Introduction
Body size is an ecologically-influential trait that determines the susceptibility of endotherms to lower temperatures, so is a crucial feature for determining the vulnerability of endotherms to the effects of climate change; which is expected to be the most severe for animals inhabiting high altitudes (Swann, Fung, Levis, Bonan, & Doney, 2010; Öztürk, Hakeem, Faridah-Hanum, & Efe, 2015). Large animals will tend to produce more heat and to lose relatively less than small animals, which may represent an advantage in cold climates (Johnson, Adler, & Cherry 2000; Meiri & Dayan 2003; Freeman, 2017). Consequently, an expected pattern among closely-related animals is that populations living in colder climates, at high altitudes and latitudes, have larger bodies than those in warmer climates (Bergmann, 1847); this pattern is known as Bergmann’s rule. Even though this rule is considered a valid ecological generalization for birds and mammals (Blackburn & Gaston, 1996), research on this ecogeographical rule has mainly found clines in body size along latitudinal gradients for medium to large endotherms and has analyzed intra-specific variations (Ashton, Tracy, & Queiroz, 2000; Blackburn & Ruggiero, 2001; Ashton, 2002; Meiri & Dayan, 2003). For tropical species, the few evaluations of Bergmann’s rule along altitudinal gradients, have shown mixed support (Blackburn& Ruggiero, 2001; Freeman, 2017). Consequently, it is still unclear how generalizable is Bergmann’s rule to altitudinal clines for tropical species of endotherms (but see Blackburn& Ruggiero, 2001; Freeman, 2017); in particular, it remains unknown whether environmental variation along altitudinal gradients drives body size variation of small-bodied endotherms.
Small endotherms have high metabolic rates (Porter & Kearney, 2009) and small geographic ranges ( Gaston &Blackburn, 1996); these traits suggest that they may respond to climatic conditions along environmental gradients. Among endotherms, smaller animals have a higher amount of energy expended per unit of body mass than larger animals (White & Kearney, 2013). Besides, the energetic cost of using metabolic heat to keep a stable body temperature increases towards low temperatures (Kendeigh, 1969). Consequently, clades of small endotherms should exhibit a strong response according to Bergmann’s rule since a large body size can be adaptive by saving energy at the low temperatures expected in high altitudes and latitudes (Kendeigh, 1969). However, in addition to body size, species differ in other traits that can influence heat production and dissipation, which should be considered when investigating Bergmann’s rule. Traits related to metabolic rate act as important constraints on how species interact with their environment by determining other life history and behavior attributes of organisms (Brown, Gillooly, Allen, Savage, & West, 2004; Woodward et al., 2005). For small endotherms, torpor represents an energy-saving strategy in response to long periods of food shortage, low temperatures, or heavy rainfalls (Turbill, Bieber, & Ruf, 2011), which allow small-bodied endotherms such as hummingbirds to live at high altitudes in tropical mountains. For Neotropical hummingbirds, characteristics of torpor (i.e., frequency, duration) are strongly affected by body size and environmental temperature, so they differ among species and vary with altitude (C. Revelo, G. Londoño and O. E. Murillo-García personal communication). Thus, torpor may dilute the strongly-expected tendency of non-passerine birds (Kendeigh, 1969), such as hummingbirds, to exhibit larger body sizes in the cold environments of Tropical mountains.
To understand the influence of environmental variation along altitudinal gradients on body size variation of small endotherms, we evaluated whether variation in interspecific body size follows predictions from Bergmann’s rule along an altitudinal gradient. Specifically, we assessed the association among body mass and a descriptor of altitudinal distribution (mid-point of altitudinal range) of hummingbirds, while accounting for phylogenetic relatedness and different evolutionary models. We predicted that hummingbirds conform to Bergmann´s rule; however, contrary to expectations for non-passerine birds, they may not show a strong response to altitude in tropical mountains.
Materials and methods
data on body mass and altitudinal range: We compiled data on altitudinal distribution (lower and upper limits) and body mass for all hummingbird species of Colombia from both Hilty and Brown (1986) and Ayerbe-Quiñones (2015). For each species, we used the mean of the lowest and highest altitudinal limits (mid-point of elevational distribution) as a descriptor of altitudinal distribution. Besides, we used body mass as a measure of body size since other traits may not accurately reflect body size (Blackburn & Ruggiero, 2001; Meiri & Dayan, 2003). For species with sexual dimorphism in size, we used the mean body mass between males and females as a measure of body size. We follow the South American Classification Committee for taxonomic designations of species (Avendaño et al., 2017).
Data analyses: For phylogenetic analyses, we used a phylogenetic hypothesis for the evolutionary relationships among Hummingbirds (McGuire et al., 2014) based on two mitochondrial genes (ND2 and ND4), flanking tRNAs, and two nuclear introns (AK1 and BFib). We excluded from this tree the species that are not present in Colombia and Colombian species for which we do not have data on elevation or body mass. Thus, the final tree included 133 of the hummingbird species that inhabit Colombia.
We tested for the phylogenetic signal and mode of evolution of both body mass and mid-point of elevational distribution, by comparing the fit of different evolutionary models. Thus, we compared the fit of Brownian Motion, Ornstein-Uhlenbeck, Pagel’s lambda, and Early Burst evolutionary models with the fit of a model without phylogenetic signal that assumes no covariance structure among species (white noise model). Then, we identified the best evolutionary model for each trait by performing model selection with Akaike’s Information Criterion corrected for small sample sizes (AICc) (Burnham & Anderson, 2002).
The residuals of non-phylogenetic regressions of body mass on the mid-point of the altitudinal range showed a strong phylogenetic signal (λ= 0.994). Consequently, to evaluate the association between body mass and altitude (mid-point of elevational distribution), we compared the fit of Phylogenetic Generalized Linear Squared (PGLS) regressions under different evolutionary models (Brownian motion, Ornstein-Uhlenbeck and Pagel’s Lambda) with Akaike’s Information Criterion corrected for small sample sizes (AICc) (Burnham & Anderson, 2002). To determine the strength of the association among body mass and elevational mid-point, we estimated the determination coefficient as the square of the correlation between the actual and predicted outcomes of the model best supported by the data. We fitted the different models by using the fitContinuous function of the R package ‘geiger’ (Harmon, Weir, Brock, Glor, & Challenger, 2008).
Results
We found a strong phylogenetic signal for both body mass and mid-point of altitudinal distribution since all evolutionary models fitted better the data than the model with no phylogenetic signal (white noise) (Table 1). Thus, there is a tendency for related species to resemble each other more than to species drawn randomly from the phylogeny (Fig. 1). For body mass, the Early Burst model provides a better fit than the other evolutionary models. It had a lowerAICc, a better fit for the data (lnL= -233.42), and more than twice empirical support (evidence ratio > 2.7) than the second best-supported model (Table 1, see A). This model indicated that the rate of body mass change in hummingbirds had decreased exponentially through time (a= -0.07). On the other hand, for the mid-point of altitudinal distribution had a strong phylogenetic signal (λ= 0.89) with a Pagel’s Lambda evolutionary model (Table 1, see B), which indicates that elevational distribution changes continuously through time from a common ancestor in hummingbird clades.
TABLE 1 Test of phylogenetic signal for A. body mass and B. elevational range of tropical hummingbirds
| Model | k | lnL | AICc | ΔAICc | w |
| (A) Body mass (g) | |||||
| EB | 3.00 | -233.42 | 473.03 | 0.00 | 0.54 |
| BM | 2.00 | -235.46 | 475.02 | 1.98 | 0.20 |
| PG | 3.00 | -234.47 | 475.11 | 2.08 | 0.19 |
| OU | 3.00 | -235.46 | 477.11 | 4.08 | 0.07 |
| WN | 2.00 | -290.10 | 586.09 | 113.06 | 0.00 |
| (B) Elevational range (m) | |||||
| PG | 3.00 | -1044.33 | 2 094.84 | 0.00 | 1.00 |
| OU | 2.00 | -1053.79 | 2 113.76 | 18.92 | 0.00 |
| BM | 3.00 | -1057.23 | 2 118.55 | 23.71 | 0.00 |
| EB | 3.00 | -1057.23 | 2 120.65 | 25.80 | 0.00 |
| WN | 2.00 | -1109.68 | 2 223.45 | 128.61 | 0.00 |
k is the number of parameters, lnL is the log likelihood, AICc is the Akaike´s Information Criterion corrected for small sample sizes, ΔAICc is the difference in AICc between a given model and the model with the lowest AICc, and w is the weight of the AICc. BM: Brownian Motion model, PG: Pagel’s lambda model, OU: Ornstein-Uhlenbeck model, EB: Early Burst model, WN: White Noise model.
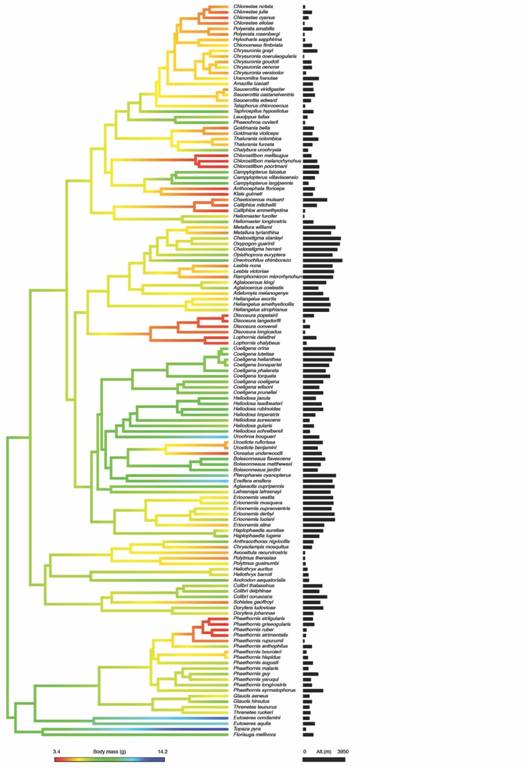
Fig. 1 Phylogeny of tropical hummingbirds illustrating the map of trait evolution for body mass and elevational mid-point. Black bars represent mid-point of elevational range (meters) for each species. We used the phylogeny of McGuire et al., 2014 and mapping of traits was accomplished by estimating states at internal nodes using maximum likelihood with the function contmap of the R package ‘phytools’ (Revell, 2012).
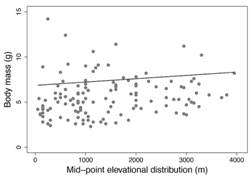
The comparison between models of phylogenetic regression analysis for the association between body mass and elevational ranges indicated that models that included altitude received more support than the null model (Σw elevation = 0.71). In particular, the Brownian model had a lower AICc, and the evidence ratio provides almost twice more empirical support for it than for Ornstein-Uhlenbeck (w BM/w OU= 1.7); besides, this Brownian model had a similar fit (lnL= -233.34) with fewer parameters (k= 3) than Ornstein-Uhlenbeck model (lnL=-232.88, k= 4). Thus, evidence indicates that a covariance under a Brownian evolutionary model provides a better fit to the data than the Ornstein-Uhlenbeck model (Table 2). Based on this model, there is a positive association between altitude and body mass (Intercept (α root )= 6.85,β altitude = 3.67 x 10-4 95 % confidence Interval [1.79 x 10-5, 7.2 x 10-4]) but it is weak (R 2 = 0.036); which indicates that hummingbirds tend to be slightly larger at higher altitudes (Fig. 2) and that body mass and altitudinal distribution are to some extent correlated through the phylogeny (Fig. 1).
TABLE 2 Evaluation of the relationship between body mass and midpoint of altitudinal range of Tropical hummingbird for different evolutionary models
| Models | k | AIC | lnL | ΔAIC | w | |
| Altitude-BM | 3 | 472.68 | -233.34 | 0.00 | 0.44 | |
| Altitude-PG | 4 | 473.76 | -232.88 | 1.08 | 0.26 | |
| Constant-BM | 2 | 474.93 | -235.46 | 2.24 | 0.14 | |
| Constant-PG | 3 | 474.93 | -234.47 | 2.24 | 0.14 | |
| Altitude-OU | 4 | 479.67 | -235.84 | 6.99 | 0.01 | |
| Constant-OU | 3 | 481.78 | -237.89 | 9.09 | 0.01 |
k is the number of parameters, lnL is the log likelihood, AICc is the Akaike´s Information Criterion corrected for small sample sizes, ΔAICc is the difference in AICc between a given model and the model with the lowest AICc, and w is the AICc weight. BM: Brownian Motion model, PG: Pagel’s lambda model, OU: Ornstein-Uhlenbeck model.
Discussion
The rate of evolution of hummingbird body mass showed a decelerating trend across time, whereas the evolutionary rate of the elevational range was slow and constant. We found that body mass and the elevational mid-point are weakly and positively associated throughout the phylogeny of hummingbirds. Therefore, our results suggest that altitudinal variation does not strongly influence body mass in hummingbirds.
Body mass and altitudinal distribution of hummingbirds showed a strong phylogenetic signal, which suggests that closely-related species are more similar for both traits than species that do not share a recent common ancestor. However, the mode of evolutionary change was different for these traits. We found that the evolution of body mass was better described by the Early Burst (EB) model of evolution, which indicates that hummingbird clades show a pattern of rapid and early morphological evolution followed by relative stasis. Evolution of body size has been characterized by a pattern of constraints to optimum values through time across different organisms (Harmon et al., 2010). Consequently, our results suggest that evolution of body size in hummingbirds follows an uncommon pattern for phylogenetic comparative studies; where morphological evolution is initially rapid and slows through time, a pattern expected under the classical model of adaptive radiation (Schluter, 2000). This pattern of body size evolution agrees with a recent analysis indicating that hummingbirds diversified after colonizing South America and the rise of the Andes and that the rate of diversification has been high but decelerating (McGuire et al., 2014). Together, these results suggest that body size has been an important trait related to initial diversification of phylogenetic clades of tropical hummingbirds. On the other hand, based on the strong phylogenetic signal found for altitudinal distribution, it is expected that tropical hummingbirds will to track their ancestral climatic regime in response to climate change. For Neotropical hummingbirds, upwards elevational shifts of between 300 and 700 m have been projected, depending on climate change scenario and original mean elevation of the target species (Buermann et al., 2011). Besides, the value for the lambda parameter for altitudinal distribution was less than one and lower than for body size; which indicates that additional factors, different from the phylogenetic history of hummingbirds, have an impact on the evolution of altitudinal distribution (Münkemüller et al., 2012). Consequently, results suggest that elevational ranges would be comparatively less affected by phylogenetic relationships and more affected by the ecological context than body mass, as has been reported for fishes (Hernández et al., 2013).
As expected under Bergmann’s rule, we found that hummingbird species have larger body masses at higher elevations where temperatures are coldest. This pattern, particularly prevalent for birds, has been frequently reported for endotherms along latitudinal gradients (Ashton, 2002; Meiri & Dayan, 2003). However, the assessment of variation in body size along elevational gradients for tropical endotherms has shown mixed results. For Andean passerine birds, body mass was positively correlated with the elevational range of the species, but there was no significant relationship with latitudinal range (Blackburn & Ruggiero, 2001). On the other hand, in a more comprehensive study, different assemblages of tropical montane passerines from different biogeographical regions did not show larger body masses at higher elevations where temperatures are colder (Freeman, 2017). For tropical mammals, studies of interspecific variation in body mass show result not consistent with Bergmann’s rule, as in birds. Gohli and Voje (2016), found no reliable support for Bergmann’s rule among 22 mammalian families (many tropical); which suggests that neither variation in latitude nor temperature is an important interspecific phenomenon for body size in mammals at the family level. These results suggest that temperature is not a generally-important driver of body size evolution in tropical endotherms as birds and mammals.
Our results indicate that body mass and altitudinal distribution are correlated through the phylogeny. It is expected that extreme environmental conditions should be significant drivers of body size evolution (Root, 1988) and that birds would conform to Bergmann’s rule. In particular, non-passerine species are more affected by cold than passerine species (Kendeigh, 1969). This phenomenon may imply that the selective pressures exerted by low temperatures are stronger for non-passerines, and thus make them more inclined to vary in their body sizes in agreement with Bergmann’s rule (Meiri & Dayan, 2003). Additionally, the fact that large-bodied species tend to have lower critical temperatures at lower ambient temperatures than small-bodied species indicates that small species are more affected by cold than large species (Kendeigh, 1969). Birds and mammals of lower body size (4-500 g) show a significant tendency to follow Bergmann’s rule (Meiri& Dayan, 2003). Therefore, being non-passerine and having small body sizes, it would be expected that hummingbirds show a strong cline in body mass as a response to elevational changes in environmental temperature. However, contrary to those expectations, the degree of association between body mass and elevational distribution was not as substantial as would be expected for small non-passerine birds such as hummingbirds.
In addition to body size, metabolic rate act as an important constraint to interact with environmental conditions (Brown et al., 2004; Woodward et al., 2005). Frequency of torpor is affected by both environmental temperature (altitude) and body mass. In an assessment of torpor use for 28 hummingbird species along a 1 900-meter elevational gradient in the Colombian Western Andes (C. Revelo, G. Londoño and O. E. Murillo-García personal communication), bigger-bodied hummingbirds used torpor less frequently. Furthermore, species inhabiting high elevations use torpor more frequently and for longer durations, and have lower body temperatures than lowland species. Together, these findings indicate consistently that in hummingbirds, smaller species can use torpor more frequently in response to low environmental temperatures than larger species. Therefore, small hummingbirds could, due to the ability to use torpor more frequently and for more extended periods in response to lower environmental temperatures, live at higher altitudes in the tropical mountains that would be expected for their body size; which could account, in part, for the lack of conformity with Bergmann’s rule.
Even though temperature effects are size specific (Lindmark, Huss, Ohlberger, & Gårdmark, 2018) and seasonal and daily fluctuations in temperature may influence body size, we found that body mass slightly predicted the values of altitudinal ranges of hummingbirds. In the same way, the lower body size categories of mammals have shown a lesser tendency to conform to the rule (Meiri & Dayan, 2003); in particular, small tropical species such as Murid rodents and Phyllostomid bats do not follow the predictions of Bergmann’s rule (Gohli & Voje, 2016). Alternatively, physiological mechanisms involved in thermoregulation may be an essential aspect of determining body size variation along altitudinal gradients. For Andean hummingbirds, small species tend to have a higher frequency and duration of torpor at higher altitudes than at lower altitudes (C. Revelo, G. Londoño y O. E. Murillo-García personal communication). Consequently, our result suggests that torpor can disrupt the strongly expected relationship between environmental temperature and body size for small-bodied and non-passerine endotherms such as hummingbirds. Thus, we conclude that environmental variations across altitudinal gradients seems to weakly influence body size in small tropical endotherms.
Ethical statement: authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.












 uBio
uBio