Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.59 n.4 San José Dec. 2011
Dinora Acosta Zamorano1, Gorgonio Ruiz Campos1, Asunción Andreu Soler1,2 & Claudia Alejandra Reyes Valdez1
1. Laboratorio de Vertebrados, Facultad de Ciencias, Universidad Autónoma de Baja California, km. 106 Carr. Tijuana- Ensenada, 22800, Ensenada, Baja California, México; U.S. mailing address: PMB 064, P.O. Box 189003, Coronado, California, USA; dinora002@hotmail.com, gruiz@uabc.edu.mx, reyesc@uabc.edu.m2. Departamento de Ecología e Hidrología, Facultad de Biología, Universidad de Murcia, Campus de Espinardo 30100, Murcia, España; asun@um.es
Dirección de correspondencia
Abstract
Fundulus lima inhabits river drainage systems and is threatened after the introduction of cichlids in the area. To support conservation programs, the spatial and temporal variation of the diet composition of this endangered killifish, was determined in two oasis systems of Baja California Sur, Mexico (San Ignacio and La Purisima river drainages), during rainy and dry seasons. F. lima was captured by using passive and active capture techniques. A total of 192 stomach contents of F. lima was analyzed. The contribution of each prey item in the diet composition was quantified by means of the indices of occurrence frequency (% OF), numerical (%N) and volume (%V) percentages. The relative importance of each prey item was determined according to the percentage of the Relative Importance Index (%RII). The similarity of the diet was calculated between hydrological basins (populations combined by basin), seasons (rainy versus dry months), sexes and size classes, by using Schoener’s resource overlap index. We used two ecological indices to determine the type of feeding strategy exhibited by the fish: (1) niche breadth of Levins and (2) proportional similarity of Feisinger. Sand was the most abundant item in the stomach content of killifishes from both drainages (39% and 47%, respectively). Diet composition was similar for both drainages (74%) as well as among their respective size classes; however, it was different between sexes. In both drainages, F. lima predated mainly on diatom algae, dipterous and trichopteran larvae, and fish scales during the dry season; while it preferred dipterous larvae, filamentous algae and ostracods in the rainy season. A feeding strategy of opportunist type was exhibited by F. lima during the rainy season, changing to specialist type during the dry season. This information will be the basis for future investigations related to the conservation of this endangered species and its habitat. Rev. Biol. Trop. 59 (4): 1669-1678. Epub 2011 December 01.
Key words: Fundulus lima, diet, feeding strategy, oasis systems, Baja California Sur.
Resumen
Fundulus lima habita sistemas de aguas continentales, y se encuentra amenazado tras la introducción de cíclidos en el área de estudio. Para poder llevar a cabo programas de conservación, fue determinada la dieta de la sardinilla peninsular en peligro de extinción, en dos sistemas de oasis de Baja California Sur, México (cuencas de los rios San Ignacio y La Purísima), durante las épocas secas y de lluvias. Una gran proporción de arena fue encontrada en el contenido estomacal de este pez para ambas cuencas (39% y 47%, respectivamente). La composición de la dieta fue similar entre ambas cuencas (74%), como también dentro de sus respectivas clases de tallas; sin embargo, fue diferente entre sexos. Durante la época de secas, F. lima consumió principalmente diatomeas, larvas de dípteros y tricópteros, además de escamas de peces; mientras que en la época de lluvias tuvo preferencia por las larvas de dípteros, algas filamentosas y ostrácodos. Este pez exhibe una estrategia alimentaria de tipo oportunista durante la época de lluvias, la cual cambia a tipo especialista en la época de secas. Esta información será la base para futuras investigaciones relacionadas con la conservación de esta especie, en peligro de extinción, y su hábitat.
Palabras clave: Fundulus lima, alimentación, estrategia alimenticia, oasis, Baja California Sur
The Baja California killifish, Fundulus lima (Vaillant 1894) is one of the two killifishes with distribution in the Eastern Pacific drainage (Parenti 1981), and one of the three freshwater fishes endemic to the Baja California peninsula (Ruiz-Campos 2000, Ruiz-Campos et al. 2002). In the last decade, F. lima has been declining in abundance in more than 75% of its original distribution range due to competitive interactions with the exotic cichlid Tilapia sp. cf. zillii (Ruiz-Campos et al. 2002, 2006). Currently, F. lima is categorized as endangered (Jelks et al. 2008) on the basis of monitoring from 1998 to 2004 through its distribution range (Ruiz-Campos et al. 2006).
Fundulus lima inhabits the oases of the Pacific drainage of the Baja California peninsula, from the San Ignacio to Las Pocitas river basins (Ruiz-Campos et al. 2002), with extirpated populations in San Javier, San Luis and San Pedro de La Presa basins (Ruiz-Campos et al. 2008). These oases were formed during the radical ecological transformation that suffered the central Baja California from a wet region to dry one after the late Pleistocene (Axelrod 1948). These oases, like the islands, are ecosystems very fragile to disturbed by anthropogenic activities, therefore the holistic knowledge of their biota and habitat are critical to develop future programs of conservation.
In spite of, that the diet composition of F. lima was previously studied in the typical locality of San Ignacio oasis (Alaníz-García et al. 2004), a comparison of the diet composition among sites of the same drainage, between different drainages or contrasting conditions (rainy and dry seasons), had not been performed. In this study, we analyzed the spatial and temporal variation of the diet composition of F. lima in the San Ignacio and La Purísima drainages, embracing rainy and dry seasons. This information will be the basis for future investigations related to the conservation of this endangered species and its habitat.
The two studied drainages (Fig. 1) are part of the Priority Hydrological Regions of San Ignacio and La Purísima, both belonging to the Desert El Vizcaino floristic province (Arriaga- Cabrera et al. 2000). The San Ignacio river (RSI) is located South of the Sierra San Francisco mountain range, heads at a spring on the Bavisuri Plain and intermittently flows westward through the small towns of Santa Lucía, Cueva Colorada, Piñuela, and Guamuchil before reaching San Ignacio oasis (Fig. 1). There, a dam that also serves as a bridge to enter the town from Mexican Highway 1 impounds it. The river then flows intermittently southwestward, passing the ranches Los Estribos, Los Corralitos and San Sabas, previous to completely disappearing in a wide sandy plain about 20km Northeast of the large hypersaline coastal lagoon of San Ignacio (Ruiz-Campos et al. 2006). The fluvial valley is bordered by basaltic terraces and hills of sedimentary rocks. General climate is extremely arid with sparse winter rains (<100mm), with mean annual temperature ranging from 18 to 24°C (Arriaga- Cabrera et al. 2000).
Another major regional drainage, La Purísima river (RLP, Fig. 1), originates on the Western slope of the Sierra La Giganta mountain range and is the largest perennial stream in the State of Baja California Sur. It flows above ground for nearly 32km, through the villages of La Mochila, La Pintada, Huerta Vieja, Carambuche, San Isidro, La Purísima, El Saucito, Los Corrales and San Gregorio, before entering the coastal San Gregorio estuary (Ruiz-Campos et al. 2006). Similarly, general climate is arid with winter rains delivering average annual precipitation up to 200mm (Arriaga-Cabrera et al. 2000).
In the RSI, the salinity increases from upstream to downstream, exhibiting a gradient from 0.1 to 4.3ppt. In the RLP, salinity also increases from upstream to downstream, but with lesser range of variation (0.1 to 1.0ppt) (Ruiz-Campos et al. 2008). The riparian vegetation of the RSI is represented by native Mexican Fan Palm (Washingtonia robusta), Southern Cattail (Typha domingensis), Spiny Rush (Juncus acutus), Vinorama (Acacia brandegeana), Western Honey Mesquite (Prosopis glandulosa), and exotic Date Palm (Phoenix dactylifera), Giant Reed (Arundo donax), and Tamarisk (Tamarix sp.) (Arriaga-Cabrera et al. 1997, Ruiz-Campos, unpublish. data). Along the RLP, dominant riparian plants are date palm, common reed or "carrizo" (Phragmites australis), Southern Cattail, Mule fat or "batamote" (Baccharis salicifolia), vinorama, western honey mesquite, Bonpland willow (Salix bonplandiana), and the conspicuous exotic woody vine or "bejuco" (Cryptostegia grandiflora). Aquatic vegetation in both basins is represented by the genera Ceratophyllum, Enteromorpha, Chara and Potamogeton.
Material and methods
Samples were taken during rainy (January and March 2004) and dry (February and September 2003, and July 2004), seasons at six stations through the main river course of the San Ignacio (Poza Larga, Los Corralitos and San Sabas) and La Purísima (Ojo de Agua, Presa Carambuche and El Pilón) drainages (Fig. 1). F. lima was captured by using passive and active capture techniques. Passive capture devices consisted of one experimental gill net composed of 2 panels 4.5m long x 1.80m high, with bar meshes of 1.3 and 3.8cm, respectively. Deployment times for the gill net ranged from 12 to 22h per sampling event (mean=17hr). The active capture devices were a cast net (4m diameter with 2.54cm bar mesh) and a minnow seine (7.8m long x 1.9m high, with 3.5mm bar mesh). For the analysis of stomach contents of F. lima, a sample of 15 individuals was obtained by sampling event for each site. The individuals were fixed in formalin 10% (buffered with sodium borate) solution. Each fish was measured [standard length (SL) to nearest 0.01mm], and then dissected to remove the stomach. Three classes of size were determined on the basis of size frequencies: class I (<40mm SL), II (40-60mm SL) and II (>60mm SL). The stomach contents were fixed in formalin (10%) and later analyzed under a stereoscope.
Prey availability: The abundance of prey in the water column in each site was quantified using Wildco plankton net (diameter 22cm and mesh size 153μ), that was towed along a 40m-transect. The samples were fixed in formalin (5%) solution (buffered with sodium borate) to be transported to the laboratory for analysis. Each sample was weighed (to nearest 0.001g) and the contribution of each item prey was estimated visually as the percentage (%) that occupies in a Petri disc (Sánchez-Gonzáles et al. 2001).
For quantifying the availability of benthic prey in each site, a sample of 1kg of sediment was taken using a core sampler. Each sample was preserved on ice for transportation to the laboratory. The samples were washed and sieved to obtain the present preys, which were separated into major food items, counted, and weighed on an analytical scale to 0.001g precision. Since small prey tend to float in a test tube, the volume of each prey item in the stomach content was quantified under a dissecting microscope from the area occupied by each prey item on a grid of transparency film consisting of 100 cells placed under a Petri dish (Sánchez-Gonzáles et al. 2001).
Diet composition: The relative importance of each prey item was determined according to the percentage of the Relative Importance Index (%RII; Pinkas et al. 1971): RII=(%N+%V)x% OF; where %N=(number of prey i)/(number of total prey)x100; %V=(number of cells occupied by the prey i) ⁄ (number of cells occupied by all the prey taxa in each individual stomach x100) and %OF=(number of stomachs with prey i ⁄ number of stomachs with content analyzed) x100. This index combines the absolute numerical values, frequency, and the gravimetric index, providing a comparable value for the different aspects. For comparative purposes, the absolute value of RII of each prey taxa was expressed in percentage of the total summa of RII for all prey taxa.
The similarity of the diet was calculated between hydrological basins (populations combined by basin), seasons (rainy versus dry months), sexes and size classes, by using Schoener’s resource overlap Index (Schoener 1970): α=[1-0.5(Σ|Pxj-Pyj|)]x100, where Pxj=proportion of the prey taxon j (%RII) in the diet of the group x (basin, season, sex or size class x) and Pyj=proportion of the same prey taxon j in the diet of the group y (basin, season, sex or size class y). The overlap was considered significant for values ≥60% (0.60; Zaret & Rand 1971). Finally, the Pearson’s parametric correlation (r) between the average size of consumed prey and the size of the fish was also calculated (Sokal & Rohlf 1981).
Trophic niche breadth: We used two ecological indices to determine the type of feeding strategy exhibited by the fish in the RSI: (1) niche breadth of Levins (1968): NB=[1/ (RxΣPj2)], where Pj=proportion of the prey taxon j (%RII) in the diet of the fish, and R=number of prey resources (types) available in the environment; and (2) proportional similarity of Feisinger (Feisinger et al. 1981): PS=1-[0.5Σ|pj–qj|], where, Pj=proportion of the prey taxon j in the diet, and qj=proportion of that same prey taxon in the environment (%N). The criterion to assign the type of feeding strategy was based on Rachlin et al. (1989). Statistical analyses were performed with Statistica 5.0 software package (StatSoft, Inc., Tulsa, OK, 1995) and accepted at a significance level (p<0.05).
Results
Prey availability: In the RSI, 19 major prey taxa were identified for combined samples of water column and bottom. Sand represented 39% of the volume of these samples followed by dipterous larvae (18%) and filamentous green algae (15%) (Fig. 2). In the RLP, had a less variety of prey (n=10), but with the same dominance of sand (47%), followed by dipterous larvae (26%) and filamentous green algae (22%) (Fig. 2). The presence of dipterous larvae and filamentous green algae was evident during the dry months, while invertebrates as trichopterans, plecopterans, copepods and ostracods occurred during the rainy months.
Diet Composition: According to the Relative Importance Index (RII), the stomach contents of F. lima (n=125) in the San Ignacio river showed high proportions of sand (47%) and in lesser proportions dipterous larvae (19%) and filamentous algae (17%). In the RLP, the killifishes (n=67) also had a similar consumption pattern for sand (41%) and dipterous larvae (21%), as well as trichopteran larvae (18%). The diet similarity of F. lima between RSI and RLP basins was significant (α=74%). During the dry months the most important prey items in the stomach contents were as follows: in February 2003 the presence of sand (58%) was predominant, followed by diatom algae (14%) and trichopteran larvae (11%); in September 2003, again the dominium of sand (70%) was notable, with secondary importance of filamentous green algae (15%) and fish scales (13%). In July 2004, sand (35%) and dipterous larvae (26%) were the most consumed food items. During the rainy season (January 2004), a high percentage of sand (47%) was found in the stomach contents, seconded by dipterous larvae (26%) and filamentous green algae (22%). In March 2004, the most consumed prey items were dipterous larvae (39%), sand (20%), ostracods (17%) and filamentous green algae (16%); the remaining prey had values of RII below 4% (Table 1).
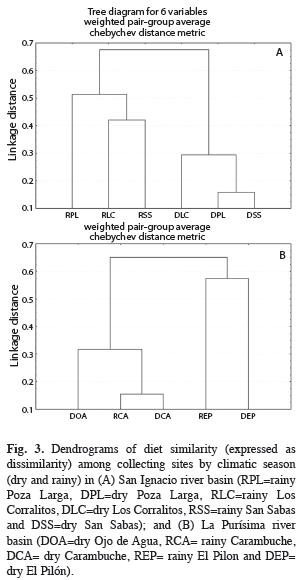
The dendrogram of the diet similarity among sampling sites by climatic season (dry and rainy) in the RSI showed two groups, one formed by the sites sampled during the dry season and other composed by those sampled in the rainy season (Fig. 3). The highest similarities occurred among sites during the dry season (>0.70). In the case of the diet similarity among sampling sites by climatic season in the RLP, the higher values of similarity (≥0.76) were registered among the sites of Carambuche (dry and rainy seasons) and Ojo de Agua (dry season), but not for El Pilón in both climatic seasons (Fig. 3).
Diet composition by size and sex: The standard length for 192 individuals of F. lima examined ranged from 20 to 75mm. In Table 2 are depicted the values of %RII for the prey items found in the three size classes of F. lima by basin. In the RSI, the class I (<40 mm) recorded a high percentage of sand (62%), followed by filamentous green algae (15%). Class II (40-60mm), also exhibited a notable amount of sand (59%), a lesser proportion of filamentous green algae (21%), dipterous larvae (12%), and diatom algae (5%). The diet of the class II (>60mm) included a 54% of sand, 21% of dipterous larvae and 11% of filamentous algae. The diet for females was dominated by dipterous larvae (60%) and ostracods (15%), while males registered high proportions of sand (53%), dipterous larvae (23%) and filamentous green algae (14%) (Table 2). Both sexes had a diet overlap value of 51.9%.
The diet composition of F. lima by size classes in the RLP (Table 2) was as follows: class I (<40mm) consumed mainly dipterous larvae (62%) and nematodes (19%); class II (40-60mm) included in their diet a high proportion of sand (66%) and filamentous green algae (28%); and class II (>60mm) consumed dipterous larvae (39%), filamentous green algae (29%) and sand (23%). Both females and males showed respectively a high consumption of sand (60 and 72%, respectively), filamentous green algae (25 and 13%, respectively) and dipterous larvae (15 and 12%, respectively), habits that are reflected in a high value of diet similarity (74.5%).
The diet similarity among size classes of F. lima from combined basins was significant between the classes I and II (84.2%), classes I and II (80.2%), and classes II and II (82.5%). When comparing a same size class between basins, only between classes II was significant (85.8%). The diet similarity for the same sex between basins, exhibited no significant values (males 18.0%, and females 31.7%, respectively).
Consumed prey size and trophic niche breadth: The overall average prey size (all he prey taxa and basins combined) consumed by F. lima showed a significant correlation with the mouth size (r=0.71, p<0.001). When analyzed by basin separately, only the individuals from RSI showed a significant correlation (r=0.82, p=0.001).
The feeding niche breadth (NB) of F. lima in the RSI was low with values ranging from 0.05 to 0.09, both corresponding to Poza Larga site. The Proportional Similarity Index (PSI) in the diet varied between 0.19 (Los Corralitos) and 0.65 (Poza Larga). During the rainy period (January 2004 and March 2004), F. lima showed an opportunist feeding strategy (low NB and high PSI) consuming the most abundant prey in the environment. In the dry period (February 2003, September 2003 and July 2004) the ish showed a specialist feeding strategy (low NB and low PSI), where the fish predated on certain types of prey independent of their abundances or availability in the environment.
Discusion
During the study period that embraced both dry and rainy seasons, a wide variation in the level of flooding for both rivers was observed. In the dry season, the flows decreased significantly for forming isolated ponds, increasing the proliferation of filamentous algae and the sedimentation. After the rainy months, the flow increased and connected the ponds again, favoring thus the abundance of aquatic invertebrates as trichopterans, plecopterans, and coleopterans. On 22-23 September 2003, the Marty hurricane affected the study area, where the flooding caused alterations in the morphology of the river and in the riparian vegetation (Ruiz-Campos et al. 2006). After the flooding, the washing of the ponds was noted because of the little amount of prey and sand in the benthos.
The diet composition of F. lima in the two basins studied, included as the most important prey to dipterous larvae, filamentous green algae, ostracods, diatom algae and copepods. It is important to note the significant contribution of sand to the stomach contents, representing until a 47% of the total volume, condition that denoted its marked preference by benthic habitats (Alaníz-García et al. 2004). In addition, this fish possess sharp teeth slightly long and flexible to scrape the periphyton of the substratum (Alaniz-García et al. 2004).
Fundulus lima showed a strong preference by consuming dipterous larvae (19%) and filamentous green algae (17%) through the study, but exhibiting a change from iatom algae to ostracods during the dry months. The significant consumption of algal material in F. lima contrasts with that reported for other species of killifishes in the Atlantic drainage such as F. majalis, F. heteroclitus and F. diaphanus, which consume algae in a smaller proportion (Baker-Dittus 1978, Rozas & LaSalle 1990). The diet composition in F. lima is similar than that reported for the omnivorous killifish F. grandis (Simpson & Gunter 1956, Springer & Woodburn 1960, Harrington & Harrington 1961). The high consumption of filamentous green algae has also been reported by Rakes (1989) for F. julisia, in spring conditions. Kneib & Steven (1978) observed for F. heteroclitus that individuals >30mm standard length consume a significant amount of living plant material. It is important to mention, the occasional presence of fish remains (%OF<10%) in the stomach content of F. lima, particularly in adult individuals.
The feeding spectrum of F. lima in the two studied basins did not show significant differences in the type, size and proportion of prey consumed among size classes. In both sexes, all size classes showed preference of consumption by dipterous larvae, filamentous green algae and ostracods. Our results differed from those reported by Alaníz-García et al. (2004) for this same species of killifish, where the diet changed with the length of the fish, and by Vince et al. (1976) for F. heteroclitus, who observed in feeding preference experiments in laboratory that the maximum size of prey eaten increased with the size of the predator. The diet composition of F. lima is very similar in both basins (74.4%), however within the same basin it differed among sites and seasons. Seasonally, the diet of F. lima in the dry months was very different than that for rainy months, which is due to changes in the composition and availability of prey in the environment. The poor correlation found between consumed prey size and length of F. lima might be consequence of a shortage of the availability of prey. After the flooding event on 23 September 2003, the density of prey in the ponds decreased by effect of removing and eventually increased by the colonization events (Ruiz-Campos et al. 2008). Thus F. lima shows a specialist strategy during conditions of high density of prey, changing to one opportunist when the density of prey is low. Both strategies are indicatives of the feeding plasticity of this killifish for exploiting efficiently prey in relation to its availability (Gerking 1994).
Although, this work covers only the feeding spectrum of F. lima in two oasis systems of the Baja California peninsula, it is possible to assume that the introduction of exotic species (Tilapia cf. sp. zillii, Poecilia reticulata, Gambusia affinis or Xiphophorus hellerii, among others), in these habitat has had a negative impact on the native killifish, mainly as resulted from the competition pressure that limits the access to food resources (Ruiz-Campos et al. 2006, 2008,). This kind of stress on native fish species is only one of many that should be considered in achieving conservation targets; however, this has recently emerged as a priority in the planning process and in behavior studies.
Acknowledgments
We would like to thank F. Camarena, J.A. Echánove, R. Guzmán, A. Jullian, G. De León, I.A. Peraza by their valuable help in the fish sampling. Also thanks to late A. Espinoza by logistical support and information of the study area. This study was funded by the Universidad Autónoma de Baja California (projects: DGIP 1275, 173), and the Secretaría de Medio Ambiente y Recursos Naturales (SEMARNAT, project: 173/A-1). The collection permit was provided by the Dirección General de Vida Silvestre de México (SGPA/DGVS-6559). The third author received a postdoctoral fellowship from Programa Seneca 2009 (Fundación Séneca, Agencia Regional de Ciencia y Tecnología, Murcia, España).
References
Alaníz-García, J., G. Ruiz-Campos, F.J. Abarca-González & A. Valdez-González. 2004. Interacción trófica entre dos especies ícticas sintópicas, una nativa (Fundulus lima) y la otra exótica (Xiphophorus helleri), en el oasis San Ignacio, Baja California Sur, México, p. 193-216. In M.L. Lozano-Vilano & A.J. Contreras- Balderas (eds.). Homenaje al Doctor Andrés Reséndez Medina: un ictiólogo mexicano. Universidad Autónoma de Nuevo León, Monterrey, México. [ Links ]
Arriaga-Cabrera, L., S. Díaz, R. Domínguez & J.L. León. 1997. Composición florística y vegetación, p. 69-106. In L. Arriaga & R. Rodríguez Estrella (eds.). Los oasis de la península de Baja California. SIMAC and CIB-NOR, La Paz, Baja California Sur, México. [ Links ]
Arriaga-Cabrera, L., V. Aguilar-Sierra & J. Alcocer- Durand. 2000. Aguas continentales y diversidad biológica de México. Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, México D.F., México. [ Links ]
Axelrod, D.I. 1948. Climate and evolution in western North America during middle Pliocene time. Evolution 2: 127-144. [ Links ]
Baker-Dittus, A.M. 1978. Foraging patterns of three sympatric killifish. Copeia 1978: 383-389. [ Links ]
Feisinger, P., E.E. Spears & R.W. Poole. 1981. A simple measure of niche breadth. Ecology 62: 27-32. [ Links ]
Gerking, S.D. 1994. Feeding ecology of fish. Academic, California, USA. [ Links ]
Harrington, R.W. & E.S. Harrington. 1961. Food selection among fishes invading a high subtropical salt marsh from onset of flooding through the progress of a mosquito brood. Ecology 42: 646-656. [ Links ]
Hyslop, E.J. 1980. Stomach contents analysis-a review of methods and their application. J. Fish Biol. 17: 411-429. [ Links ]
Jelks, H.L., S.J. Walsh, N.M. Burkhead, S. Contreras- Balderas, E. Díaz-Pardo, D.A. Hendrickson, J. Lyons, N.E. Mandrak, F.McCormick, J.S. Nelson, S.P. Platania, B.A. Porter, C.B. Renaud, J.J. Schmitter-Soto, E.B. Taylor & M.L. Warren Jr. 2008. Conservation status of imperiled North American freshwater and diadromous fishes. Fisheries 33: 372-407. [ Links ]
Kneib, R.T. & A.E. Steven. 1978. Growth, reproduction, and feeding of Fundulus heteroclitus (L.) on a North Carolina salt marsh. J. Exp. Mar. Biol. Ecol. 31: 121-140. [ Links ]
Lagler, K.F. 1978. Freshwater Fishery Biology. W.M.C. Brown, Dubuque, Iowa, USA. [ Links ]
Levins, R. 1968. Evolution in changing environments. Princeton University, Princeton, New Jersey, USA. Parenti, L.R. 1981. A phylogenetic and biogeographic analysis of cyprinodontiform fishes (Teleostei: Atherinomorpha). B. Am. Mus. Nat. Hist. 168: 341-557. [ Links ]
Parenti, L.R. 1981. A phylogenetic and biogeographic analysis of cyprinodontiform fishes (Teleostei: Atherinomorpha). B. Am. Mus. Nat. Hist. 168: 341-557. [ Links ]
Pinkas, L., M.S. Oliphant & I.L. Iverson. 1971. Food habitats of albacore, bluefin and bonito in California waters. Fish. Biol. 152: 1-105. [ Links ]
Rachlin, J.W., B.E. Warkentine & A. Papantoniou. 1989. The use of niche breadth and proportional similarity in feeding to stipulate California peninsula, Mexico. Proc. Desert Fish. Coun. 17: 105-117. [ Links ]
Rakes, P.L. 1989. Life history and ecology of the barrens topminnow, Fundulus julisia Willians and Etnier (Pisces, Fundulidae). M.S. Thesis, University of Tennessee, Tennessee, USA. [ Links ]
Rozas, L.P. & M.W. LaSalle. 1990. A comparison of the diets of Gulf Killifish, Fundulus grandis Baird and Girard, entering and leaving a Mississippi brackish marsh. Estuaries 13: 332-336. [ Links ]
Ruiz-Campos, G. 2000. Threatened fishes of the world: Fundulus lima Vaillant, 1894 (Fundulidae). Env. Biol. Fish. 59: 20. [ Links ]
Ruiz-Campos, G., J.L. Castro-Aguirre, S. Contreras-Balderas, M.L. Lozano-Villano, A.F. González-Acosta & S. Sánchez-González. 2002. An annotated distributional checklist of the freshwater fish from Baja California Sur, Mexico. Rev. Fish Biol. Fisher. 12: 143-155. [ Links ]
Ruiz-Campos, G., F. Camarena-Rosales, S. Contreras-Balderas, C.A. Reyes-Valdez, J. De La Cruz-Agüero & E. Torres-Balcazar. 2006. Distribution and abundance of the endangered killifish, Fundulus lima, and its interaction with exotic fishes in oases of Central Baja California, México. Southwest. Nat. 51: 502-509. [ Links ]
Ruiz-Campos, G., F. Camarena-Rosales, S. Contreras- Balderas, G. Bernardi & J. De La Cruz-Agüero. 2008. Evaluación ecológica y distribución de peces exóticos en las regiones hidrológicas de San Ignacio y La Purísima, Baja California Sur, y su impacto en las poblaciones del pez amenazado Fundulus lima. Final Tech. Report. Project: Semarnat-Conacyt- 2002-C01-173. México. [ Links ]
Sánchez-Gonzáles, S., G. Ruiz-Campos & S. Contreras- Balderas. 2001. Feeding ecology and habitat of the threes pine stickleback, Gasterosteus aculeatus microcephalus, in a remnant population of northwestern Baja California, Mexico. Ecol. Freshw. Fish. 10: 191-197. [ Links ]
Schoener, T.W. 1970. Nonsynchronous spatial overlap of lizards in patchy habitats. Ecology 51: 408-418. [ Links ]
Simpson, P.G. & G. Gunter. 1956. Notes on habitats, Saltwater Cyprinodontes. Tul. Stud. Zool. 4: 115-134. [ Links ]
Sokal, R.R. & F.J. Rohlf. 1981. Biometry: the principles and practice of statistics in biological research. W.H. Freeman and Company, New York, USA. [ Links ]
Springer, V.G. & K.D. Woodburn. 1960. An ecological study of the fishes of the Tampa Bay Area. Prof. Pap., Ser. No I, Florida State Board of Conservation, St. Petersburg, USA. [ Links ]
Vince, S., I. Valiela, N. Backus & J.M. Teal. 1976. Predation by the salt marsh killifish Fundulus heteroclitus (L.) in relation to prey size and habitat structure: Consequences for prey distribution and abundance. J. Exp. Mar. Biol. Ecol. 13: 255-266. [ Links ]
Zaret, T.M. & A.S. Rand. 1971. Competition in tropical stream fishes: support for the competitive exclusion principle. Ecology 52: 336-342. [ Links ]
Correspondencia a: Dinora Acosta Zamorano, Gorgonio Ruiz Campos & Claudia Alejandra Reyes Valdez. Laboratorio de Vertebrados, Facultad de Ciencias, Universidad Autónoma de Baja California, km. 106 Carr. Tijuana- Ensenada, 22800, Ensenada, Baja California, México; U.S. mailing address: PMB 064, P.O. Box 189003, Coronado, California, USA; dinora002@hotmail.com, gruiz@uabc.edu.mx, reyesc@uabc.edu.mx
Asunción Andreu Soler. Laboratorio de Vertebrados, Facultad de Ciencias, Universidad Autónoma de Baja California, km. 106 Carr. Tijuana- Ensenada, 22800, Ensenada, Baja California, México; U.S. mailing address: PMB 064, P.O. Box 189003, Coronado, California, USA / Departamento de Ecología e Hidrología, Facultad de Biología, Universidad de Murcia, Campus de Espinardo 30100, Murcia, España; asun@um.es
Received 22-IX-2010. Corrected 02-III-2011. Accepted 05-IV-2011.