supplying the supply of honeydew for ants; in large colonies, this effort is less because more individuals contribute to supply the demand for this substance (Katayama & Suzuki, 2002).
Studies carried out by Hosseini et al., (2017) and de Siqueira, Fagundes, Sperber and Fernandes (2011), indicate that this ant aphid interaction generates positive effects on the yield and biomass production of crop plants, among others, because the spread of microorganisms is prevented by avoiding the accumulation of aphid secretions, and the abundance of other herbivores is reduced. The interaction between A. gossypii and ants is of interest in biological control because ants interfere in the predation of this aphid by aphidophagous generalists, such as Hippodamia convergens Guérin-Méneville, 1842 (Coleoptera: Coccinellidae) and Chrysoperla carnea (Stephens) (Neuroptera: Chrysopidae) (Kaplan & Eubanks, 2002; Powell & Silverman, 2010a). Similarly, ant care reduces parasitism rates in A. gossypii colonies (Powell & Silverman, 2010a). Despite the fact that direct ant interference by potential predators or parasitoids was not observed in the field, our results indicate a positive effect of ant care on the reproductive effectiveness of the colony (increased production of nymphs). In the context of aphids as virus vectors, the mutualistic interaction between aphids and ants of the Solenopsis invicta Buren species in tomato (Solanum lycopersicum L.) promotes aphid abundance and results in an increase in the number of plants affected by CMV (Cooper, 2005). Though, it is necessary to consider the aphid-ant interaction role in the propagation of viral diseases in commercial crops of Capsicum spp. and to determine how this interaction may affect regional biological control strategies.
The aphid species that infest commercial crops of cayenne pepper C. annuum and tabasco pepper C. frutescens are A. gossypii and M. persicae, which are potential vectors of the virus that affect Capsicum spp. in Southwestern Colombia. A total of 19 ant species were found attending the A. gossypii aphids, whose attended colonies were significantly larger than those not attended by ants in commercial crops. The mutualistic aphid-ant relationship thus promotes an increase in the size of the colonies of this aphid species. It is advisable to explore the effect that ants can have on the effectiveness of aphidophagous generalists and specialists in the context of integrated pest management for aphids in pepper crops in Southwestern Colombia.
Ethical statement: authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.












 uBio
uBio 


