Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.58 suppl.3 San José Oct. 2010
Implications of coral harvest and transplantation on reefs in northwestern Dominica
Andrew W. Bruckner1,2 & Eric H. Borneman3
1. Khaled bin Sultan Living Oceans Foundation, Landover, Maryland 20978, USA; bruckner@livingoceansfoundation.org2. NOAA Fisheries Office of Habitat Conservation, NOAA Coral reef Conservation Program, Silver Spring, MD 20910 USA.
3. University of Houston, Houston TX USA.
Abstract
Key words: coral mariculture, fragmentation, restoration, coral health and disease, transplantation.
Resumen
En junio del año 2002 el gobierno de Dominica solicitó asistencia para evaluar el Instituto Oceanográfico de Dominica (OID), una operación de cultivo de corales del océano Atlántico y del Indo Pacifico para propósitos de restauración y comercio. Evaluamos las facilidades de cultivo del OID, la condición de los arrecifes y el impacto potencial de la recolección de corales y los posibles beneficios del transplante de colonias. Los arrecifes de coral (9 arrecifes de profundidades entre 3 y 20m se caracterizaban por 35 especies escleractíneos y una cobertura viva de coral entre 8 y 35%. Las especies que liberan larvas tales como Porites astreoides (14.8 % de todos los corales), P. porites (14.8%), Meandrina meandrites (14.7%) y Agaricia agaricites (9.1%) fueron los más abundantes, pero sus colonias eran pequeñas (promedio de 25cm de diámetro). El complejo de Montastraea annularis fue otro grupo dominante (20.8% de todos los corales) y sus colonias eran mayores (promedio de 70cm de diámetro). Entre todas las especies los corales habían perdido el 20% de sus tejidos, con un promedio de 1.4% por mortandad reciente. Las enfermedades de coral afectaron 6.4% de todas las colonias, con la incidencia mayor en Cabrits Oeste (11%), Bahía Douglas (12.2%) y el arrecife de Coconut Afuera (20.7%). Plaga blanca y la enfermedad de la banda amarilla causaron la mayoría de la pérdida de tejido vivo, especialmente en el complejo de M. annularis, con impactos localizados por caracoles coralívoros, sobre crecimiento por macroalgas, impactos de tormentas y sedimentación. Mientras los arrecifes parecen estar declinando substancialmente, los esfuerzos de restauración de OID no parecen entablar su recuperación debido a que las especies escogidas para transplantar con las mayores tasas de sobrevivencia incluyen especies que liberan larvas (Agaricia y Porites) que fueron abundantes en las restauraciones, al igual que los organismos potencialmente parasíticos (Palythoa y Erythropodium) que pueden monopolizar el sustrato y sobre crecer a los corales. Las especies de mayor valor para la restauración (corales masivos de liberación de gametos) tuvieron baja sobrevivencia, sufrieron mas de las enfermedades u otros impactos biológicos e impactos antropogénicos los cuales deben ser atendidos para garantizar la sobrevivencia de los corales transplantados. Algunos problemas con la operación de cultivo de OID tales como temperaturas e irradiación altas al igual que sobre crecimiento de algas podrían ser controladas con algunos cambios en los sistemas de irrigación, iluminación y mantenimiento. Sin embargo los altos niveles de enfermedades y otros impactos (macroalgas, ausencia de herbívoros, evidencia de sobre pesca, sedimentación y eutrofización) son de mayor preocupación por sus impactos sobre los corales que proveen el armazón del arrecife ya que estas especies reciben menos beneficio de la restauración, su colección puede amenazar la sobrevivencia de sus poblaciones la recuperación de estos corales podría tomar siglosPalabras claves: maricultura de coral, fragmentación, restauración, enfermedad y salud de coral, transplantes.
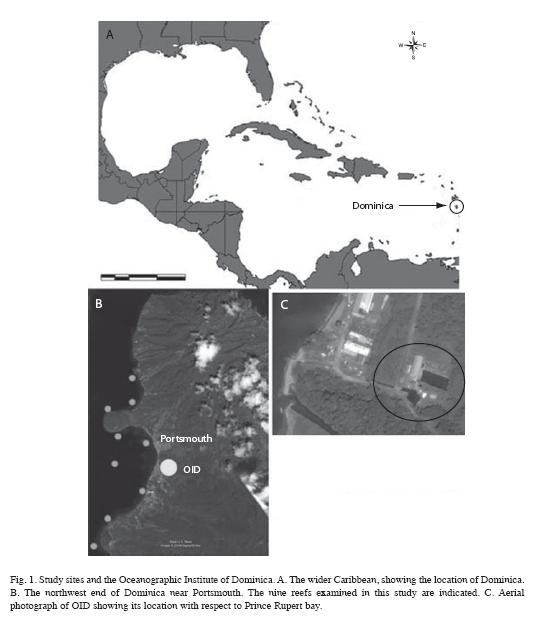
A land-based coral farm was established by Associated Marine Technologies (AMT, later renamed Oceanographic Institute of Dominica) off the northwest coast of Dominica, near Portsmouth in 1998. This farm was interested in growing corals to supply the aquarium trade, and also proposed to restore neighboring reefs using corals collected from local waters and propagated on the farm. The farm was initially established with brood-stock imported from Indonesia, including hundreds of soft and stony corals and colonial anemones. Caribbean species were introduced to the facility after receiving permission to harvest corals from surrounding reefs, with the agreement that 10 corals would be transplanted back to the reef for each colony that was harvested. As of 2002, AMT was primarily growing stony and soft corals in shallow outdoor tanks from small fragments and clippings removed from wild-harvested colonies, using natural seawater piped in from Prince Rupert Bay. These were fragmented and attached with adhesive to small (8cm) cross-shaped discs and exported to the United States for the aquarium trade. Following a shift in management, the owner undertook an extensive review of existing practices and future options for mariculture, including a proposed shift for coral culture from IndoPacific specimens to Caribbean corals. The primary goal of this effort was to grow corals for use in coral reef restoration, with a secondary goal of producing second or third "generation" corals for international trade, to supply aquarium hobbyists in the U.S. and other locations. The owner also wanted to support local communities by employing individuals from Portsmouth and neighboring towns.
Materials and methods
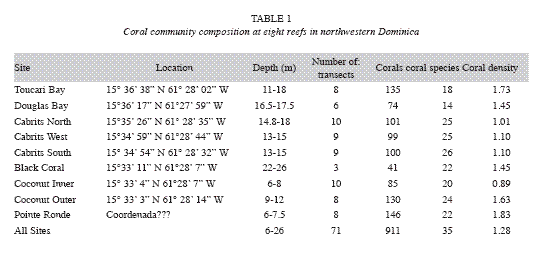
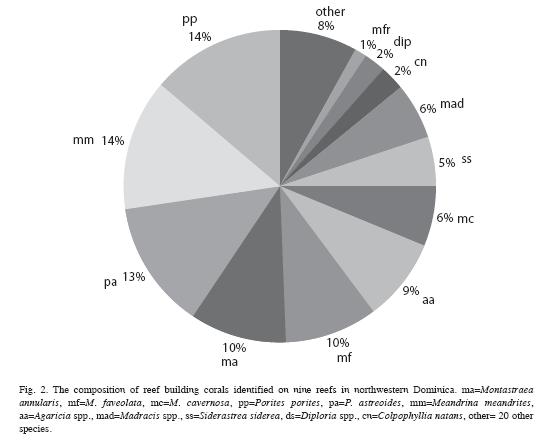
Field surveys: Baseline data on the condition of reef-building corals were obtained for 8 reefs (Toucarie, Douglas Bay, Cabrits North, Cabrits West, Cabrits South, Black Coral Gardens, Coconut Outer, Coconut Inner, Pointe Rounde (Table 1, Fig 1) off the west coast of Dominica using the benthic protocol from the Atlantic and Gulf Rapid Reef Assessment (AGGRA) Version 2 (http://www.coral.noaa. gov/agra/index.html) assessment methodology, with minor modifications. At each site, 10m transects were extended parallel to depth contours, and all corals 5cm in diameter and larger that touched or lay directly below the transect tape were recorded to species, measured (maximum diameter and height, to nearest 1cm). For each coral an estimate of the amount of live tissue, recently killed tissue and condition (s) causing coral mortality was recorded. Live coral cover was determined by calculating the total number of centimeters of live coral tissue located directly under each 10m transect. In addition to corals, other benthic invertebrates including corallimorphs, octocorals, anemones, gorgonians and sponges were counted within 1 m of either side of the line. All stony corals were identified to species. Colonies of Montastraea annularis complex were separated according to Weil and Knowlton (1994) as M. annularis, M. faveolata or M. franksi. Forms or morphotypes of Agaricia agaricites Colpophyllia natans, Meandrina meandrites and Porites porites were combined under the respective species.
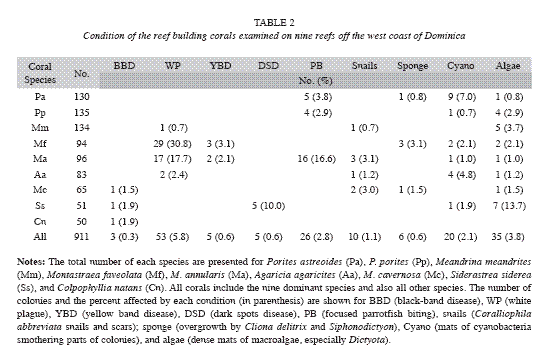
Recent mortality was defined in this study as any tissue loss occurring within approximately the last 30 days, using signs that included: (1) white coral skeleton that lacked algae (surfaces denuded of tissue within the last 5-7 days); (2) skeletal areas with readily recognizable corallites that had not been substantially eroded but were colonized by fine green filamentous algae or (3) white, exposed skeletal surfaces, or eroded skeletal surfaces with fine filamentous algae, that had been physically abraded by fish or other agents but had not yet been colonized by epibionts. Old mortality was defined as areas on a colony that were dead for longer than 30 days and included exposed skeletal surfaces with eroded corallites, and denuded areas on a colony colonized by crustose coralline algae, macroalgae or encrusting invertebrates; in most cases the cause of mortality could not be definitively determined. Causes of recent mortality were identified as disease (separated into black band disease, white plague, yellow band disease, dark-spots disease, or other disease), corallivory [parrotfish bites, damselfish (primarily Stegastes planifrons) algal lawns, fireworm (Hermodice carunculata) or snail (Coralliophila abbreviata) predation], overgrowth by algae or invertebrates (cnidarian, sponge or tunicate), sedimentation or storm damage, based on the key identification features described in Bruckner (2001). In the event that a cause of mortality could not be determined, it was recorded as unknown. The following abbreviations are used throughout this manuscript: BBD (black band disease), WP (white plague), YBD (yellow band disease), DSD (dark spots disease), PB (focused parrotfish biting), snails (Coralliophila abbreviata snails and scars), sponge (overgrowth by Cliona spp. and Siphonodictyon spp.), cyano (mats of cyanobacteria smothering parts of colonies) and algae (dense mats of macroalgae, especially Dictyota and Lobophora).
Assessment of coral husbandry practices: A tour of the facility was undertaken to collect information on the size of the facility,methods of water circulation and decontamination, coral fragmentation, and culture and husbandry practices. OID employees provided demonstrations of the technique used to fragment corals and attach them to bases for grow-out, and efforts to maintain the health of the corals, including removal of algae and corallivores. A survey of the species, numbers of fragments, and condition of the fragments on hand was undertaken, along with general observations on culture facilities including water quality, water flow, exchange between tanks, quarantine practices, and light and temperature levels.
Efficacy of restoration efforts: Two sites with pilot restoration projects were examined. Transect surveys as described above were conducted within these sites to provide an indication of the community composition, structure and condition. Transplanted corals were located and assessed in terms of survival and growth and effects on the surrounding reef.
Results
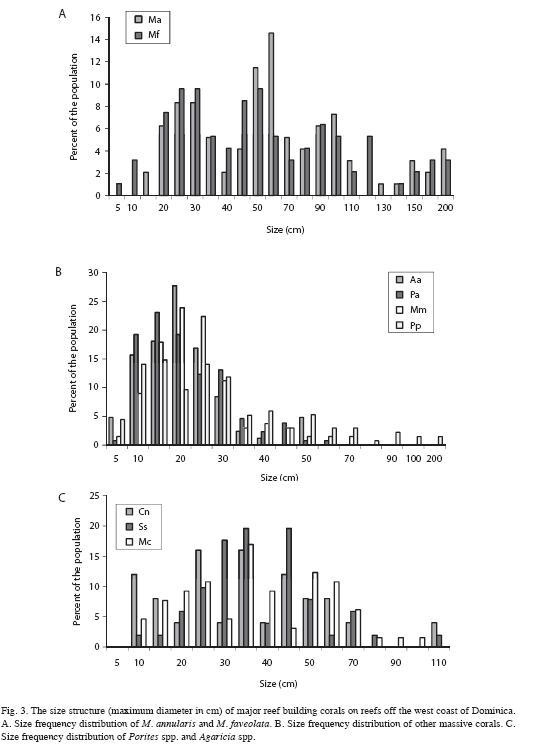
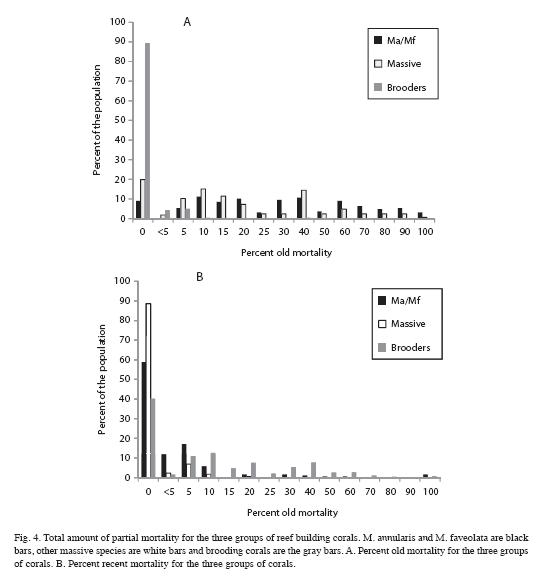
Extent and causes of mortality: Most corals examined on these reefs had experienced partial mortality, except for colonies that were small in size (20cm or less). Mean old mortality for all corals combined was 20.4% compared to 1.4% recent mortality. Recent mortality was attributed to coral diseases (6.4% of all colonies), predation (3.6%), overgrowth by algae (6.1%), invertebrate predation (0.8%), storm damage (0.8%; Hurricane Lenny, 1999) and sedimentation (0.5%). While most colonies of M. annularis (90%) had experienced partial mortality in the past, only 20% of the other massive broadcast spawning corals and 10% of brooding corals showed signs of old mortality. In contrast, a larger proportion (60%) of brooding corals showed signs of recent mortality, compared to about 40% of the M. annularis complex and 10% of other massive corals (Fig. 4). The mean amount of recent mortality (tissue loss) among all corals (n=911) was low (1.4%).
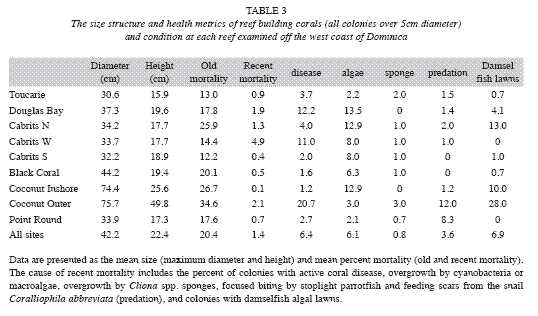
Disease and predation: A number of coral diseases were observed along transects, including white plague, black band disease, Caribbean yellow band disease, and dark spots disease. White plague was observed on all reefs, while other diseases were restricted to specific locations and generally occurred at a low prevalence (<5%). The highest prevalence of disease overall was recorded at Cabrits West (11.0%), Douglas Bay (12.2%) and Coconut Outer (20.7%) (Table 3). In addition, corallivores including coral-eating snails (C. abbreviata), fireworms (H. carunculata) and stoplight parrotfish (Sparisoma viride) were prominent, and in some locations had caused substantial mortality. Coconut Outer was dominated by large (1-5m diameter), very old colonies of M. annularis and M. faveolata, many of which had signs of recent mortality (colonies were missing a mean of 45% of their tissue). White plague affected 19.2% of all corals and 30% (n=23) of the M. annularis (species complex). In addition, 13% of all M. annularis colonies exhibited recent lesions created by Sparisoma viride (focused biting); 26% also had older lesions that were either colonized by damselfish algal lawns or were showing signs of tissue recovery.
Damselfish algal lawns: Stegastes planifrons territories were observed on all reefs except Cabrits West, affecting 6.9% of all corals, but they were of particular concern in three locations, Coconut Inner (10% colonies affected), Coconut Outer (28% colonies affected) and Cabrits North (13%). In these locations, fish established territories among massive and plating corals and had created extensive lesions on corals.
Coral mariculture: The Oceanographic Institute of Dominica (OID) was primarily culturing corals in 2002, along with limited experimentation with other aquaculture products. The facility had a large quarantine tank, a deeper (50 000L) tank and 160 shallow (0.5m deep, 1.5m diameter) plastic circular tanks for coral propagation (Fig. 5). An indoor, shallow flowing-seawater table was used for fragmenting corals and attaching them to a substrate. All outdoor tanks were located under dense shade cloth which reduced ambient light by 50-60%. Sea water was pumped from several hundred meters offshore in Prince Rupert´s Bay from about 30 m depth. The water was pumped to the 50 000L tank, which serves as a settling tank and then into each smaller propagation tank before being circulated through the quarantine tank and returned to the sea. The water was reported to be UV-sterilized and ozonated before discharge, but this sterilization system was not working properly during the site visit. Each individual coral propagation tank was self-contained; water was not exchanged among those tanks.
In June, 2002, the main propagation area contained stony corals and gorgonian (soft) corals, all originally obtained from waters surrounding Dominica, along with a small number of tanks with Pacific soft corals and stony corals. Each tank was subdivided with PVC pipe into six pie-shaped sections, with each section holding 55 coral fragments (330 corals/tank; approx. 52 000 corals on hand) of one to three species of corals. One group of six tanks in a separate location contained Indo-Pacific soft corals, including genera such as Clavularia, Pacific Briareum (formerly Pachyclavularia), Sarcophyton, Xenia, Sinularia, Tubipora, Actinodiscus, Lobophytum, and other species. The facility also had one display tank that contained seven gorgonian species, 3 corallimorph species, 2 zoanthid species, and 21 species of stony corals including 3 Indo-Pacific genera (Fungia, Caulastrea and Leptoria). The holding (quarantine) tank contained a few large colonies of stony corals, including M. faveolata, M. cavernosa, P. astreoides and gorgonians Pseudopterogorgia), as well as a large pile of live rock. The main settlement tank (50 000L tank formerly used for tourist-related helmet diving) had a large central pile of reef rock with a few isolated corals and numerous Caribbean reef fishes, including angelfish, doctorfish, wrasses and parrotfish. For coral propagation, fragments or pieces of colonies averaging 2-5cm across or in length were used. These included small clippings taken from mature gorgonian colonies using clippers; fragments from branching corals; and square, rectangular, or irregular "plugs" (explants) from massive and plating corals removed with a rotary tool and a cutting wheel. The explants were attached to a base using an adhesive. The base was shaped like a cross and made of resin and sand with one small hole for attachment and a separate hole for attachment to the reef or other substrate (Fig. 5). Second and third "generation" clippings were marked using colored bands.
Restoration efforts: The former owner of AMT had used fragments propagated at this facility for a reef restoration effort in Mustique, and in experimental trials on reefs in Dominica. In Dominica, fragments at different locations were attached to Reef Balls (Reef Ball Development Group, LTD, Doraville, Georgia) and also anchored directly onto the reef. The initial pilot study involved 443 propagules of 26 species, consisting of 1) ten stony corals in the genera Montastraea, Porites, Eusmilia, Mycetophyllia, Diploria, Agaricia and Meandrina; 2) two colonial anemones in the genera Palythoa and Zoanthus; and 3) eleven gorgonians in the genera Eunicea, Pseudopterigorgia, Muricea, Plexaura, Pseudoplexaura, Erythropodium, Muriceopsis and Pterogorgia. Outplants (27) were identified and assessed in one location. This included 18 that had died, including all of the (12) massive corals, four gorgonians, and two brooding corals (Agaricia). Two fragments, both gorgonians (Psuedopterogorgia bipinnata) exhibited substantial upward growth (colonies were 15-20cm in height) while two encrusting Erythropodium caribaeorum colonies and one colony of the colonial anemone Palythoa had expanded off the base and were overgrowing and smothering adjacent corals. Two other survivors were stony corals (P. porites) that appeared healthy, but remained small (5-15cm) with little evidence of new growth and dense mats of algae encircling the base of the colony (Fig. 5). A thorough search of the restoration site, examination of a reef ball placed at a second site, and examination of the substrate surrounding the outflow pipe did not reveal any Indo-Pacific stony corals or soft corals.
Discussion
Production of corals for home aquaria: The Oceanographic Institute of Dominica was established originally as a for-profit coral farm with the goal of producing corals and other cnidarians from fragments taken from wild-harvested specimens to supply international markets with home aquarium specimens. The facility started with Indo-Pacific corals obtained from Indonesia, with exports of several shipments of ´second generation" fragments to the U.S. These were supplemented with, and eventually replaced by western Atlantic corals harvested from waters in Dominica.
Over the last decade there has been a 10-30% annual increase in the export of live stony corals (primarily scleractinian corals for home aquaria, with over 98% taken from the wild and more than 1.5 million corals in trade during 2007 (Bruckner 2005, unpubl data). Most of these are from Indonesia, as well as several other southeast Asian and Pacific Island nations, with less than 1% from western Atlantic reefs; over 80% are destined for the U.S. Because of the increasing threats to coral reefs, including recent climate-induced bleaching events, and worldwide declines in living coral cover, there are growing concerns of the sustainability of the wild harvest of corals. Concurrently, there is recognition of the importance of this trade as a source of revenue for developing countries and potential educational value of aquarium displays. In recent years, there has been a move to switch from wild harvest to in situ coral production facilities, especially in the Indo-Pacific. Most of these efforts involve the fragmentation and grow-out of corals on racks placed in shallow lagoonal environments. While there are economic risks associated with this practice, especially losses due to storm damage, bleaching events and other stressors, this approach supports entire communities and can be much less detrimental to wild populations if done in an environmentally friendly way. Further improvements to coral mariculture practices, to the point it can replace wild harvesting for the ornamental trade, is a priority measure to enhance conservation of reefs while still allowing trade in corals for home aquaria. Nevertheless, there is a critical need to develop internationally recognized environmental standards, criteria for certification in best practices, and improved reporting guidelines for coral farms to minimize impacts to the wild associated with collection of brood stock, and reduce potential risks of introductions of non-native species. For instance, it is possible to take only a portion of an individual colony for each preferred species and color variety, and use this as the donor colony to produce first generation fragments. These fragments could be allowed to grow up beyond marketable size, with periodic removal of small branches or fragments that are grown to market size and exported. This would minimize the need to supplement production with additional specimens removed from the wild. Other standards regarding the sizes of fragments and the amount of time for grow-out prior to export need to be established, possibly including the use of some sort of pit tag to allow rapid verification that the corals were in fact produced following recognized mariculture standards. In addition, coral farms should only use locally harvested corals as source colonies to reduce potential introductions of pathogens, diseases and non-native species. Utilization of sexual reproduction, especially for brooding species, is also possible.
CITES requirements: There are specific international requirements that must be addressed to legally export of all stony corals (but not currently for soft corals) associated with their Appendix II listing in CITES. CITES requires that any export of a listed specimen must be authorized with an export permit based on a management program implemented by the country´s CITES scientific authority, and administered by its CITES management authority. The management plan should include a variety of control measures such as quotas for wild harvest, which are based on the abundance and population dynamics of the species in trade (Armstrong & Crawford 1998; Green and Hendry 1999). CITES permits can be issued for export only if they address three criteria: 1) the specimens were acquired legally, in accordance with the country´s laws and regulations; 2) the export will not be detrimental to the survival of the species or its role in the ecosystem; and 3) live specimens will be prepared and shipped so as to minimize the risk of injury, damage to health or cruel treatment. One of the difficulties for corals has been a lack of baseline data on the status and trends of CITES listed corals and a simple approach to determine what constitutes sustainable levels of harvest. An approach to manage coral fisheries was proposed for Indonesia, which relied on a determination of the total amount of habitat occupied by the species of interest, the abundance and size structure of the species within its preferred habitats, and the coral life history (Bruckner 2003, Bruckner & Borneman 2006).
Ensuring sustainable harvest: At the time of this study, the government of Dominica had not yet implemented many of the CITES requirements and it lacked the information necessary to make a determination that exports are non-detrimental. OID was the first western Atlantic-based coral farm intending to produce corals for international markets. Other Caribbean nations, as well as the United States, currently ban the wild harvest of stony corals because of the importance of these as reef builders, habitat for other species, sources of food, and important sources of primary productivity (Bruckner 2005). Furthermore, western Atlantic reefs have experienced widespread declines in living coral cover, and additional collection may severely compromise the health and resilience of these reefs. Dominica´s reefs are affected by many of the same stressors observed throughout the region, including threats from overfishing, coastal development, land based pollution, sedimentation and effects from climate change. Benthic reef surveys were conducted on the west coast of Dominica in the early to mid 1980s by Goodwin (1985) to assess the potential value of artificial reefs. The fringing reefs of Toucarie Bay and Douglas Bay were considered at the time to be the finest in Dominica, with high coral species diversity and living coral cover, although some algal overgrowth of corals was observed and macroalgae were conspicuous between corals (Putney et al. 1983; Goodwin 1985). In addition, west coast reefs were reported to be affected by industrial/urban activity and river input, with considerable turbidity and silt observed on reefs in northern Prince Rupert´s Bay, Scott´s Head, and in areas between Layou and Pte. Tarou (Goodwin 1985). During the present surveys, coral cover was moderate to low and it appeared to be declining. A high prevalence of disease and other biotic stressors were recorded, which had also been previously reported (Borger 2003), and corals exhibited high levels of recent mortality and ongoing degradation.
The extensive and ongoing decline of Caribbean reefs emphasizes the need to limit harvest of corals. In Indonesia, recommendations were developed that included a conservative harvest of no more than 5% of the population of the target species for fast growing species with high recruitment, with 1-2% allowable take of other slow growing species provided their populations exceeded some minimal baseline abundance and there was evidence of recruitment (Bruckner & Borneman 2006). More conservative estimates need to be adopted for the Caribbean and certain species from the Caribbean should be completely avoided. This includes a ban on the collection of Acropora palmata and Montastraea annularis (complex) due to unusually low levels of recruitment, widespread losses experienced over the last two decades, and an ongoing regional decline of these species due to disease and bleaching (Bruckner & Hill 2009).
Reef restoration: The benefits of the pilot restoration experiments conducted by AMT need to be carefully evaluated before additional restoration efforts are undertaken. OID initially obtained a permit from the government of Dominica to harvest over 3 000 colonies from local reefs, with agreement that they would replace each colony that was removed with 10 new corals. There were few details on the types or sizes of corals that would be harvested and returned to the reefs, sites for collection, or the approach used to identify sites for restoration and implement and monitor restoration efforts. Before allowing additional harvest and restoration efforts, a thorough analysis of the benefits from a conservation perspective should be undertaken. This should include an evaluation of existing threats and the likelihood that these threats will impact the survival of transplanted corals, the condition of reefs and their need for restoration, optimal species and sizes for transplant and methods/substrates for attachment. For instance, one of the primarily sites for the pilot restoration project, Toucari Bay reef, was in good condition. This site contained a high diversity and cover of stony corals and a low prevalence of disease and recent mortality; coral restoration was likely to have minimal benefits at this site. Corals transplanted onto the Reef Balls at other sites included many different coral species with vastly different life histories, and these corals were planted within 5-10cm of each other, suggesting the possibility of competition and potential mortality of the weaker competitor in the near future.
Furthermore, benefits and limitations of natural substrates versus artificial substrates such as a Reef Ball need to be carefully considered before more of the structures are placed into the marine environment.
Invasives: While it does not appear that MT purposely introduced Pacific corals to Dominica´s reefs, the onshore culture of Pacific corals presents a significant threat because the facility is not sufficiently isolated from the sea. Although AMT had systems of filtration and UV sterilization in place, Dominican officials should be permitted to inspect the system in detail to ensure the filtration method is adequate to treat effluent water and not subject to failure. In addition to the danger that imported species will spatially outcompete indigenous corals, there is the danger of introducing exotic diseases. The risk of growing Indo-Pacific corals in a Caribbean-based facility with direct connection to the ocean is unacceptable, even in situations where filtration and UV sterilization of water is undertaken before release into the sea, due to a high potential for accidental release (e.g. if the filtration system is not functioning properly or the facility is hit by a hurricane).
Conclusions
Acknowledgments
References
Armstrong, J. & J. Crawford. 1998. Convention of International Trade in Endangered Species of Wild Fauna and Flora, p. 65- 67. In M.E. Hatziolos, A.J. Hooten & M. Fodor (eds.) Coral Reefs Challenges and Opportunities for Sustainable Management, The World Bank, Washington, D.C., USA. [ Links ]
Bruckner A.W. 2001. Coral health and mortality. Recognizing the signs of coral diseases and predators, p. 240-271. In P. Humann & N. Deloach. Reef Coral Identification: Florida Caribbean Bahamas Including Marine Plants. [ Links ]
Bruckner, A.W. 2003. Sustainable Management Guidelines for Stony Coral Fisheries, p.167-184. In J. Cato & C. Brown (eds.). Marine Ornamental Species Collection, Culture and Conservation. Blackwell Scientific, Iowa State University, Iowa,USA. [ Links ]
Bruckner, A.W. 2005. The importance of the marine ornamental reef fish trade in the wider Caribbean. Rev. Biol. Trop. 53 (Suppl. 1): 127-138. [ Links ]
Bruckner, A.W. & E. Borneman. 2006. Developing a sustainable harvest regime for Indonesias stony coral fishery.Proc. 10th Intern. Coral Reef Symp. 1692-1697. [ Links ]
Bruckner, A.W. & R.J. Bruckner. 2006. Restoration outcomes of the Fortuna Reefer Grounding at Mona Island, Puerto Rico, Chapter 19. In W.F. Precht (ed.). Coral Reef Restoration Handbook - The Rehabilitation of an Ecosystem Under Siege CRC, Boca Raton, Florida, USA. [ Links ]
Bruckner, A., R. Bruckner & R. Hill. 2009. Rethinking restoration approaches for Acropora palmata: lessons from the Fortuna Reefer grounding in Puerto Rico. Proc. 11th Coral Reef Symp., Ft. Lauderdale 2: 1205-1209. [ Links ]
Goodwin, M.H. 1985. Feasibility study on the establishment of artificial reefs. TCP/DMI/4402. FAO Draft Technical Report. [ Links ]
Putney, A.D., G. Butler, S. Edwards & A. James. 1983. Background information on the proposed Cabrits National Park. Draft Report. [ Links ]
Spalding, M.D., C. Ravilious & E.P. Green. 2001. World Atlas of Coral Reefs. Chapter 6 Eastern Caribbean and Atlantic, p 162-163. UNEP World Conservation Monitoring Centre, University of California, Berkeley, USA. [ Links ]
Steiner, S.C.C. & J.L. Borger. 2001. Coral assemblages of Dominica, West Indies: An Introduction. Reef Encounter 28: 20-23. [ Links ]
Correspondencia: Andrew W. Bruckner. Khaled bin Sultan Living Oceans Foundation, Landover, Maryland 20978, USA;bruckner@livingoceansfoundation.org / NOAA Fisheries Office of Habitat Conservation, NOAA Coral reef Conservation Program, Silver Spring, MD 20910 USA. Eric H. Borneman. University of Houston, Houston TX USA.
Received 20-VII-2009. Corrected 06-I-2010. Accepted 12-VII-2010.