Introducción
Among Meliaceae species, mahogany (Swietenia macrophylla King) is a valuable tree naturally distributed from Mexico to Colombia (Amin, 2012). Its wood is used commercially for furniture manufacturing due to its hardness, strength, beauty and quality (Azad & Matin, 2015). In recent decades, intensive exploitation and inadequate use of this species have resulted in the loss of natural populations (Maruyama, 2009). Conventional propagation of mahogany by seeds is difficult due to the low germination rate (Amin, 2012). In addition, the establishment of commercial plantations of mahogany is limited by the shoot borer (Hypsipyla grandella Zeller, Lepidoptera: Pyralidae), which is the most harmful pest of tree species in the Meliaceae family (Maruyama, 2009). As a result, mahogany is considered an endangered species by the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) (Maruyama, 2009; Pérez-Flores, Aguilar-Vega, & Roca-Tripepi, 2012).
Somatic embryogenesis (SE) is used to propagate plants commercially or to study basic aspects of induction and development of somatic embryos. Also, SE offers an efficient in vitro regeneration approach as a fundamental step in plant genetic improvement (Loyola-Vargas, 2016). SE is defined as the development of somatic embryos from haploid or diploid somatic cells without gamete fusion (Jiménez, 2001; Jiménez, 2005). Several factors affect SE, such as genotype, kind of explant and plant growth regulators. Appropriate conditions for SE induction are usually established empirically through trial and error experiments (Jiménez, 2001; Jiménez, 2005; Loyola-Vargas, 2016). Morphological, anatomical, biochemical and histological studies could contribute to the understanding of the somatic embryogenesis process and provide clues for the optimization of protocols involving this regeneration route (Yeung, 1999; Silva et al., 2015).
Although protocols for somatic embryogenesis have been reported in mahogany (Collado et al., 2006; Maruyama, 2009; Collado et al., 2010), to the best of our knowledge, no histological or biochemical analyses of SE development have been described. The aim of this study was to describe the histological and biochemical characteristics of SE in order to elucidate this unexplored process in Swietenia macrophylla and to determine the embryogenic origin of the calli.
Materials and methods
Plant material: unripe fruits were collected from mahogany trees (Swietenia macrophylla) located in Parque Metropolitano La Sabana, San José (9°56’08” N & 84°06’15” W) and in Santa Rosa National Park, Guanacaste (10°53’01” N & 85°46’30” W), Costa Rica, between December 2014 and January 2015. Fruits were disinfected in a solution of sodium hypochlorite (3.5 % v/v) for 30 min with constant shaking, then soaked for 2 min in 70 % (v/v) ethanol and rinsed three times with sterile water. Then, the cotyledons were separated from the seeds and used as starting material.
Somatic embryogenesis protocol: cotyledons were cultured in culture jars with 20 ml of the somatic induction medium (SIM; Collado et al., 2010) in darkness at 25.0 oC ± 2.0. Six weeks later, primary callus and explants were transferred to culture jars with 20 ml of differentiation culture medium (Collado et al., 2010).
For germination, somatic embryos were cultured in 125 ml Erlenmeyer flasks with 20 ml of liquid media consisting of WPM (Woody Plant Medium) salts (Lloyd & McCown, 1980) containing 10 mgL-1 L-cysteine, 100 mgL-1 hydrolyzed casein, 200 mgL-1 malt extract, 250 mgL-1 citric acid, 1 mgL-1 IBA (indole butyric acid), 1.5 mgL-1 2,4-D (2, 4-dichlorophenoxyacetic acid), 2 mgL-1 iP ((N6 (D2-isopentenyl) adenine ) and 30 gL-1 sucrose with continuous agitation at 100 rpm under a 12 h photoperiod (30 μmol m-2 s-1) at 25 ± 2 ºC. After 60 days, somatic embryos were transferred to the Recipient for Automated Temporary Immersion System (RITA®) and cultured under the same conditions in 200 mL of liquid media with a 1 min immersion period every 12 h.
Then after 30 days, for plantlet conversion, somatic embryos were transferred to 20 mL of the maturation medium described by Maruyama (2009) and incubated at 25 ± 2 ºC under a 12 h photoperiod (30 μmol m-2 s-1). All media were adjusted to pH 5.6, before autoclaving for 21 min at 121 ºC.
Determination of total phenolic content: phenolic compounds were quantified following the method described by Bobo-García et al. (2015). Twenty μL of the extracts, standard solutions of gallic acid (10 - 200 μgmL-1 in distilled water) and distilled water were placed in quadruplicate in the wells of a flat-bottom 96-well polystyrene microplate (NUNC, Denmark). Subsequently, 100 μl of diluted Folin-Ciocalteu reagent (1:4) were added to each of the wells, followed by the addition of 75 μl of 100 gL-1 carbonate. Plates were maintained at room temperature (25 ± 1 °C) in darkness for 2 h. Plates were then shaken for 60 s in normal mode and the absorbance of each well was read at 750 nm. Results were reported as gallic acid equivalents per g of fresh plant material (μg GAE g-1).
Determination of total flavonoid content: flavonoid content was quantified by the method described by Magalhães, Almeida, Barreiros, Reis, & Segundo (2012) with some modifications. Fifty μl of the extracts, standard solutions of quercetin (20 - 60 μgmL-1 in methanol) and methanol were placed in quadruplicate in a 96-well flat-bottom polystyrene microplate. Using a multi-channel micropipette, 50 μl of sodium nitrite 6 gL-1 were added to each well and the plate was maintained at room temperature (25 ± 1 °C) for 5 min. Subsequently, 50 μL aluminum chloride hexahydrate (22 gL-1) were added to each of the wells using the first automatic dispenser of the microplate reader. The plate was again allowed to stand for 6 minutes and then 50 μl of 0.8 M sodium hydroxide were added to each well using the second dispenser of the equipment. The plate was shaken for 30 s and the absorbance of the contents of the wells was measured immediately at 510 nm. The procedure was carried out at room temperature (25 ± 1 °C). Results were reported as anhydrous quercetin equivalents per g of fresh plant material (μg AQE g-1).
Quantification of total protein, resistant and soluble starch, sucrose, glucose and fructose: these analyses were done using 50 mg of EC and NEC of mahogany. Protein content was analyzed by the combustion method using the rapid N exceed protein analyzer (Elementar) and arginine acid (99.4 % purity, Sigma Aldrich) as standards. Soluble and resistant starch contents were quantified using the commercial kits K-TSTA and K-RSTAR following the manufacturer’s instructions. All measurements were conducted in duplicate.
For sugar determination, 50 mg samples of mahogany EC and NEC were combined with 800 μl of grade 1 water (conductivity < 100 μS / cm at 25 °C). Samples were vortexed for 40 s, incubated at 60 °C and centrifuged. The supernatant was filtered through a regenerated cellulose membrane with a pore size of 0.2 μm and collected in an HPLC vial coupled with a 250 μl insert. Samples were analyzed using an Agilent 1260 Infinity HPLC equipped with an autosampler (G7129A), quaternary pump (G1311B), column furnace (G1316A), refractive index detector (RID) (G1362 A) and Agilent OpenLab CDS processing and integration system (EZChrom edition). Sucrose, glucose and fructose (99.0 % purity, Sigma Aldrich) were used as standards. All measurements were conducted in duplicate.
Purification and quantification of plant growth regulators: samples of mahogany EC and NEC were shock-frozen in liquid nitrogen, freeze-dried and transported on dry ice from Costa Rica to Germany for hormone analysis. Samples were individually ground in liquid nitrogen and extracted using 50 ml ice-cold 80 % MeOH overnight at 7 °C. The extracts were vacuum filtrated through size 4 glass sintered filters and reduced to a few drops of aqueous extract in a rotary evaporator. The extracts were re-suspended with 4 ml 0.01 M ammonium acetate buffer pH 8.5 in an ultrasonic bath. Then, the samples were frozen overnight at -30 °C in centrifugation tubes, thawed and centrifuged at 60 000 g for 20 min. Samples were purified using a combination of insoluble polyvinylpolypyrrolidone (PVPP; Sigma, Germany), DEAE SephadexTM A-25 (Amersham, Sweden) and SepPak C18 (Waters, Germany) columns according to Jiménez, Guevara, Herrera, and Bangerth (2001). Acidic hormones, IAA, ABA and gibberellins (GAs) were methylated with diazomethane prior to analysis. Based on the method developed by Weiler (1980), hormone analysis was performed by radioimmunoassay (RIA) with triplicate technical repeats using polyclonal antibodies as described in detail by Potchanasin et al. (2009).
Histological analysis: the morphological features of the embryogenic calli and somatic embryos, histological analyses were determined according to the literature (Boissot, Valdez, & Guiderdoni, 1990; Maataoui, Espagnac, & Michaux-Ferriere, 1990; Sandoval, 2005). Macroscopic features were photographed using a stereoscope (NIKON SMZ800 with a Moticam 5) and microphotographs were taken using an OLYMPUS IX51 inverted microscope with an Olympus DP71 camera.
Scanning electron microscopy (SEM): embryogenic calli and somatic embryos were fixed in 2.5 % glutaraldehyde and 2.0 % paraformaldehyde in 0.1 M sodium phosphate buffer pH 7.4 for 24 h at 4º C. Samples were washed three times in the same buffer and post-fixed in 2 % osmium tetroxide solution for 2 h. Fixed tissues were dehydrated in graded acetone solutions for 15 min each. Afterwards, the dehydrated specimens were critical point dried and mounted on aluminum stubs. The mounted specimens were coated with a very thin layer (200 nm) of gold-palladium and examined in a scanning electron microscope (S3700N and S-570, Hitachi) at an accelerating voltage of 15 KV (Goodhew, Humphreys, & Beanland, 2001).
Statistical analysis: The data obtained from biochemical analysis (carbohydrates, total phenols, total flavonoids, protein, and plant growth regulators content) were subjected to one-way analysis of variance (ANOVA) and means were compared using Tukey tests (P = 0.05).
Results
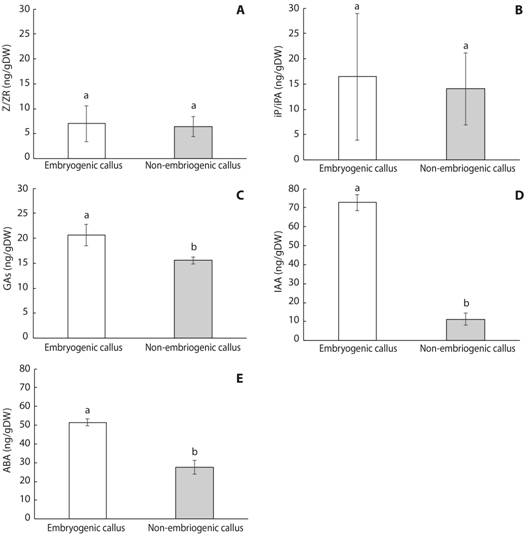
In our study, explants responded by forming three types of callus which varied in color, texture, and friability (Fig. 1). Morphologically, the calli were friable white (type I calli; Fig. 1A), compact cream-colored (type II calli; Fig. 1B), and spongy brown structures (type III calli; Fig. 1C). Friable calli were composed of small isodiametric cells with a conspicuous nucleus and a dense cytoplasm (Fig. 1D). Similarly, in type II calli, isodiametric meristematic dense cells with large distinct nuclei were observed (Fig. 1E). In contrast, type III calli possessed degenerated contracted cells without a distinct nucleus or a dense cytoplasmic region (Fig. 1F).
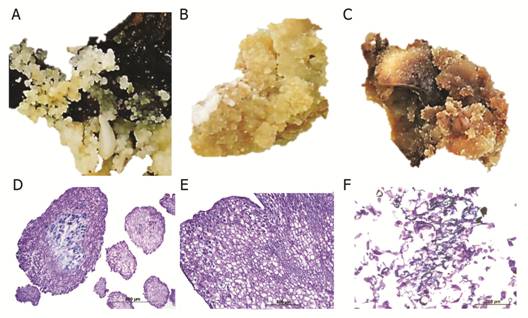
Fig. 1 Three types of callus structures induced from cotyledons of Swietenia macrophylla cultured on differentiation medium. A. Calli with globular structures, Bar: 1 cm. B. Cream-colored compact calli, Bar: 1 cm. C. Spongy calli, Bar: 1 cm. D. Histological section of calli with globular structures (10X). E. Histological section of cream-colored calli with tracheary structures (10X). H. Histological section of spongy calli showing degenerated cells (20X). Bar: 1 cm.
During the initiation phase, swelling of the explants was observed after six weeks of culture in induction medium and type II calli appeared on the surface of the immature cotyledons (Fig. 2A and Fig. 2C). Immature cotyledons of mahogany were composed mostly of parenchymatous cells (Fig. 2B). The sub-epidermal cell layers and cortical parenchyma of the cotyledons were the most embryogenic regions showing the formation of protuberances composed of cells in constant division. Somatic embryo formation was not observed in the proliferation of tissue in the medullar parenchyma of the cotyledons (Fig. 2B). In the differentiation phase, pre-embryogenic structures were observed as protuberances in the callus (Fig. 2D, Fig. 2E, Fig. 2F). In this tissue, cells with prominent nuclei and nucleoli, thick walls and a symmetrical pattern of division were observed (Fig. 2E). During the germination and maturation phases, histological and SEM analyses of somatic embryos embedded in the callus showed no apparent vascular connection with the mother tissue (Fig. 2G, Fig. 2H, Fig. 2I). The protodermis of the somatic embryos was clearly differentiated and was characterized by cells with prominent nuclei and nucleoli and thickened cell walls (Fig. 2H).
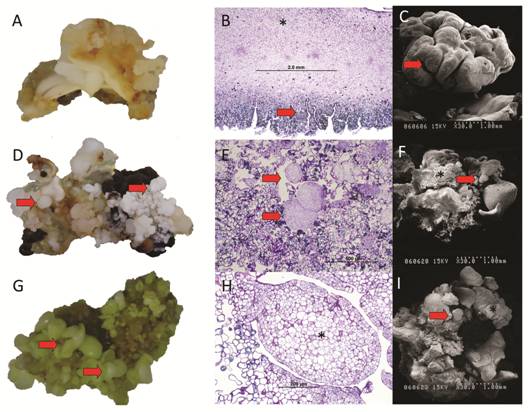
Fig. 2 Macroscopic and microscopic comparison of initial, differentiation and maturation phases of somatic embryogenesis of Swietenia macrophylla. A. Cream-colored compact calli formed after six weeks of culture in induction medium, Bar: 1 cm. B. Parenchyma of immature cotyledons (*) and callus formation (arrow) during the initial phase. C. Callus seen from a Scanning Electron Microscope (SEM). D. Pre-embryogenic structures (arrows) protruding from the callus, Bar: 1 cm. E. Microphotograph of the maturation phase showing pre-embryogenic structures (arrows). F. Maturation phase showing somatic embryos (arrows) protruding from callus (*). G. Differentiation phase showing somatic embryos (arrows), Bar: 1 cm. H. Microphotograph of the maturation phase showing somatic embryos (*). I. Differentiation stage showing embryos (arrow) and callus (*). Bar: 1 cm.
At the differentiation phase, the formation of somatic embryos was asynchronous (Fig. 3A) and plantlets with shoots and roots were obtained after cultured of mature somatic embryos in germination and plantlet conversion medium (Fig. 3B).

Fig. 3 Plant regeneration via SE from cotyledons of Swietenia macrophylla. A. Somatic embryos at different developmental stages. B. Mature somatic embryos with shoots and roots (arrows). Bar: 1 cm.
Globular-shaped somatic embryos were bright and lighter in color than callus (Fig. 4A). They were characterized by cells with a prominent nucleus, dense cytoplasm and connected to parent explants through a suspensor-like structure (Fig. 4B). Scanning electron micrographs revealed globular structures attached to the original callus (Fig. 4C). Heart-shaped embryos showed a typical notch on the tip (Fig. 4D and Fig. 4E) and a notch at the structure surface (Fig. 4F). Heart stage embryos elongated and developed into torpedo stage somatic embryos (Fig. 4G) with a well-defined procambium (Fig. 4H) and they developed into torpedo somatic embryos (Fig. 4I). Finally, the torpedo stage somatic embryos formed small plantlets with distinct cotyledon leaves, apical shoots and root meristems (Fig. 4J). The initiation of cotyledon primordia coincided with the bifurcation of the procambium into cotyledons at the shoot axis (Fig. 4K) and cotyledonary-shaped somatic embryos (Fig. 4L) were observed. In all developmental stages observed, a distinct protodermis surrounded the somatic embryos with no visible vascular connection to the parent explant.
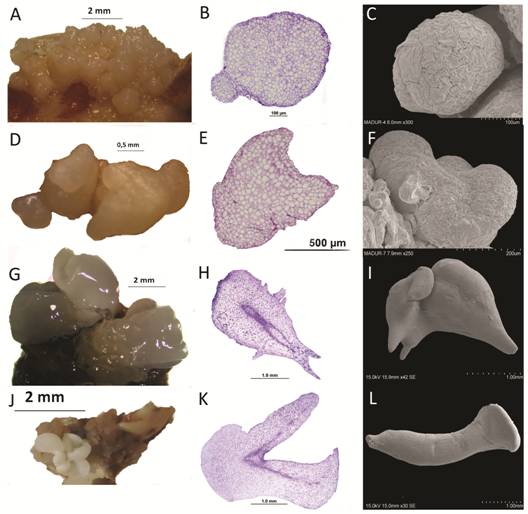
Fig. 4 Developmental stages of somatic embryos of Swietenia macrophylla. A. Globular shaped somatic embryos (arrows) forming on the surface of embryogenic callus. B. Cross section of a globular somatic embryo with a suspensor-like structure (*). C. Scanning electron microscopy (SEM) image of globular embryo. D. Heart-shaped somatic embryo (arrow). E. Cross section of a heart-shaped embryo. F. SEM image of heart-shaped embryo. G. Torpedo-shaped somatic embryos (arrow). H. Cross section of a torpedo-shaped somatic embryo showing its initial procambium (*). I. SEM image of torpedo-shaped somatic embryo. J. Cotyledonary somatic embryo (arrow). K. Cross section of a cotyledonary embryo with its initial procambium (*). L. SEM image of cotyledonary somatic embryo.
Histochemical analysis with PAS-H staining revealed numerous starch grains in the parenchyma of the globular somatic embryos (Fig. 5A and Fig. 5B).
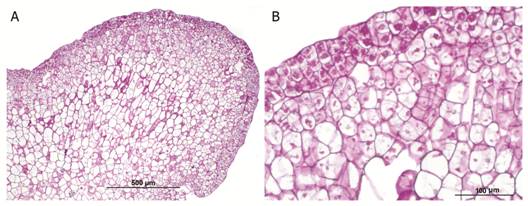
Fig. 5 Starch grains in the parenchyma of globular somatic embryo of Swietenia macrophylla. A. Microphotograph at 100X. B. Microphotograph at 400X showing starch grains (arrows).
Total phenol and total flavonoid contents were significantly higher in NEC than EC (Fig. 6A). The soluble protein content in EC did not differ significantly from that of NEC (Fig. 6B), although the concentration was slightly higher in EC. In our experiment, fructose and glucose were not present at detectable levels in EC or NEC of mahogany (data not shown). Moreover, soluble starch (742.0 ± 657.4 mg/100 g) and sucrose (0.40 ± 0.02 g/100 g) were only detectable in EC. On the other hand, the content of resistant starch was higher in NEC than EC (Fig. 6C).
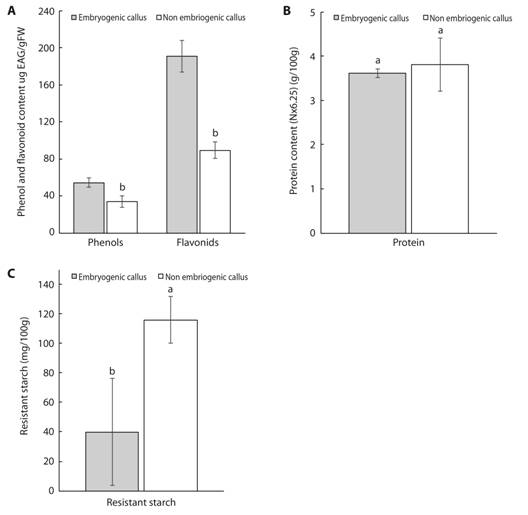
Fig. 6 Concentration of total phenols and flavonoids (A.), protein (B.), resistant starch (C.) in embryogenic and non-embryogenic calli of mahogany. Data are presented as mean ± standard deviation. Means followed by different letters are significantly different at P ≤ 0.05 according to the Tukey test.
Regarding plant growth regulators, similar concentrations of zeatin/zeatin-riboside (Z/ZR) (Fig. 7A), N6-(Δ2-isopentenyl) adenine and N6-(Δ2-isopentenyl) adenosine (iP/iPA) (Fig. 7B) were detected in mahogany EC and NEC. In contrast, EC contained significantly higher concentrations of GAs (Gibberellins 1+3+20) (Fig. 7C), IAA (Fig. 7D) and ABA (Fig. 7E) compared to NEC.
Discussion
In this study, SE was induced using cotyledons of immature zygotic embryos of mahogany following the protocol reported by Collado et al. (2010). Other previously reported SE protocols for Meliaceae used zygotic embryos as starting material (Cameron, 2010; Peña-Ramírez et al., 2011). Moreover, regarding the different callus types obtained in our study, similar observations of the cellular structures have been reported in mangosteen (Garcinia mangostana L., Clusiaceae) (Maadon, Rohani, Iismail, Baharum, & Normah, 2016) and Spanish cedar (Cedrela odorata L., Meliaceae) (Cameron, 2010; Peña-Ramírez et al., 2011). Histological and SEM analyses of somatic embryogenesis in the Meliaceae family have been reported in Cedrela fissilis Vell. (Vila, González, Rey, & Mroginski, 2009) and Cedrela odorata (Cameron, 2010; Peña-Ramírez et al., 2011).
The consumption of seed reserve compounds such as proteins, lipids, and carbohydrates has been correlated with the onset of organogenesis and somatic embryogenesis (Branca Torelli, Fermi, Altamura, & Bassi, 1994; Cangahuala-Inocente, Steiner, Santos, & Guerra, 2004). The beginning of the in vitro developmental process is characterized by high levels of polysaccharides (Branca et al., 1994). These findings suggest that starch is rapidly metabolized in embryogenic tissues to provide energy for intense metabolic and mitotic activity (Martin et al., 2000). In our study, histochemical analysis revealed starch grains in the parenchyma of the globular somatic embryos. Similarly, Silva et al. (2015) identified starch grains in developing somatic embryos of Passiflora edulis Sims (Passifloraceae). Our results differ from those of Rodríguez & Wetzstein (1998) and Cangahuala-Inocente et al. (2004) who reported the absence of starch grains in globular and heart-shaped embryos in Carya illinoinensis (Wangenh.) K. Koch (Juglandaceae) and Acca sellowiana (O. Berg) Burret.
Previously, phenolic compounds were shown to be inhibitory to in vitro regeneration (Nieves et al., 2003; Nascimento-Gavioli et al., 2017). In this sense, our results showed that NEC had higher total phenol and total flavonoid content than EC. Similar results have been reported in sugarcane (Nieves et al., 2003) and peach palm (Bactris gasipaes Kunth, Arecaceae) (Nascimento-Gavioli et al., 2017).
Proteins are primary metabolites that play an important role in somatic embryogenesis induction and the development of somatic embryos (Jiménez, 2001; Cangahuala-Inocente, Steiner, Maldonado, & Guerra, 2009). According to our results, no difference between the soluble protein content in EC and NEC was observed. On the contrary, significantly higher levels of soluble proteins have been reported in EC of sugarcane (Nieves et al., 2003), Acca sellowiana (Cangahuala-Inocente et al., 2009) and Cordyline australis Hook. f. (Asteliaceae) (Warchoł, Skrzypek, Kusibab, & Dubert, 2015). As suggested by Warchoł et al. (2015), active protein synthesis during embryogenic development might be important for the differentiation of somatic embryos.
During cell growth, carbohydrates are important compounds that provide a carbon source and regulate medium osmolarity during embryogenic cell induction, proliferation and development (Maadon, Rohani, Iismail, Baharum, & Normah, 2016). Maadon et al. (2016) reported significant accumulation of fructose, glucose, and sucrose in non-embryogenic structures of mangosteen (Garcinia mangostana) originating from seed and leaf culture. In contrast, according to our results, fructose and glucose were not present at detectable levels in EC or NEC of mahogany and soluble starch and sucrose were only detectable in EC. On the other hand, the NEC had a higher concentration of resistant starch compared to EC. As suggested by Cangahuala-Inocente et al. (2009), somatic embryogenesis requires high amounts of energy and the catabolism of starches results in glycolic compounds that provide the necessary ATP to the cell. Thus, the high turnover of soluble carbohydrates may limit their accumulation.
Endogenous hormones play an important role in regulating SE under in vitro conditions; they function as signaling molecules for the reprogramming of gene expression that allows the acquisition of embryogenic competence and explant differentiation (Jiménez, 2001; Grzyb, Kalandyk, Waligórski, & Mikuła, 2017). In our study, the concentration of Z/ZR and iP/iPA was similar in EC and NEC of mahogany. Similarly, for these cytokinins, Jiménez & Bangerth (2001c) reported no major differences between EC and NEC in the wheat cultivar “Combi”.
In contrast, EC of mahogany contained higher concentrations of GAs, IAA and ABA compared to NEC. Our results are in agreement with comparisons of EC and NEC of carrot (Jiménez & Bangerth, 2001a), maize (Jiménez & Bangerth, 2001b) and peach palm (Nascimento-Gavioli et al., 2017). In this sense, ABA and cytokinins have been shown to help regulate sugar metabolism and transport (Grzyb et al., 2017). On the other hand, the establishment of an auxin gradient during the induction phase is essential for initiating bilateral symmetry during embryogenesis in somatic and zygotic embryos in dicotyledonous and monocotyledonous species (Jiménez, 2005).
In conclusion, histological and biochemical differences between embryogenic and non-embryogenic cells of mahogany were shown. The identification of histological and metabolic differences during the induction of somatic embryogenesis could lead to improved regulation of this process and optimization of in vitro protocols for somatic embryogenesis through modifications of culture media with substances that mimic the inductive condition (Jiménez, 2001). This study opens the opportunity for future analysis of the expression of genes involved in SE, protein content and methylation patterns during this process, as well as genetic engineering of this crop.












 uBio
uBio