Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.59 n.4 San José Dec. 2011
Floristic composition and similarity of 15 hectares in Central Amazon, Brazil
Kátia Emidio da Silva1, Sebastião Venancio Martins2, Carlos Antonio Alvares Soares Ribeiro2, Nerilson Terra Santos3, Celso Paulo de Azevedo4, Francisca Dionizia de Almeida Matos5 & Ieda Leão do Amaral5
2. Federal University of Viçosa, UFV, Forestry Engineering Department, Av. Ph Rolfs s/n, post code 36570-000, Viçosa, Minas Gerais, Brasil; venancio@ufv.br, cribeiro@ufv.br
3. Federal University of Viçosa, UFV, Department of Statistics, Av. Ph Rolfs s/n, post code 36570-000,Viçosa, Minas Gerais, Brazil; nsantos@ufv.br
4. Embrapa Amazonia Ocidental, rodovia AM010, C.P.319, post code: 69010-970, Manaus, Amazonas, Brazil; celso.azevedo@cpaa.embrapa.br
5. Instituto Nacional de Pesquisas da Amazonia, INPA/CPBO, av. Andre Araújo, Aleixo, post code 69060-001, Manaus, Amazonas, Brasil; fmatos@inpa.gov.br, iamaral@inpa.gov.br
Dirección de correspondencia
Abstract
The Amazon region is one of the most diverse areas in the world. Research on high tropical forest diversity brings up relevant contributions to understand the mechanisms that result and support such diversity. In the present study we describe the species composition and diversity of 15 one-ha plots in the Amazonian terra firme dense forest in Brazil, and compare the floristic similarity of these plots with other nine one-ha plots. The 15 plots studied were randomly selected from permanent plots at the Embrapa Experimental site, Amazonas State in 2005. The diversity was analysed by using species richness and Shannon’s index, and by applying the Sorensen’s index for similarity and unweighted pair-group average (UPGMA) as clustering method. Mantel test was performed to study whether the differences in species composition between sites could be explained by the geographic distance between them. Overall, we identified 8 771 individuals, 264 species and 51 plant families. Most of the species were concentrated in few families and few had large number of individuals. Families presenting the highest species richness were Fabaceae (Faboideae: 22spp., Mimosoideae: 22spp.), Sapotaceae: 22spp., Lecythidaceae: 15 and Lauraceae: 13. Burseraceae had the largest number of individuals with 11.8% of the total. The ten most abundant species were: Protium hebetatum (1 037 individuals), Eschweilera coriacea (471), Licania oblongifolia (310), Pouteria minima (293), Ocotea cernua (258), Scleronema micranthum (197), Eschweilera collina (176), Licania apelata (172), Naucleopsis caloneura (170) and Psidium araca (152), which represented 36.5% of all individuals. Approximately 49% of species had up to ten individuals and 13% appeared only once in all sampled plots, showing a large occurrence of rare species. Our study area is on a forest presenting a high tree species diversity with Shannon’s diversity index of 4.49. The dendrogram showed two groups of plots with low similarity between them (less than 0.25), and the closer the plots were one to another, more similar in species composition (Mantel R=0.3627, p<0.01). The 15 plots in our study area share more than 50% of their species composition and represent the group of plots that have the shortest distance between each other. Overall, our results highlight the high local and regional heterogeneity of environments in terra firme forests, and the high occurrence of rare species, which should be considered in management and conservation programs in the Amazon rainforest, in order to maintain its structure on the long run. Rev. Biol. Trop. 59 (4): 1927-1938. Epub 2011 December 01.
Key words: forest composition, Central Amazonian, forest ecology, floristic similarity, diversity, terra firme, upland dense forest.
Resumen
Palabras clave: composición boscosa, Amazonía Central, ecología forestal, similitud florística, diversidad, terra firme, bosque denso de tierras altas.
Brazil holds approximately one third of the remaining world’s tropical forests being one of the most important biodiversity areas (PAS 2008), and with the most complex natural environments on Earth. In the Amazon region several landscapes are formed by a mosaic of areas with variable occurrence of plant species per environment (Pitman et al. 2001), and where the differences in tree species distribution can be attributed to habitat peculiarities and/or species’ preferences. Under this context, there has been a rising interest among scientists and societies around the world, especially in what concerns biodiversity loss due to deforestation, regional and global climate changes, and to how species and communities behave when under such changes (Houghton et al. 2000).
The Brazilian Amazon region occupies over half of all Brazilian territory and approximately 65% of its vegetation cover is classified as terra firme forests, which is characterized by high tree species diversity, presenting a low number of individuals, high floristic dissimilarity among adjacent plots, with large variation in floristic similarity (10-36%) (Prance et al. 1976, Ferreira & Prance 1998, Lima Filho et al. 2001, ter Steege et al. 2006, Oliveira et al. 2008). Therefore, several environmental conditions and geographical distances among areas at different scales may play an important role in floristic variations across the Amazon region.
The aim of this paper is to describe and analyse the floristic composition and diversity in the large survey of 15 1-ha plots in terra firme tropical rainforest at the Embrapa Experimental site, in central Amazon, Manaus, Amazonas State and to compare their floristic composition with other nine plots spread over the Amazonas State, by analysing the influence of the geographical distance on the floristic similarity between plots across Amazonas State, Brazil.
Materials and methods
Study site: The study area is located in a terra firme dense forest of Central Amazon, at the Embrapa Experimental site, Central Amazon, Manaus, Amazonas state, Brazil, with a total area of 400ha. This was subdivided into one-ha permanent plots, belonging to the forest management project developed in the Amazon region in Brazil. The climate is tropical, type "Am" (Koppen classification), with a mean annual rainfall ranging from 1 355 to 2 839mm. The mean annual temperature ranges from 25.6°C to 27.6°C, with relative humidity from 84 to 90% (RADAM 1978). On a broad scale, soils are fairly homogeneous throughout the stands; heavy-textured dystrophic yellow latosol predominates, covered mainly by dense forest with emergent trees (IBGE 1999), but with a low degree of local variation in topographic and edaphic conditions. The upland areas are plateaus formed by Tertiary sediments that cover the largest portion of the Amazon sedimentary basin, shaped into landforms dissected in extensive interfluvial plateaus and hills (Regis 1993).Field sampling: Fifteen 1ha sized plots were randomly chosen from the area of 400 in order to investigate the diversity, floristic composition and similarity as compared to nine other terra firme forests from the same Amazonas state. The 15 plots measured 100x100m and had their topographic position recorded as top, slope and base (Fig. 1). Nine were completely positioned in the top position according to topography; four presented a gradient in top and slope; another one stands completely in a base position and one was in the slope and base. All trees ≥10cm dbh (diameter at breast height) were tagged and botanically classified to species level, in the survey undertaken in 2005 (Silva 2010). The botanical material was identified (APGII system) by specialists at the herbarium of the Instituto Nacional de Pesquisas da Amazonia, Manaus, by experts and specialized literature (Ribeiro et al. 1999). The names were checked with the W3tropics database from the Missouri Botanical Garden.
Twenty four plots, consisting of 15 from our present study, called "EMB" plus the plotnumber, together with nine plots coming from different surveys in the Amazonas State, located also in terra firme forests, were used for the floristic similarity analysis (Table 1). All plots inventoried were one-ha sized with dbh≥10cm trees. The nine plots species list was obtained from published and unpublished surveys carried out by INPA’s researchers. The coordinates of all plots were defined by their centroid.
Results
Out of 264 species recorded, the ten most abundant were: Protium hebetatum Daly (1 037 individuals), Eschweilera coriacea (DC.) S.A. Mori (471), Licania oblongifolia Standl. (310), Pouteria minima T.D.Penn. (293), Ocotea cernua (Nees) Mez s.l. (258), Scleronema micranthum Ducke (197), Eschweilera collina Eyma (176), Licania apelata (E.Mey.) Fritsch (172), Naucleopsis caloneura (Huber) Ducke (170) and Psidium araca Raddi (152) (Fig. 3). These species represent 36.5% of the total number of individuals, and only Protium hebetatum, Burseraceae had 11.8% of the relative abundance.
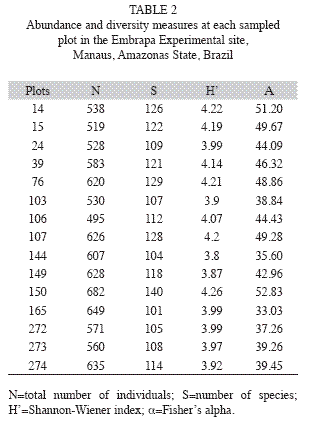
The number of individuals per plot ranged from 495 to 682, with SD equal to 56.4 (Table 2), showing a great variability in the abundance of species measured in all the 15 plots of the study area. Figure 4 shows the species distribution by class of number of individuals within each sampled plot. Thirtyfour species (13%) appeared only once when we summed the abundances over all 15 plots, showing the large occurrence of rare species. Plots 39 and 165 had no occurrence of species with only one individual.
The Shannon-Wiener’s diversity index varied between 3.8 and 4.22 among the 15 plots (Table 2).
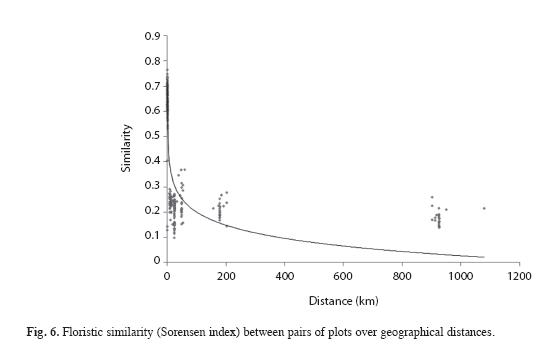
For the 15 EMB plots the distances between pair of plots varied from a minimum of 100m to a maximum of 1 657m (from "EMB39" to "EMB103" plot). The other nine plots had larger distances between pairs, varying from a minimum of 100m to a maximum of 918km (from "JURUA" to "ITAP" plots). The Mantel test, carried out to study the association between differences in species composition and geographic distances among plots, showed positive and significant correlation when all sites were analysed (Fig. 6, R=0.3627, p<0.01). For our 15 EMB plots we also found a significant correlation (R=0.4012, p<0.001). However, the nine plots used to compare with our 15plots, showed no significant correlation between floristic similarity and geographical distances, when analysed alone (R=0.3049, p>0.05).
Discussion
It seems that the environmental heterogeneity and distance played effect in separating plots, which could be observed for the "ZF2B" and "JURUA" plots, once the former plot is located at the base position in the topography and the "JURUA" is the farthest plot located in other watershed, probably having different features when compared to the others. Despite the positive correlation between geographical distance and species composition, the Mantel coefficient was not significant. Through the dendrogram analysis, we could better see the tendency of adjacent plots sharing more species, like the plots ZF2P and ZF2V, which are close to each other (100m apart).
In addition, our study area shows high tree diversity and calls our attention to the large occurrence of low-abundance species across the whole area and to the necessity of considering such rare species in management projects in order to prevent local extinctions. Our results fall in the range of the floristic composition and diversity’s index for Amazon terra firme forests, and the comparison between our 15 plots in the present study and other nine in the Amazonas state, showed that the closer to one another the plots the more similar they are in species composition. Also, we point out that the mosaic of environments in the Amazon terra firme forests, with regional and local variations, coupled with the biological processes, may be responsible for the low similarity observed between some plots compared in this study, showing once more the importance of studies that can keep such variability, helping to understand the mechanisms that bring about and maintain the high tree diversity in tropical rainforests.
Acknowledgments
We thank the Brazilian Agricultural Research Corporation (EMBRAPA) for the financial support, and the Federal University of Viçosa for the opportunity to accomplish the study and the data analysis and the National Council for Scientific and Technological Development (CNPq).
References
Alves, J.C.Z.O. & I.S. Miranda. 2008. Analise da estrutura de comunidades arbóreas de uma floresta amazónica de terra firme aplicada ao manejo florestal. Acta Amaz. 38: 657-666. [ Links ]
Amaral, I.L., F.D.A. Matos & J. Lima. 2000. Composição florística e parâmetros estruturais de um hectare de floresta densa de terra firme no rio Uatumã, Amazonia, Brazil. Acta Amaz. 30: 377-392. [ Links ]
Barrantes, G. & L. Sandoval. 2009. Conceptual and statistical problems associated with the use of diversity indices in ecology. Rev. Biol. Trop. 57: 451-460. [ Links ]
Caiafa, A.N., S.B. Martins, J.A. Nunes & P.V. Eisenlohr. 2009. Espécies arbóreas raras, p. 245-261. In S.V. Martins (ed). Ecologia de florestas tropicais do Brasil. Editora UFV, Universidade Federal de Viçosa, Viçosa, Brasil. [ Links ]
Cientec. 2006. Software Mata Nativa 2: Sistema para Análise Fitossociológica, Elaboração de Inventários e Planos de Manejo de Florestas Nativas. Viçosa, Brasil. [ Links ]
Comita, L.S., R. Condit & S. Hubbell. 2007. Developmental changes in habitat associations of tropical trees. J. Ecol. 95: 482-492. [ Links ]
Coronado, E.N.H., T.R. Baker, O.L. Phillips, N.C.A. Pitman, R.T. Pennington, R.V. Martınez, A. Monteagudo, H. Mogollón, N.D. Cardozo, M. Ríos, R. García-Villacorta, E. Valderrama, M. Ahuite, I. Huamantupa, D.A. Neill, W.F. Laurance, H.E.M. Nascimento, S.S. de Almeida, T.J. Killeen, L. Arroyo, P. Núnez & L.F. Alvarado. 2009. Integrating regional and continental scale comparisons of tree composition in Amazonian terra firme forests. Biog. Discuss. 6: 1421-1451. [ Links ]
Ferreira, L.V. & G.T. Prance. 1998. Species richness and floristic composition in four hectares in the Jaú National Park in upland forests in Central Amazonia. Biod. Cons. 7: 1349-1364. [ Links ]
Formiga, K.M. 2004. Avaliação dos parâmetros estruturais, diversidade e similaridade florística de uma floresta de baixio, na ZF2, reserva do Cuieiras, Amazonas, Brasil. Instituto de Tecnologia do Amazonas/UEA. (Monography), Manaus, Brasil. [ Links ]
Fortin, M.J & M.R.T. Dale. 2005. Spatial analysis: A Guide for Ecologists. Cambridge University, New York, USA. [ Links ]
Gama, J.R.V., A.L. Souza, S.V. Martins & D.R. Souza. 2005. Comparação entre florestas de várzea e de terra firme do Estado do Pará. Rev. Árv. 29: 607-616. [ Links ]
Gentry, A.H. 1988. Changes in plant community diversity and floristic composition on environmental and geographical gradients. Ann. Miss. Bot. Garden 75: 1-34. [ Links ]
Hammer, O., D.A.T. Harper & P.D. Ryan. 2001. PAST: Paleontological Statistics Software Package for Education and Data Analysis. V. 1.92. Palaeontologia Eletronic. [ Links ]
Houghton, R.A., D.L. Skole, C.A. Nobre, J.L. Hackler, K.T. Lawrence & W.H. Chomentowski. 2000. Annual fluxes of carbon from deforestation and regrowth in the Brazilian Amazon. Nature 301-304. [ Links ]
Hubbell, S.P., J.A. Ahumada, R. Condit & R.B. Foster. 2001. Local neighborhood effects on long-term survival of Individual trees in a neotropical forest. Ecol. Res. 16: 859-875. [ Links ]
Instituto Brasileiro de Geografia e Estatística (IBGE).1999. Mapa digital temático de vegetação, Banco de dados Sipam, Brasil. [ Links ]
Knight, D.H. 1975. A phytosociological analysis of species- rich tropical forest on Barro Colorado Island, Panama. Ecol. Monog. 45: 259-28. [ Links ]
Lima Filho, D.A., F.D.A. Matos, I.L. Amaral, J. Revilla, L.S. Coêlho, J.F. Ramos & J.L. Santos. 2001. Inventário florístico de floresta ombrófila densa de terra firme, na região do Rio Urucu-Amazonas, Brasil. Acta Amaz. 31: 565-579. [ Links ]
Magurran, A.E. 1988. Ecological diversity and its measurement. Cambridge University, New York, USA. [ Links ]
Malheiros, A.F., N. Higuchi & J. Santos. 2009. Análise estrutural da floresta tropical úmida do município de Alta Floresta, Mato Grosso, Brasil. Acta Amaz. 39: 539-548. [ Links ]
Martins, S.V., N.R.S. Silva, A.L. Souza & J.A.A. Meira Neto. 2003. Distribuição de espécies arbóreas em um gradiente topográfico de Floresta Estacional Semidecidual em Viçosa, MG. Scie. For. 64: 172-181. [ Links ]
Matos, F.D.A. 2006. Estimativa de biomassa e carbono em floresta ombrófila densa de terra firme na Amazônia Central, Manaus-AM, Brasil, por meio de dados de satélites de média e alta resolução espacial.Tese de Doutorado, Universidade Federal do Paraná, Curitiba- PR, Brasil. [ Links ]
Milliken, W. 1998. Structure and composition of one hectare of central Amazonian terra firme Forest. Biotropica 30: 530-537. [ Links ]
Oliveira, A.A. & S.A. Mori.1999. A central Amazonian terra firme forest. I. High tree species richness on poor soils. Biod. Cons. 8: 1219-1244. [ Links ]
Oliveira, A.N. & I.L. Amaral. 2004. Florística e fitossociologia de uma floresta de vertente na Amazônia Central, Amazonas, Brasil. Acta Amaz. 34: 21-34. [ Links ]
Oliveira, A.N. & I.L. Amaral. 2005. Aspectos florísticos, fitossociológicos e ecológicos de um sub-bosque de terra firme na Amazônia Central, Amazonas, Brasil. Acta Amaz. 35: 1-16. [ Links ]
Oliveira, A.N., I.L. Amaral, M.B.P. Ramos, A.D. Nobre, L.B. Couto & R.M. Sahdor. 2008. Composição e diversidade florístico-estrutural de um hectare de floresta densa de terra firme na Amazonia Central, Amazonas, Brasil. Acta Amaz. 38: 627-642. [ Links ]
Plano Amazônia Sustentável (PAS). 2008. Governo Federal (also available on-line: http://www.mma.gov.br) [ Links ]
Phillips, O.L., P. Hall, A.H. Gentry, S.A. Sawyer & R. Vásquez. 1994. Dynamics and species richness of tropical rain forests. Proc. Nat. Acad. Sci. 91: 2805-2809. [ Links ]
Pinto, S.I.C., S.V. Martins, N.F. Barros, H.C.T. Dias & S.H. Kunz. 2008. Influence of environmental variables on the shrub and tree species distribution in two Semideciduous Forest sites in Viçosa, Minas Gerais, Brazil. Rev. Biol. Trop. 56: 1557-1569. [ Links ]
Pitman, N.C.M., J.W. Terborg, S.R. Silvan, P.V. Nunes, D.A. Neil, C.E. Ceron & W.A. Palacios. 2001. Dominance and distribution of tree species in upper Amazonia terra firme. Ecology 82: 2101-2117. [ Links ]
Poulsen, A.D., H. Tuomisto & H. Balslev. 2006. Edaphic and floristic variation within a 1-ha plot of lowland Amazonian rain forest. Biotropica 38: 468-478. [ Links ]
Prance, G.T., W.A. Rodrigues & M.F. Silva. 1976. Inventário florestal de um hectare de mata de terra firme, km 30 da estrada Manaus-Itacoatiara. Acta Amaz. 6: 9-35. [ Links ]
Quesada, C.A., J. Lloyd, M. Schwarz, T.R. Baker, O.L. Phillips, S. Patiño, C. Czimczic, M.G. Hodnett, R. Herrera, A. Arneth, G. Lloyd, Y. Malhi, N. Dezzeo, F.J. Luizão, A.J.B. Santos, J. Schmerler, L. Arroyo, M. Silveira, N. Priante Filho, E.M. Jimenez, R. Paiva, I. Vieira, D.A. Neill, N. Silva, M.C. Peñuela, A. Monteagudo, R. Vásquez, A. Prieto, A. Rudas, S. Almeida, N. Higuchi, A.T. Lezama, G. López-Gonzalez, J. Peacock, N.M. Fyllas, E. Alvarez Dávilla, T. Erwin, A. di Fiore, K.J. Cao, E. Honorio, T. Kileen, A. Peña Cruz, N. Pitman, P. Nuñez Vargas, R. Salomão, J. Terborgh & H. Ramirez. 2009. Regional and large-scale patterns in Amazon forest structure and function are mediated by variations in soil physical and chemical properties. Biog. Discuss. 6: 3993-4057. [ Links ]
RADAM. 1978. Programa de Integração Nacional. Levantamentos de Recursos Naturais. Manaus. DNPM, Ministério das Minas e Energia. Brasilia-DF, Brazil. [ Links ]
Rankin-de-Merona, J.M. 1987. Estudos populacionais de árvores em florestas fragmentadas e as implicações para a conservação In Situ das mesmas na floresta tropical da Amazonia central. IPEF 35: 47-59. [ Links ]
Regis, W.D.E. 1993. Unidades de Relêvo, p.39-45. In S.S. Caldeiron (ed.). Recursos Naturais e Meio Ambiente: Uma visão do Brasil. Fundação Instituto Brasileiro de Geografia e Estatística-IBGE, Rio de Janeiro, Brasil. [ Links ]
Ribeiro, J.E.L.S., B.W. Nelson, M.F. Silva, L.S.S. Martins & M. Hopkins. 1999. Reserva florestal ducke- diversidade e composição da flora vascular. Acta Amaz. 24: 19-30. [ Links ]
Rosenberg, M.S. 2005. Passage 1 (Pattern Analysis, Spatial Statistics, and Geographic Exegesis). V.1.1. Arizona State University. [ Links ]
Ruokolainen, K., H. Tuomisto, M.J. Maciá, M.A. Higgins & M. Y-Hallas. 2007. Are floristic and edaphic patterns in Amazonian rain forests congruent for 4 trees, pteridophytes and Melastomataceae? J. Trop. Ecol. 23: 13-25. [ Links ]
Silva, K.E. 2010. Florística e estrutura espacial: 15 hectares de parcelas permanentes na floresta densa de terra firme na Amazônia Central. Ph. D. Thesis, Federal University of Viçosa, Viçosa-MG, Brasil. [ Links ]
Sposito, T.C., T.I. Matzer, I.L. Amaral, A.C. Oliveira, M. Horta, D.A. Clark & D.B. Clark. 2007. Dynamic and structure of tropical forest in South and Central América: an across-site analyses of one hectare plots, p.23-30. In The third TEAM network science meeting. Panamá. The tropical ecology assessment and monitoring network. Panamá, Panamá [ Links ].
Tello, J.C.R. 1995. Aspectos fitossociológicos das comunidades vegetais de uma toposseqüência da Reserva Florestal Ducke do INPA. Tese de Doutorado, Instituto Nacional de Pesquisas da Amazônia/Universidade Federal do Amazonas, Manaus, Amazonas, Brasil. [ Links ]
ter Steege, H., D. Sabatier, H. Castellanos, T.V. Andel, J. Duivenvoorden, A.A. Oliveira, R. Ek, R. Lilwah, P. Maas & S. Mori. 2000. An analysis of the floristic composition and diversity of Amazonian forests including those of the Guiana Shield. J. Trop. Ecol. 16: 801-828. [ Links ]
ter Steege, H., N.C.A. Pitman, O.L. Phillips, J. Chave, D. Sabatier, A. Duque, J.F. Molino, M.F. Prévost, R. Spichiger, H. Castellanos, P.V. Hildebrand & R. Vásquez. 2006. Continental-scale patterns of canopy tree composition and function across Amazonia. Nature 443: 444-447. [ Links ]
Torres, R.B., F.R. Martins & L.S. Kinoshita. 1997. Climate, soil and tree flora relationships in forests in the state of São Paulo, southeastern Brazil. Rev. Bras. Bot. 20: 41-49. [ Links ]
Tuomisto, H. & K. Ruokolainen. 1997. The role of ecological knowledge in explaining biogeography and biodiversity in Amazonia. Biodivers. Conserv. 6: 347-357. [ Links ]
Tuomisto, H. & K. Ruokolainen. 2005. Environmental heterogeneity and the diversity of pteridophytes and Melastomataceae in Western Amazonia. Biol. Skr. 55: 37-56. [ Links ]
Sebastião Venancio Martins & Carlos Antonio Alvares Soares Ribeiro. Federal University of Viçosa, UFV, Forestry Engineering Department, Av. Ph Rolfs s/n, post code 36570-000, Viçosa, Minas Gerais, Brasil; venancio@ufv.br, cribeiro@ufv.br
Nerilson Terra Santos. Federal University of Viçosa, UFV, Department of Statistics, Av. Ph Rolfs s/n, post code 36570-000,Viçosa, Minas Gerais, Brazil; nsantos@ufv.br Celso Paulo de Azevedo. Embrapa Amazonia Ocidental, rodovia AM010, C.P.319, post code: 69010-970, Manaus, Amazonas, Brazil; celso.azevedo@cpaa.embrapa.br
Francisca Dionizia de Almeida Matos & Ieda Leão do Amaral. Instituto Nacional de Pesquisas da Amazonia, INPA/CPBO, av. Andre Araújo, Aleixo, post code 69060-001, Manaus, Amazonas, Brasil; fmatos@inpa.gov.br, iamaral@inpa.gov.br
Received 10-VIII-2010. Corrected 10-II-2011. Accepted 15-III-2011.