Introduction
Peru’s territory is located according to the biogeographical zoning in the world’s neotropical area (Udvardy, 1975). It is included in one of the world’s 25 biodiversity hotspots (Myers, Mittermeier, Mittermeier, Da Fonseca, & Kent, 2000). The uniqueness of the climatic conditions caused primarily by the presence of mountain barriers that have influenced the formation of the neotropical flora and fauna over several million years (Antonelli, Nylander, Persson, & Sanmartín, 2009). Recent biogeographic partitioning of this region rests predominately on the studies of mosses, ferns, flowering plants, insects, birds, and small mammals (Morrone 2001, 2006, 2017; Hoorn et al., 2010; Herzog, Martínez, Jørgensen, & Tiessen, 2011). Less attention is paid to unicellular organisms, which macroscale patterns may considerably differ from those of macroorganisms (Martiny et al., 2006; Azovsky & Mazei, 2013, Azovsky, Tikhonenkov, & Mazei, 2016). Among protists, testate amoebae represent a common diverse and abundant component of both aquatic and terrestrial habitats, playing a vital role in ecosystem functioning. For example, in the soils of the southern taiga, they occupy in terms of biomass second place after fungi, exceeding the biomass of bacteria (Schröter, Wolters, & De Ruiter, 2003). These microorganisms are among the larger protist groups (most taxa 5-500 μm in length) and play important roles as consumers of smaller microorganisms and in the case of some species, in primary production of endosymbiotic algae (Wilkinson & Mitchell, 2010; Jassey et al., 2015). Thus, soil-inhabiting testate amoebae as part of terrestrial ecosystems can be used as biological indicators (Payne, 2013). However, their geographical distribution remains poorly understood, although we know that most of the species are cosmopolites with several cases of limited geographic distribution (Foissner, 2006; Smith & Wilkinson, 2007; Smith, Bobrov, & Lara, 2008; Heger et al., 2011a; Azovsky & Mazei, 2013; Azovsky et al., 2016).
There are a limited number of publications on Peru and contiguous areas of testate amoebae. The data represent modern and palaeo wetlands, lake sediments and terrestrial habitats (Bonnet, 1966; Haman, 1994; Krashevska, Bonkowski, & Maraun, 2007; Krashevska, Maraun, & Scheu, 2012; Swindles et al., 2014; Swindles, Lamentowicz, Reczuga, & Galloway, 2016; Patterson, Huckerby, Kelly, Swindles, & Nasser, 2015; Reczuga, Swindles, Grewling, & Lamentowicz, 2015). Two hundred and fifty four taxa of testate amoebae were found in Mexico in another study in the neotropics region (Bobrov, Krasilnikov, & García-Calderón, 2013).
The purpose of this study is to investigate the diversity and distribution of testate amoebae in Peru in different habitats as well as to discuss the geographic distribution in the neotropics of testate amoebae.
Materials and methods
The study is based upon the diversity of testate amoebae from 28 surface (0-3 cm depth) soil samples collected in Peru in March 2016 (Table 1). The collection took place nearby Cuzco (6 samples), the ancient city of Machu Piсchu (17 samples) and in nearby Aguas Calientes (5 samples). Furthermore, one bottom sediment sample was extracted from Lake Titicaca, near the city of Puno (Fig. 1).
TABLE 1 Sample sites description
| Nº | Place of sampling | Altitude (m) | Coordinates | Number of testate amoeba species identified |
| 1 | Cuzco. Grassland | 3 607 | S13° 30.254' W71° 58.864' | 1 |
| 2 | Cuzco. Under the tree crown | 3 800 | S13° 28.719' W71° 58.060' | 5 |
| 3 | Cuzco. Grassland | 3 821 | S13° 28.719' W71° 58.060' | 7 |
| 4 | Cuzco. Grassland | 3 643 | S13° 30.514' W71° 58.278' | 7 |
| 5 | Cuzco. Grassland | 3 449 | S13° 24.368' W71° 50.686' | 4 |
| 6 | Cuzco. Under the cactus | 3 450 | S13° 24.488' W71° 50.651' | 5 |
| 7 | Machu Picchu. Rainforest | 2 468 | S13° 09.917' W72° 32.621' | 36 |
| 8 | Machu Picchu. Rainforest | 2 468 | S13° 09.917' W72° 32.621' | 32 |
| 9 | Machu Picchu. Rainforest | 2 468 | S13° 09.917' W72° 32.621' | 52 |
| 10 | Machu Picchu. Rainforest | 2 554 | S13° 10.107' W72° 32.539' | 23 |
| 11 | Machu Picchu. Rainforest | 2 554 | S13° 10.107' W72° 32.539' | 35 |
| 12 | Machu Picchu. Rainforest | 2 554 | S13° 10.107' W72° 32.539' | 49 |
| 13 | Machu Picchu. Rainforest | 2 648 | S13° 10.234' W72° 32.237' | 14 |
| 14 | Machu Picchu. Lichen | 2 694 | S13° 10.177' W72° 32.082' | 11 |
| 15 | Machu Picchu. Grassland | 2 727 | S13° 10.182' W72° 32.036' | 34 |
| 16 | Machu Picchu. Grassland | 2 706 | S13° 10.185' W72° 32.117' | 4 |
| 17 | Machu Picchu. Rainforest | 2 692 | S13° 10.205' W72° 32.172' | 24 |
| 18 | Machu Picchu. Rainforest | 2 623 | S13° 10.201' W72° 32.397' | 28 |
| 19 | Machu Picchu. Grassland | 2 529 | S13° 09.988' W72° 32.636' | 1 |
| 20 | Machu Picchu. Rainforest | 2 461 | S13° 09.700' W72° 32.749' | 3 |
| 21 | Machu Picchu. Rainforest | 2 456 | S13° 09.709' W72° 32.750' | 28 |
| 22 | Machu Picchu. Rainforest | 2 444 | S13° 09.808' W72° 32.663' | 1 |
| 23 | Machu Picchu. Agave | 2 450 | S13° 09.909' W72° 32.604' | 7 |
| 24 | Aguas Calientes (Sacred Valley). Rainforest | 2 056 | S13° 09.291' W72° 31.839' | 1 |
| 25 | Aguas Calientes (Sacred Valley). Rainforest | 2 056 | S13° 09.291' W72° 31.839' | 13 |
| 26 | Aguas Calientes (Sacred Valley). Rainforest | 2 056 | S13° 09.291' W72° 31.839' | 21 |
| 27 | Aguas Calientes (Sacred Valley). Rainforest | 2 056 | S13° 09.291' W72° 31.839' | 16 |
| 28 | Aguas Calientes (Sacred Valley). Rainforest | 2 056 | S13° 09.291' W72° 31.839' | 12 |
| 29 | Lake Titicaca, sediments, near the town of Puno | 3 825 | S15° 50.120' W70° 00.935' | 2 |
The region of study (Cuzco and Machu Picchu) belongs to the system of cultivated land, forest and scrubland and is located on the western border of biodiversity priority areas with a high degree of endemity (Neugarten et al., 2015). The region has been influenced by human activity for many centuries, leading to a combination of manmade habitats, paramo grassland, Polylepis thickets, partially degraded virgin forest and former cultivated lands that has reverted back to forests or scrubs (Parker, Parker, & Plenge, 1982). Soils in Machu-Picchu represent Dystric Regosols (Arenic) type, and soils in Cuzco represent Eutric Regosols type (Gardi et al., 2015).
Samples for testate amoebae were prepared using a method based on wet sieving. 1 cm3 of sample was soaked in water for 24 h, stirred, filtered at 0.5 mm, the suspension left to settle for a further 24 h, and supernatant decanted off following the methods applied by Mazei, Blinokhvatova, and Embulaeva (2011). No back-filtering step was used as this leads to the loss of small taxa; relatively large mesh size (500 µm) was used to retain the largest shells of testate amoebae (Payne, 2009; Avel & Pensa, 2013). The specimens were revealed under a light using a biological microscope (Zeiss Axioplan 2) at x200 and x400 magnifications.
Species accumulation curve was constructed based on rarefaction procedure performed in PRIMER 6.1.6 (Clarke & Gorley, 2006). The maximum expected number of species was calculated in PRIMER 6.1.6 by the nonparametric Chao2 method, which takes into consideration the theoretical number of expected rare species (Clarke & Warwick, 2001).
Results
A total of 144 taxa were found in 29 samples:
Arcella arenaria compressa
Centropyxis aculeata
C. acuminata
C. aerophila
C. aerophila cornata
C. aerophila deflandrei
C. aerophila minuta
C. aerophila sphagnicola
C. cassis
C. constricta
C. constricta gigas
C. constricta minuta
C. deflandriana
C. delicatula
C. discoides
C. elongata
C. elongata minor
C. gibba
C. latideflandriana
C. minuta
C. orbicularis
C. ovalis
C. cf. pannosus
C. plagiostoma
C. plagiostoma lata
C. plagiostoma longa
C. plagiostoma minor
C. plagiostoma terricola
C. pyriformis
C. stenodeflandriana
C. sylvatica
C. sylvatica minor
C. sp. (1-9)
Cyclopyxis cf. arcelloides
C. arcelloides gibbosa
C. eurystoma
C. eurystoma parvula
C. intermedia
C. kahli
C. kahli cyclostoma
C. lithostoma
C. machadoi
C. penardi
C. plana
C. puteus
C. sp.
Trigonopyxis arcula
T. arcula major
T. minuta
Plagiopyxis barrosi
P. bathystoma
P. callida
P. callida grandis
P. declivis
P. declivis oblonga
P. labiata
P. minuta
P. minuta oblonga
P. penardi
P. penardi oblonga
Geamphorella lucida
Schoenbornia humicola, Sch. smithi
Difflugia decloitrei
D. lucida
D. oblonga
D. pristis
Awerintzewia cyclostoma
A. sp.
Heleopera foissneri
H. petricola
H. petricola amethystea
H. petricola humicola
H. petricola major
H. sphagni
H. sylvatica
Hyalopshenia insecta
H. minuta
Nebela bohemica
N. collaris
N. griseola
N. lageniformis
N. minor
N. parvula
N. tubulata
N. wailesi, N. sp. (1-2)
Certesella certesi
Argynnia caudata
A. dentistoma
A. retorta
A. spicata
A. vitraea
Apodera vas
Porosia bigibbosa
Quadrulella quadrigera
Q. symmetrica
Phryganella acropodia
Valkanovia elegans
V. delicatula
Assulina muscorum
Euglypha cf. acantophora
E. ciliata glabra
E. cristata
E. cristata decora
E. compressa
E. cuspidata
E. dolioliformis
E. filifera pyriformis
E. laevis
E. polylepis
Tracheleuglypha acolla
T. acolla minor
Sphenoderia fissirostris
S. macrolepis
S. sp.
Corythion dubium
C. dubium minima
C. orbicularis
C. pulchellum
Trinema complanatum
T. grandis
T. enchelys
T. lineare
T. lineare minuscula
T. lineare terricola
T. penardi
T. cf. caudatum
Cryptodifflugia minuta
Testate amoebae unidentified (spec. 1-6)
Nineteen taxa (13.2 % of the total number of taxa recorded) have not been identified to the species level. Twenty three taxa (16.0 %) have a limited geographical distribution: Centropyxis acuminata, C. cf. compressa, C. deflandriana, C. delicatula, C. latideflandriana, C. cf. ovalis, C. cf. pannosus, C. pyriformis, C. stenodeflandriana, Cyclopyxis lithostoma, C. machadoi, C. plana, Geamphorella lucida, Awerintzewia cyclostoma, Plagiopyxis barrosi, Heleopera foissneri, Certesella certesi, Argynnia retorta, A. spicata, Apodera vas, Quadrulella quadrigera, Sphenoderia macrolepis, Trinema cf. caudatum (Fig. 2). Over a half of the species found in the samples belong to five genera: Centropyxis, Cyclopyxis, Plagiopyxis, Euglypha, and Nebela.
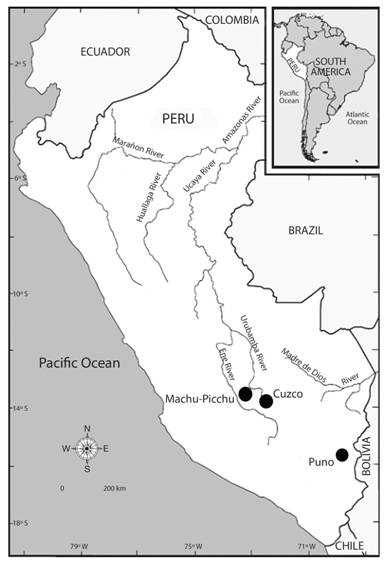
Fig. 2 Micrographs of some rare species of testate amoebae in studied regions (magnification 400x): A. - Apodera vas; B. - Argynnia retorta; C. - Centropyxis cf. compressa; D. - Certesella certesi.
Different habitats yielded various species richness (Table 1). Results of rarefaction procedure (Fig. 3) show that species-accumulation curve does not reach a plateau. The curve is well fitted (R2 = 0.98) by the power function S = 20.56N 0.57 (where S is the number of species revealed, N is the number of samples investigated). Low value of power coefficient (0.57) reflects an unsaturated community with 20.56 as an average number of taxa per sample. Expected total number of species (Chao2) in the studied area is estimated at 189.
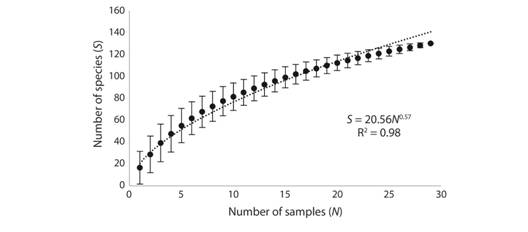
Fig. 3 Relations between number of samples investigated (N) and number of species identified (S). Whiskers - standard deviation.
Six samples from various types of habitats near the city of Cuzco (altitudes from 3607 m to 3450 m) yielded twenty taxa of testate amoebae of the cosmopolitan group. Most of these also belong to eurybiontic species, with the exception of several taxa, such as Plagiopyxis labiata, Heleopera petricola, Valkanovia delicatula, Cryptodifflugia minuta, each which usually inhabit permanently or temporarily in wet habitats enriched with organic substances.
Twelve samples from the rainforest near Machu Picchu (heights from 2 444 m to 2 692 m) revealed the most diverse testate amoebae community (108 taxa). In addition, 17 testate amoebae from these samples were not determined to the species level. Besides, the location has yielded almost all species with a limited geographical distribution. Representatives from all ecological groups of testate amoebae were noted here with the prevalence of hygro- and hydrophilic species from the genera Arcella, Difflugia, Trigonopyxis, Centropyxis, Cyclopyxis, Heleopera, Hyalosphenia, Nebela, Porosia, Argynnia, Quadrulella, and Sphenoderia. The environmental condition is due to high humidity resulting from significant seasonal amount of precipitation, fog and cloudiness. The highest diversity (52 taxa) was observed in the sample # 9 (Table 1), a population which is represented by all ecological groups of testate amoebae - that is eurybiont, pedobiont, sphagnobiont, hydrobiont, acidophilic, and calcific.
In a single sample from lichen habitat near Machu Picchu, 11 taxa were found, represented by xerophilic species from the genera Assulina, Corythion, as well as eurybionts from the genera Centropyxis (including rare species C. deflandriana and C. latideflandriana), Cyclopyxis, and Trinema.
In three samples from grassland habitats near Machu Picchu, 37 testate amoebae taxa were identified. Communities include high number of hygrophilous species Arcella arenaria compressa, Trigonopyxis arcula, T. minuta, Centropyxis cassis, C. constricta, C. elongata, C. orbicularis, Heleopera petricola amethystea, H. petricola humicola, H. sylvatica, Nebela tubulata, N. wailesi, Argynnia caudata, A. spicata, Quadrulella quadrigera, and Difflugia lucida, reflecting a wet hydrological regime of the grasslands due to specific climatic features of this mountainous region of Peru. The calciphilic testate amoebae species dwelling the region, are for example Centropyxis plagiostoma, C. plagiostoma lata, C. plagiostoma minor, and Cyclopyxis kahli indicate near-neutral pH of the soil fluids. Among the rare species, only one was found, namely Centropyxis deflandriana.
Eleven testate amoebae taxa were found in one sample taken from the soils near agave, among which one can distinguish a group of calciphilic species: Centropyxis plagiostoma, C. plagiostoma minor, and exclusively pedobiont species Plagiopyxis minuta and P. penardi. The findings of a moderately hygrophilous species H. petricola humicola indicate relatively favorable soil moisture conditions. Besides, the location provided a rare species of Centropyxis acuminata.
Forty-two testate amoebae taxa were found in five samples at Aguas Calientes at an altitude of 2 056 m.a.s.l. When comparing with the higher elevated forests of Machu-Picchu (above 2 444 m.a.s.l.), the number of hydrophilic taxa decreased. There is no species of the genus Trigonopyxis. Species from the genera Heleopera, Nebela, Argynnia are rare. All of this most likely reflects decreasing humidity levels below the slope. Topography is a predictor of species composition (Basnet, 1992).
The only bottom sample from Lake Titicaca yielded only two hydrobiont species from the genus Difflugia, i.e. D. oblonga and D. pristis.
Discussion
The broadleaved forest biome, also known as Amazonia or Amazon Jungle, covers most of the Amazon River basin and forms part of the Neotropic bioregion. A significant part of the Amazon basin falls on the territory of Peru. The age of the rainforest is estimated to be about several tens of millions of years (Mark, Yadvinder, Oliver, & Sharon, 2005). In fact, the age of the Amazonia region has been determined to be approximately 55 million years (Burnham & Johnson, 2004; Morley, 2000). The largest in South America, the Amazon rainforest is a place of an unprecedented biodiversity (Turner, 2001). The diversity of plant species is the highest in the world: as has been reported, 0.25 km2 (62 acres) in the Ecuadorian rainforest supports more than 1 100 species of trees (Wright, 2001). Considering that Ecuador holds about 2 % of the area that comprises the Amazon, the exceptional biodiversity of the entire forest need be noted.
In this study, we have attempted to understand to what extent the region of the Peruvian rainforests is unique in respect to testate amoebae as a common eukaryotic microbial component of terrestrial ecosystems (Geisen et al., 2017). The findings of 144 testate amoebae taxa in 29 samples, including 19 taxa not identified to the species level, is indicative of a high potential level of terricolous testate amoebae diversity, which can be caused by a diversity of microhabitat types produced by higher vascular plants and the antiquity of the Amazonian landscapes, and accentuate the importance of this part of the neotropics as a hotspot of biodiversity (Myers et al., 2000).
Previously, even a greater percentage of testate amoeba species of an uncertain taxonomic position was noted for Mesoamerica, in particular Mexico, where this figure exceeded 30% of the total biodiversity described (Bobrov et al., 2013). In the more northern part of the neotropics, 254 taxa of testate amoebae were found (Bobrov et al., 2013). The highest species diversity of testate amoebae was attributed to the soils of the tropical rainforests (126 taxa) and wetlands (144 taxa), including organisms with a limited geographical distribution (from the genera Cornuapyxis, Ellipsopyxis, Hoogenraadia, Planhoogenraadia, Apolimia, Certesella, Apodera and Alocodera).
In the course of the study of terricolous testate amoebae of the neotropical area of Ecuador’s mountain habitats (Krashevska et al., 2007, 2012) 135 testate amoebae taxa were identified in 36 samples. The vast majority of the identified testate amoebae were rather common and can be found also in temperate areas. Nine species (6.7 % of the total list) were tropical, thus were reckoned by authors as relicts of Gondwana (Krashevska et al., 2007; Krashevska et al., 2012).
Earlier studies of testate amoebae in Amazonian Sphagnum ombrotrophic bogs revealed 47 taxa (Swindles et al., 2014, 2016) and provided the description of a new species, namely Arcella peruviana (Reczuga et al., 2015). Another study revealed the possibility to use testate amoebae as a bioindicator of the aquatic environment in the Amazon basin (Arrieira, Schwind, Alves, & Lansac-Tôha, 2017).
This study revealed several rare species. Most located in limited geographical areas (genera Certesia, Apodera, Argynnia, and some representatives of the genera Centropyxis and Cyclopyxis) inhabiting the rainforests. Most likely, due to the high humidity of the upper soil layer and the microhabitat diversity of environmental conditions contributed to the limited distribution. Likely, high spatial heterogeneity appears in the region’s diversity of soil types because of the features of its history and its complex relief (Quesada et al., 2011). Both factors, namely high spatial heterogeneity and high humidity, created the proper conditions for the high biodiversity, maintenance, including that of the soil-dwelling testate amoebae.
The distribution of Centropyxis latideflandriana and C. stenodeflandriana includes the Australian biogeographical regions (Bonnet, 1979; Meisterfeld & Tan, 1998), oriental (Bonnet, 1979; Balik, 1995; Bobrov, Mazei, & Tiunov, 2010), and the Southern part of Palearctic zone (Bonnet, 1979; Bobrov, 2001; Bobrov et al., 2013). Nine species of the genus Centropyxis are now found exclusively outside the Holarctic: Centropyxis acuminata, C. deflandriana, C. delicatula, C. latideflandriana, C. cf. ovalis, C. cf. pannosus, C. pyriformis, C. serrahni, C. stenodeflandriana (Bobrov et al., 2010).
Two “flagship” (sensu Foissner, 2006) species, namely Apodera vas and Certesella certesi were found in the same locations during our study. The former species seemed to be more common for Mesoamerica (Heger, Lara, & Mitchell, 2011b) and tends to inhabit in wetter habitats (freshwater sediments, wetlands) in comparison to the later taxon (Lansac-Tôha, Velho, Takahashi, Aoyagui, & Bonecker, 2001; Miranda & Mazzoni, 2015). Geographic distribution of Apodera vas is well documented beyond the limits of Holarctic (Mitchell & Meisterfeld, 2005; Smith & Wilkinson, 2007; Smith et al., 2008). The hypothesis on gondwanian origin of Apodera vas and later distribution through Pacific Ocean islands has previously been advanced (Smith & Wilkinson, 2007). In South America, the main findings of Apodera vas is limited to the narrow band of the Pacific coast along the Andes mountains from Tierra del Fuego, Cape Horn and the Patagonia in the South to Mexico in the North: Argentina (Vucetich, 1975), Chile (Zapata & Fernandez, 2008; Fernandez & Zapata, 2010-2011; Fernandez, Zapata, Meisterfeld, & Baessolo, 2012; Fernandez, Lara, & Mitchell, 2015), Southern Ecuador (Krashevska et al., 2007), Costa Rica, Guatemala (Laminger, 1973), Mexico (Golemansky, 1967; Laminger, 1973; Heger et al., 2011b; Bobrov et al., 2013). Almost all of the above-mentioned publications indicate joint occurrences of Apodera vas and Certesella certesi, latter sometimes replaced by the morphologically close Certesella martiali (Vucetich, 1978).
Heger et al. (2011b) investigated the northern limits of Apodera vas and Certesella certesi distribution within Mesoamerica based on the Great American Interchange idea (Simpson, 1940). The analysis of more than 200 samples has revealed a continuous geographic distribution of both the Apodera vas and the Certesella certesi within the intermediate zone ranging from the Neotropical realm toward the Nearctic realm, i.e. Panama, Costa Rica, Nicaragua, Salvador, Honduras, Guatemala, and Mexico. Also, joint occurrences were noted in four countries: Panama, Costa Rica, Guatemala, and Mexico.
In the eastern plains of South America outside the Andes, Apodera vas was found in water ponds (Lansac-Tôha et al., 2001; Miranda & Mazzoni, 2015). Neither there, nor in the swamps of the Peruvian Amazon, the joint findings of Apodera vas and Certesella certesi has ever been detected (Swindles et al., 2014).
Our study fills a geographical gap that consisted in an insufficiency of collected data on the findings of Apodera vas and Certesella certesi in Peru, and illustrates the continuity of these species expansion along the Pacific coast.












 uBio
uBio