Pollination services are provided by certain organisms such as bees, butterflies, moths, flies, beetles, wasps, birds, among others. In this paper, we focus on bees, because they are the most important pollinators at the ecological and commercial level, by pollinating 35 % of flowering crops worldwide (Klein et al., 2007). Therefore, floral resources are important for pollinator’s communities, where the abundance of bees is positively associated with the richness of flower species (Potts, Vulliami, Dafni, Ne’eman, & Wilmer, 2003). Bees also have a preference for those plants that provide a better reward, which could result in a greater amount of pollen, nectar, or both (Roubik, 1989). Derived from this some bees produce honey, which presents a variation related to composition, bee species, geographic region, botanic and storage condition (Karabagias, Badeka, Kontakos, Karabournioti, & Kontominas, 2014). Honey is mainly composed of fructose and glucose, enzymes, proteins, organic acids, minerals, pollen and wax (Sant’Ana, Sousa, Salqueiro, Lorenzon, & Castro, 2012; Manzanares, García, Galdon, Rodríguez, & Romero, 2014). This sweet natural substance is not only produced by honeybees, in the tropics, where stingless bees produce it and is called “pot honey”, which is appreciated and used as sweetener, and as a treatment for various types of infections (Vit, Medina, & Enríquez, 2004; Guerrini et al., 2009). Furthermore of physicochemical properties and botanical origin, sensorial studies play an important role to determine consumer acceptance; some authors report that stingless bee honey have different attributes, such as; bitter taste, salty, sweet, and astringent, with citrus scent, floral, fermented, viscosity and a wide variety of colors, registered in honeys from Geotrigona leucogastra, Melipona grandis and Scaptotrigona sp in Ecuador, Tetragonula carbonaria in Australia, and Melipona fasciculata in Brazil (Ferreira, Aires, Barreira & Estevinho, 2009; Batista de Sousa et al., 2016; Vit, Pedro, Vergara, & Deliza, 2017). On the other hand, palynological studies allow us to determine botanical and geographic origin of honey in order to control its quality, as well as to recognize the species used for pollen and therefore nectariferous flora of an area (Piedras & Quiroz, 2007). Nonetheless, there is not enough information to characterize honey, or to establish international standards to promote its trade, and use in pharmaceutical area. There is a need to contribute to its knowledge in all the places where the stingless bees are located, in order to understand their foraging behavior. Thus, this study aimed to characterize stingless bee honeys from the Soconusco region by the following; a) physicochemically analysis, b) acceptance tests and c) palynological origin, in order to learn and contribute to their knowledge.
Materials and methods
Honey samples: Stingless bee honeys were collected from February to March 2016 at the “Asociación de Meliponicultores del Soconusco S.C. del R. L.” located in municipalities of the Soconusco region; Tapachula, Tuxtla-chico and Cacahoatan, South of Mexico. Predominant vegetation is mainly coffe, rambutan, cocoa, marañon, African palm, mango, sesame, tobacco, banana, soybean, citrics and other endemic plants (Instituto Nacional de Estadística y Geografía, 2015). We gathered nine honey samples of the following species: Melipona solani from Trinidad (Msta), San Jeronimo (Mssjb), Izapa (MsIc), Scaptotrigona mexicana from Cacahoatán (Smca), Francisco y Madero (Smfmb), Izapa I (Smic), Izapa II (Smid), M. beecheii from two different places in Tapachula I (Mbta) and Tapachula II (Mbtb). All honeys were collected from three rational hives in each study in order to obtain a mixed sample of the meliponary (100 ml in total). First pots were opened aseptically with a 20 ml sterile syringes, then stingless bee honeys were kept in sterile labelled flasks and stored in the fridge at -4 °C until analysis.
Physicochemical analysis: Phisycochemical parameters were determined by triplicate using AOAC (2001) methods, which included moisture, pH, free acidity, electrical conductivity (EC), water activity (aw), color, reducing sugars, hidroxymethylfurfural (HMF) and diastase activity.
Moisture content was determined by refractometry (AOAC method 919.38) using an ATAGO digital refractometer at 20 °C. pH and free acidity: pH was measured according to AOAC (method 962.19, 2006) with an OHAUS pHmeter. Free acidity was determined by the titrimetric method; ten g of honey was dissolved in 75 ml distilled water, and this solution was titrated with 0.05 M NaOH solution until the pH reached 8.5. Ten ml of 0.05 M NaOH was added immediately, and back-titrated with 0.05 M HCl solution until the pH reached 8.3, to determine the acidity. Electrical conductivity (EC): Electrical conductivity was determined according to Bogdanov et al. (2004), and it was measured in a 20 % (w/v) solution of stingless bee honey in distilled water using a digital conductometer.Water activity (a w ): Water activity is a gravimetric determination after oven drying at 25 ºC, by using an automatic hygrometer (THERMOCONSTANTER NOVASINA T4-2).
Color: According to Pfund scale, color was determined by absorbance measurement at 635 nm (Montenegro, Avallone, Crazov, & Aztarbe, 2005), directly on diluted honey samples.
Reducing sugars: Reducing sugars and apparent sucrose were determined by potentiometric titration using the Fehling’s test (Lane and Eyon modified method).
Hydroxymethylfurfural (HMF): It was determined by the AOAC method (958.09) where the absorbance of a filtered honey solution, after clarification with carrez solutions and addition of p-toluidine and barbituric acid solutions, is measured at 284 and 336 nm against a blank in the maximum color intensity (3-4 minutes after the barbituric acid addition) (Bogdanov et al., 2002).
Diastase activity: It was measured photometrically, which was expressed in DN (diastase number), and corresponded to g starch hydrolysed/100 g honey / h, at pH 5.2 and T = 40 °C, in Schade units (one diastase unit is the enzyme activity of 1 g of honey, which can hydrolyse 0.0 I g of starch in one hour at 40 °C) (Vit & Pulcini, 1996).
Acceptance test: This test was performed with a panel of 80 untrained judges; students from “Instituto de Biociencias” between 18-22 years old. They were provided with 1 ml of honey in small transparent containers. This test was performed during 9 d (a tasting per d) from 8:00 - 9:00; once they tasted the honey, they were asked to express the acceptance level by a facial hedonistic scale format with 7 categories, where 1= I really like and 7= really don`t like (Singh-Ackbaralil & Maharaj, 2014).
Palynological analysis: Stingless bee honeys were qualitatively and quantitatively pollen analyzed, by using the acetolysis technique (Louveaux, Maurizio, & Vorwohl 1978). The sediment was fixed in glycerin gelatin on slides (three slides per sample). The pollen grains were observed under the 100X target along with an image analyzer. For pollen identification we used reference manuals (Palacios, Ludlow, & Villanueva, 1991; Roubik & Moreno, 1991; Martínez et al., 1993). Pollen grains were classified according to Louveaux et al. (1978) as predominant (> 45 % of the grains counted in a sample), secondary (16-45 %), important minor pollen (3-15 %) and minor pollen (≤ 3 %).
Statistical analysis: An analysis of variance (ANOVA) was used to determine physicochemical differences between the stingless bee honey, followed by a principal component analysis (PCA) to explain the association with the physico-chemical parameters using InfoStat software (Di Rienzo et al., 2015). For the acceptance test, a non-parametric analysis of variance was performed using the KruskallWallis test with the INFOSTAT software (Di Rienzo et al., 2015). In the palynological analysis the samples of honey were analyzed with Shannon-Wiener and Pielou indexes by using PAST software (Hammer, Harper, & Ryan, 2001).
Results
Physical-chemical analysis: We observed differences in stingless bee honeys from different species and origin (Table 1). pH values ranged between 2.8-4.8, which showed significant differences (P < 0.05). In contrast, the electrical conductivity (EC) values varied between 114-1211 μS/cm, free acidity oscillated in the range of 24-100 meq/kg. The aw varied between 59-71 g/100 g, and showed significant differences (P < 0.05). The moisture content was in the range of 35-38 %. With regard to color, which is the first sensory property perceived by consumers and that could influence acceptance and / or rejection, honey samples registered variation from 0.13-0.75 Pfund scale, where honeys varied from dark white-amber color, showing significant differences between the samples (P < 0.05). The reducing sugars showed values of 47-71 %, with significant differences, in this study the HMF content in fresh honeys is low. The samples in the work showed HMF values below 2.72 mg/100 g, the samples MsTa, SmCa and MbTb, showed absence of this compound. On the other hand, diastase activity is also used as marker for the freshness of honeys because its amount decreases in old or heated honeys. The results show ranges of (1.92-11.22 DN), the samples did not present significant differences (P > 0.05), thus showing the presence of the enzyme in honey samples.
Table 1 Physicochemical parameters of honey simples from the Soconusco regios in Chipas.
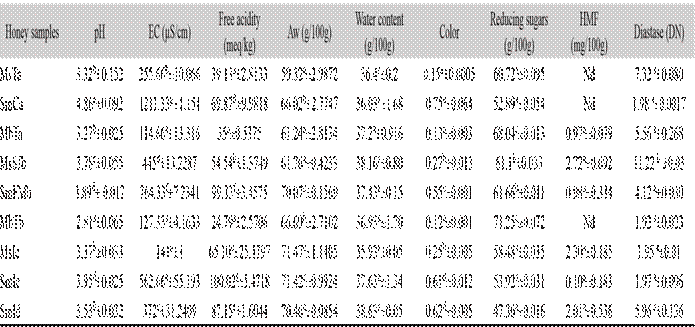
EC = Electrical conductivity; aw = Water activity; HMF = Hydroxymethylfurfural; Nd = Not detected. Different letters showed significant differences (p> 0.05).
MsTa=M. solani site: Trinidad, SmCa=S. mexicana site: Cacahoatan, MbTa=M. beecheii site: Tapachula, MsSJb=S. mexicana site: Francisco I Madero, MbTb=M. beecheii site: Tapachula, SmLc=S. mexicana site: Izapa, MsIc=M. solani site: Izapa, SmId=S. mexicana site: Izapa. Chiapas.
Acceptance analysis: The acceptance results are shown in Fig. 1, where seven of the evaluated honeys were accepted by consumers in categories 2 (I like a lot) 3 (I lighthly like). In addition, these honeys had greater acceptability for the panelists. In contrast the SmCa sample registered the category 5 (I lightly dislike) with a 41 %, followed by the SmFMb simple with the category 4 /nor like nor dislike), which reported a bitter taste.
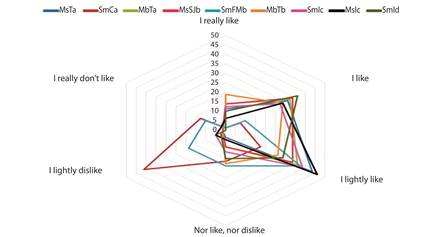
Fig. 1 Preference test for stingless bee honeys; MsTa=M. solani site: Trinidad, SmCa=S. mexicana site: Cacahoatan, MbTa=M. beecheii site: Tapachula, MsSJb=M. solani site: San Jeronimo, SmFMb=S. mexicana site: Francisco I Madero, MbTb=M. beecheii site: Tapachula, SmIc=S. mexicana site: Izapa, MsIc=M. solani site: Izapa, SmId=S. mexicana site: Izapa. The categories range from 1 (I really like) to 6 (I really don`t like).
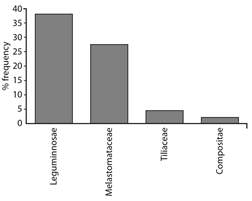
Palynological analysis: Our results showed differences in diversity and foraging uniformity; the diversity index (H) showed values of 1.10-2.36 with a uniformity distribution of species in each sample, meanwhile the Pielou index (0.40-0.75) explained that when the dominance of the species is higher over the others, the value will tend more strongly toward zero, indicating an oligolectic behavior from bees. On the other hand, the Jaccard index showed the similarity between each sample of honey in terms of the presence / absence of botanical species, where four groups are observed, among them the first and last, since there is a dissimilarity between these honey samples (MsTa, MbTb and MbTa). The frequencies of the pollen grains present in the honeys are shown in Fig. 2 and the different views of the main pollen families in Fig. 3. Honey samples of M. beecheii recorded a percentage higher than 45 % of pollen grains corresponding to the Fabaceae family, showing monofloral honey. In relation to the honeys of S. mexicana the secondary pollen spectrum of the samples corresponded to the families Melastomataceae, Malvaceae, Asteraceae and Fabaceae in 25, 28, 5, 15 and 18 %; 12 % 0.66 % and 0.33 % frequency, these samples being poly or multifloral honeys.
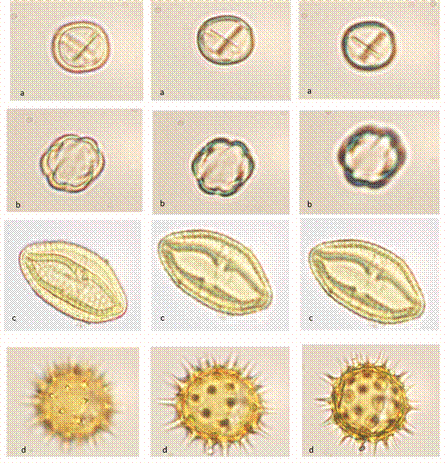
Fig. 3 Different views of the pollen grains observed in stingless bee honeys from Soconusco, Chiapas. A) Fabaceae, B) Melastomataceae, C) Malvaceae, D) Asteraceae.
Quality indicators: The principal component analysis (PCA) represents 86.9 % of the total variability between the species with the parameters analyzed (Fig. 4), the MbTa, MbTb and MsTa honeys are negatively correlated with the reducing sugars variable, which was higher (> 60 %), the MsIc sample was related to HMF being in the same way one of the higher values of this compound (2.30 mg/100 g), in the MsSJb sample the diastase activity registered the highest enzymatic activity, the SmCa honey is closely related to the pH and EC variables; the samples SmId, SmIc and SmFMb have a positive correlation with the variables HMF, aw, humidity, free acidity and color, with higher moisture, more acidic, with water activity superior to samples of Meliponas, and were the darkest ones. In addition to the correlations between the samples and the variables there is also a clear separation of the genera Melipona and Scaptotrigona.
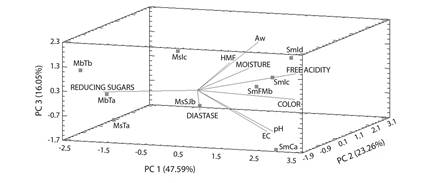
Fig. 4 Principal Component Analysis of the physicochemical properties of stingless bee honeys analyzed
Based on the results obtained in the PCA, the quality indicators were obtained, which showed the ranges of each parameter that these species must present in the Soconusco area of Chiapas, where honeys can be grouped as follow, in Scaptotrigona honeys humidity values are higher than 30 %, aw and free acidity is higher than 60 g/100 g, pH between 2.8 and 4.8, relatively low HMF less than 2 mg/100g, dark amber honeys. Melipona honeys showed higher sugar ranges (> 60 %) and lower enzymatic activity.
Discussion
The parameters of pH agreed with the parameters proposed by the International Honey Commission (IHC) for Melipona and Scaptotrigona genera (Vit et al., 2004). In contrast, our EC values are above the required by the Codex Alimentarius (800 μS/cm); some studies reported EC values between 300-670 μS/cm (Batista de Sousa et al., 2016). However, in some honeys from Brazil, Biluca, Braghini, Gonzaga, Costa, & Fett (2016) have registered different values; 1 340 μS/cm for Tetragonisca angustula, 1 010 μS/cm for Tetragona clavipens and 830 μS/cm for M. quadriasciata (Biluca et al., 2016). These differences could be related to bee species, floral sources visited, due to there are similar results reported when floral source is similar (Almeida-Muradian et al., 2013). On the other hand, when comparing our free acidity results with previous studies in the genera: Homotrigone, Lepidotrigone, Lisotrigone, Tetragonilla, Tetragonula and Tetrigona from Thailand, we can observe that our honeys agreed with the values previously reported 44-121 meq/kg (Chuttong, Chanbang, Sringarm, & Burgett, 2016), similar to the registered for stingless bees from South America with 139 meq/kg (Biluca et al., 2016). Higher values in fresh honeys could be related to organic acid from nectar or the floral source. The results of aw compared with other studies are higher than those reported by Batista de Sousa et al. (2016) 23.9-28.9 g/100 g. The moisture content of these honeys is higher than the proposed by Vit et al. (2004) (maximum of 30.0 %), some studies on honey of Plebeia sp., presented moisture values from 30.26 %, (Dardón & Enríquez, 2008). With regard to color, considered as the first sensory property perceived by consumers, could influence the acceptance and / or rejection of the product. Studies performed with stingless bee honeys have reported color from light, amber and brown (Finola, Lasagno, & Marioli, 2007; Batista de Sousa et al., 2016), which agreed with our results. On the other hand, the reducing sugars values are not only within the ranges proposed by the IHC for these same genera (Vit et al., 2004), but also correspond to the values observed in other studies (Andrade et al., 1999; Rodriguez, Ferrer, Ferrer, & 2004; Kucuk et al., 2007). The HMF content in higher concentration is observed in old honeys and in honey that has been heated, stored in non-adequate conditions, or adulterated with invert sugar or syrup. The samples MsTa, SmCa and MbTb in the study showed absence of this compound which contrasted with the limit established by Vit et al. (2004), which suggested 40 mg/kg, as well as the absent of this value in other studies (Vit, 2008; Habib, Al Meqbali, Kamal, Souka, & Ibrahim, 2014). Batista de Sousa et al. (2016) have reported the absent of HMF in monofloral honeys from the genus Melipona in different regions from Brazil, and Biluca et al. (2016) have registered HMF values lower than 0.31 mg/L (Biluca et al., 2016). The diastase activity is also used as marker for the freshness of honeys because its amount decreases in old or heated honeys. According to the Honey Quality and International Regulatory Standards, from the International Honey Commission, the diastase activity must not be less than or equal to 8, expressed as diastase number (DN), but the Codex Alimentarius (FAO, 2001) has established the minimum diastase activity value of 3, for honeys with natural low enzyme content. The low content of Diastase was only present in four samples of honey, but the Codex Alimentarius mentions that HMF in honeys with low content of this enzyme is not higher than 15 mg/kg, therefore the content of HMF in these were very low therefore these quality honeys are considered. The values of the parameters analyzed in this study vary due to different factors such as botanical origin, geographical origin, climatic conditions, bee species, storage, date of harvest, as well as the presence of organic acids, minerals, proteins, polyols, gluconic acid, some antioxidants, such as carotenoids and flavonoids (Terrab, Dıez, & Heredia, 2003; Acquarone, Buera, & Elizalde, 2007; Baltrusaityte, Venskutonis, & Ceksteryte, 2007; Silvano, Varela, Palacio, Ruffunengo, & Yamul, 2014).
On the other hand, our results agreed with the reported by Deliza & Vit (2013), who found that S. mexicana honeys showed fewer acceptances due to bitter taste. Other unusual descriptors considered as unpleasant are sour/ acid/moldy/alcoholic/urine/rape/wine, gave the lowest acceptance values, and have been reported in M. scutelaris honeys (Ferreira et al., 2009). In a similar way to the physicochemical characteristics, the preference of honey is related to bee species, botanical origin, maturation time and storage conditions (Sousa et al., 2016). Sensorial characteristics are also related to physico-chemical properties (Habib et al., 2014) and in our study we observed that honeys well accepted (MbTb) registered the highest reducing sugar value, similar to the observed by Ferrerira et al. (2009). It has also observed that monofloral honeys showed a better acceptance and registered higher reducing sugar values (Sousa et al., 2016). Thus, the most important aspects that influence the preference or acceptance of consumers and therefore the commercialization of honey is the botanical and geographical origin (Estevinho, Feás, Seijas, & Vázquez-Tato, 2012).
Based on our results, we observed similarity with Roubik (1996), who registered pollen dominance of Leguminosae family in Melipona honeys; in this sense, Almeida-Muradian et al. (2013) in M. subnitida honeys, predominant pollen of Alternanthera sp and Mimosa verrucosa, belonging the last one to the Fabaceae family. In relation to S. mexicana honeys, Ramírez-Arriaga and Martínez-Hernández (2007) indicated that S. mexicana has very particular preferences for Melastomataceae and Malvaceae also found in this study, as well as Asteraceae and Piperaceae.
Once we compared our results with Vit, Persano, Marano, & Salas de Mejías (1998) results, we agreed, by founding a separation within Trigonas and Meliponas. The PCA showed that the Scaptotrigona honeys are positively correlated with the variables aw, humidity, HMF, free acidity, color, pH and CE and the reducing sugars with the Melipona samples, however we could infer that the most important parameters to analyze and establish quality indicates are humidity, HMF and diastase activity. The acceptance test showed that all the honeys were prefered by the consumers except for the SmCa sample. Based on the palynological analysis M. beecheii honeys are monofloral and those of M. solani and the S. mexicana honey are honey multifloral. These studies at the physico-chemical, sensorial and palynological level could help us to preserve native bees, besides we can implicate researchers with producers, so they could have access to stingless bee honey analysis. On the other hand palynological studies will help producers to improve meliponaries management, in order to identify and introduce stingless bee flora.












 uBio
uBio