Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.58 n.2 San José Jun. 2010
Distribution and size of benthic marine habitats in Dominica, Lesser Antilles
Sascha Claus Christoff Steiner1 & Demian Alexander Willette1,2
1. Institute for Tropical Marine Ecology Inc. (ITME), P.O. Box 944, Roseau, Dominica; admin@itme.org
2. Department of Environmental Health Sciences, UCLA, CHS Box 951772, Los Angeles, CA 90095-1772, USA; demianwillette@ucla.edu
Abstract
Surveys of benthic marine habitats encompassing 1 814.7ha and lining 90% of Dominica’s shoreline were carried out to build the first composite picture of the distribution and size of the island’s near-shore sublittoral habitats, and the epibenthic communities they harbor. Field survey sites covered areas ranging from 1 425 to 29.6ha, lining the shore in bands ranging between 50 and 250m in width, in waters no deeper than 30m. Thus a total of 755ha of benthos were surveyed in October and November of 2007. The benthic habitat composition of an additional 1 059.7ha was inferred with the help of unpublished data and satellite imagery. Seagrass beds were the most widespread organism-built habitat type with 265ha. Coral reefs covered 72.2ha. Both of these habitats were predominantly established along the West and North coasts, which included the island’s most habitatdiverse regions. Rocky environments (911.5ha) dominated the East and South coast and together with sandy areas (566ha) constituted 81% of the island’s marine benthos. It is apparent that seagrass beds, which include four native and one invasive seagrass species, had not been surveyed as previous distribution reports could not be confirmed. Similarly, the benthic cover of Dominica’s coral reefs is evidently far below the previously reported 7 000ha. Such discrepancies highlight the advantage of environmental assessments based on field surveys and systematic data compilation, particularly in cases like Dominica where a narrow island shelf stages marginal marine resources in spatial proximity to each other and human settlements. This study has demonstrated how low-tech field methods can be applied on an island-wide scale to build an inventory of marine resources in the form of habitat maps and data repositories publicly accessible for future use. In the absence of such efforts, the development of conservation measures and status reports will remain ill founded. Rev. Biol. Trop. 58 (2): 589- 602. Epub 2010 June 02.
Key words: benthic habitats, Dominica, habitat maps, seagrasses, coral reefs.
Resumen
Los ambientes béntico-marinos de Dominica, Antillas Menores, incluyen 1 814.7ha y cerca del 90% de la costa. Este es el primer trabajo sobre el tamaño y distribución de los ambientes costeros sublitorales y sus comunidades epibénticas. Los pastos marinos constituyeron el ambiente más extenso, con 265ha. Los arrecifes coralinos presentaron una cobertura de 72.2ha. Ambos tipos de ambientes se encontraron principalmente en la costa oeste y norte, regiones con la mayor diversidad de ambientes marinos. Las áreas rocosas (911.5ha) dominaron las costas este y sur de la isla, y junto con las áreas arenosas (566ha) constituyeron el 81% de los ambientes bénticos. La distribución de pastos marinos, que incluyeron cuatro especies nativas y una invasiva, no se pudo corroborar con los pocos informes previos. La cobertura béntica de los arrecifes coralinos de Dominica fue mucho menor que las 7 000ha que se habían reportado previamente. Estas discrepancias ilustran la ventaja de los estudios ambientales basados en trabajo de campo y la compilación sistemática de datos ambientales, especialmente en casos como Dominica donde por lo angosto de la plataforma insular, los ambientes marinos sublitorales quedan muy cerca de los centros urbanos. Se demuestra que técnicas simples pueden ser utilizadas a gran escala alrededor de una isla para realizar inventarios de los recursos marino-costeros, mediante mapas y repositorios públicos de datos para usos futuros, que permiten el desarrollo de medidas de conservación efectivas.
Palabras clave: ambientes bénticos, Dominica, mapas, pastos marinos, arrecifes coralinos.The scientific assessment of benthic marine habitats along the mountainous eastern Caribbean island of Dominica has only recently received increased attention. As part of the Bredin-Archbold-Smithsonian Biological Survey of Dominica, early investigations focused on the distribution of marine algae (Randolph & Rhine 1970), burrowing sponges (Rützler 1971), balanomorph cirripeds (Ross 1968), decapods (Kristeuer 1967, Raymond 1970) and echinoids (Porter 1966) from a few locations. Recently, thematically broader studies have been carried out on the distribution of stony corals and reefs (Steiner 2003), coral diseases (Borger 2003, 2005, Borger & Steiner 2005) and bleaching events (Steiner & Kerr 2008), the abundance and distribution of the echinoid Diadema antillarum (Steiner & Williams 2006a, 2006b), and the distribution dynamics of the invasive seagrass Halophila stipulacea (Willette & Ambrose 2009). Non-peer reviewed reports, primarily on reefs and reef fishes from the West coast, include the work of Judge et al. (1987), Weyerman et al. (1996), Lucas et al. (2001), Williams et al. (2001), Smith et al. (2002), Diamond (2003), Green (2003), Knuth (2003), McDonald (2003), Ishikawa et al. (2004), Byrd et al. (2005) & Davis et al. (2006).
Prior to this study, however, a comprehensive large-scale quantitative survey of the coastal marine habitats had not been carried out in Dominica, leaving over 90% of the island’s near shore waters un-assessed. Therefore, previous reports could not be viewed within an island-wide context as concrete figures regarding the distribution, size, makeup and condition of the island’s marine habitats did not exist. The possibility to gauge environmental change within and the value of Dominica’s benthic communities outside of a few specific locations has consequently been very limited. For example, the impact of the 2005 coral bleaching episode was assessed by comparing coral communities at 16 locations during and after the episode (Steiner & Kerr 2008), yet information on how many hectares of reef exist along the island or how extensive Dominica’s coral reef resources are, had not yet been established. Only a narrow window into the scale and overall ramifications of such events was therefore opened. Similarly, the degradation of habitats that cover a comparatively small area of the island’s sub-littoral, may lead to the decrease in biodiversity and other ecological parameters, but to fewer socio-economic consequences than the degradation of habitats covering larger areas. In the absence of quantitative distinctions between common and rare habitats, decisions as to whether or not priority shall be given to conservation measures aimed at the larger or the smaller epibenthic communities were not an option. Despite these information gaps, status reports including the distribution and condition of Dominica’s benthic sublittoral resources continue to be written (Bruke & Maidens 2004, Gov. of Dominica 2005) without supporting field investigations, and become the basis for the development of local environmental policy.
Dominica’s steep terrestrial terrain and narrow island shelf put the benthic sublittoral habitats within a few tens to hundreds of meters of the island’s coastal settlements. Although the population has gradually declined since the early 1900’s and is currently at approximately 71 000 (2002 Census Report, Gov. of Dominica), urban development along coastal zones is growing as Dominicans are moving away from the mountainous interior of the island, abandoning agricultural and self- sustaining lifestyles. This is resulting in an increased infrastructural and consumer footprint in the form of larger concrete houses with comparatively fewer inhabitants and a greater dependency on industrially manufactured imported goods. Visible signs of detrimental inputs of terrestrial sediments, degradable and non-degradable "contaminants" sensu GESAMP (1990) are rising in number and remain unabated. The most evident sources of these inputs (solid wastes, sewage, chemicals, and sediments from poor land use) are currently areas of urban expansion, smallscale manufacturing, dumping sites, and quarries along the West coast.
Dominica’s benthic marine habitats support a variety of extractive (e.g. artisanal fisheries targeting a broad range of demersal and pelagic fishes, some invertebrates, and algae), and non-extractive resource uses (e.g. tourism). A variety of extraction practices (e.g. fishing, coral mining into the 1950´s), particularly along the western and northern shores, have occurred over the past three centuries. To date, coastal fishing practices including the use of fish pots, spear guns, seine nets, and occasionally gill nets continue to be sources of chronic local disturbances, in addition to the aforementioned disturbances from coastal developments. Together with regional disturbances (e.g. elevated sea surface temperatures) an unprecedented number of stressors are affecting the island’s benthic communities and the dwindling resources they provide. In this situation, an island-wide comprehensive assessment and quantification of Dominica’s benthos is an essential prerequisite for building the baseline of insightful resource conservation measures.
This study reduces the abovementioned information gaps by: (1) identifying the principal benthic marine habitats found along Dominica’s shoreline; (2) categorizing the habitats using common biotic and abiotic attributes applicable in island-wide field surveys; (3) determining the geographic distribution and size of each habitat type in order to (4) build a composite picture (habitat maps and publicly accessible survey data) of Dominica’s near shore benthos as context for both previous studies and a reference point for future studies. Low-tech field methods, local knowledge, and the systematic compilation of published and unpublished field data form the foundations of this study, to demonstrate its applicability in regions where financial and technical support for large scale habitat assessments are limited.
Materials and methods
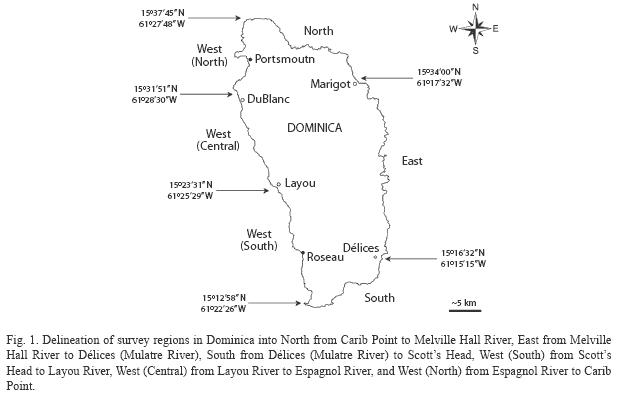
Habitat categories and survey regions: Four habitat categories, rock, sand, seagrass and coral reef, each including up to four habitat types (Table 1) were selected and defined based on field data recorded by S. Steiner between 1998 and 2007, and habitat descriptions in Steiner (2003), Borger & Steiner (2005), Steiner & Williams (2006a), Steiner & Kerr (2008), and Willette & Ambrose (2009). Two principal criteria guided the selection process: (1) habitat categories and types had to reflect the known heterogeneity of habitats, and (2) the number of categories and types had to be kept at a minimum to allow for a rapid classification during large-scale field surveys (≥1 000m2). In order to facilitate the assessment of the island’s coastal environments along its 176km of shoreline, six regions were delineated (Fig. 1). The regional boundaries were selected to include geographic features easily identifiable by future surveyors (e.g. river mouth) and by the diversity of habitats to be expected within each region based on aforementioned studies. Therefore, the high energy eastern shore of Dominica’s windward coast was expected to harbor a homogenous array of near shore benthic habitats and was not subdivided.
Surveys: Field survey sites covered areas ranging from 1 425 to 29.6ha, lining the shore in bands ranging between 50 and 250m in width, in waters no deeper than 30m. Thus a total of 755ha of benthos were surveyed in October and November of 2007. At each site, the surface cover of each habitat category and type were estimated. In addition, the live cover of eight epibenthic biotic characteristics (erect macroalgae, turf algae, calcareous rhodophytes in the genus Porolithon, seagrasses, sponges, hydrocorals in the genus Millepora, encrusting non- scleractinian anthozoans, and scleractinians were ranked as follows: 1(0%), 2(1- 5%), 3(6-10%), 4(11-25%), 5(26-50%), 6(51-75) and 7(76-100%). The nominal ranking system, which focuses on the 1-25% range, was chosen based on benthic cover data from the aforementioned authors.
Extensive portions of Dominica’s coastal marine habitats, particularly along the East coast, are marked by treacherous marine conditions making field surveys using SCUBA or by snorkeling unsafe or impossible on most days of the year. However, the presence of habitat categories and types in such areas could be inferred for some sites for which either published or unpublished field observations, or quantitative data were available. In the absence of previous data, inferences on the near-shore sublittoral habitat categories were made, where possible, using the criteria of exposure (windward or leeward), turbulence (reflective or dissipative shores), headland morphology (cliff, slope, valley, rivers), shoreline or beach type (sandy or rocky), available imagery and data from adjacent areas or locations with similar attributes. This allowed for the composition assessment of an additional 1 059.7ha of benthos. Sections of shoreline for which field surveys or inferences could not be made, and which remain un-assessed (see Fig. 2a-2g), added up to 17.2km.
Data collection, compilation and analysis: Three to six surveyors collected field data simultaneously by snorkeling parallel to shore and each other (evenly spread out throughout the width of the survey band) from the predetermined starting points to the ending points of each site. Where the sublittoral zones were steep or marked by structurally simple habitats (e.g. homogenous sandy environments) survey bands remained narrow (50-100m). SCUBA dives were carried out to survey structurally more complex sites, particularly reefs, in depths beyond 8m. A total of 203 hours of field assessments were thus carried out. Observations by individual surveyors (including estimates of habitat dimensions, habitat map sketches, references to landmarks along the shore and underwater, as well as the benthic cover rankings of epibenthic biotic characteristics), were compared and corroborated to produce a single data entry for each site. Sixteen hours of onsistency training in the field, and with the aid of projected images, were allocated to ensure that surveyors estimated the percent cover congruently. Survey and habitat areas were determined using in-field estimates and landmarks. Images by Google Earth were compared to map sketches and notes produced by surveyors in the field to assist in establishing the location and benthic cover of individual habitat types. The island circumference measurement used to build habitat distribution maps was derived by the sum of linear segments no longer than 100m, contouring the island 25m off shore. Many locations in Dominica have multiple names. Site names used here are based on the maps published by the British Government’s Ministry of Overseas Development, series E803, edition 4 DOS 1978, scale 1:25 000. A repository of all coordinates delineating individual sites as well as numerical data used in generating the site maps (Fig. 2a-2g) was compiled and stored as an appendix at http://www.itme.org/data.htm for public access. Similarities among the habitat types assessed in the field, based on their benthic cover rankings of biotic attributes, were discerned with the Bray-Curtis similarity cluster analysis. All calculations were performed in Primer v5 (Clarke & Groley, 2001).
Results
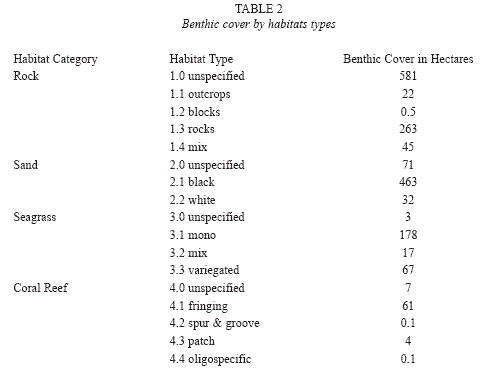
Regional characteristics: Of the 1 814.7ha surveyed, 50.2% were composed of rocky habitats and 31.2% of sandy environments (Table 2). Habitats in which living organisms are highly involved in shaping and building the substrate constituted 14.5% (seagrasses) and 4.0% (coral reefs). All but one seagrass bed and most coral reefs were found along the western (Fig. 2a-2c) and northern regions (Fig. 2d), which also contained 88% of the island’s sandy environments. The most heterogeneous regions, with regards to the number of habitat categories and types, were also the western and northern regions (Fig. 3a, 3b). In contrast, more homogenous rock dominated environments characterized eastern and southern regions.
Western region: The west coast (Fig. 2a-2c) harbored the largest sandy coastal environments in conjunction with the most extensive seagrass beds and the second largest area of coral reefs on the island (Fig. 3b). Where rocky substrates characterized the eulittoral zone, near-shore sublittoral areas were also rocky. Seagrass beds in the western central region (248ha) were dominated by the seagrass Syringodium filiforme growing in depths between 2 and 18m. Halophila decipiens and H. stipulacea were also identified within this depth range, commonly along the shallow and deep perimeter of continuous S. filiforme beds. They occasionally also occurred in "erosional holes" and as patches within S. filiforme beds. The depth limit of both Halophila species could not be determined. Halodule wrightii was occasionally identified along the shallow margins of S. filiforme beds. River mouths, steep sublittoral areas (lining steep headlands) and coral reefs were the main features interrupting the otherwise continuous band of seagrass beds between Fond Colé (Fig. 2a) and Anse Lamothe (Fig. 2c). Towards their northern and southern distribution limits along the West coast, seagrass beds were patchy and with limited or no spatial overlap between seagrass species, in particular where the invasive H.stipulacea had established itself.
Dominica’s second largest coral reef area (approx. 15.7ha) comprises the reef system of the Grande Savane and the reefs of Mero (Fig. 2b). Located in depths between 5 and 40m, they include well-developed spur-and-grove zones, structurally complex and rugose reefs, as well as oligospecific coral assemblages dominated by the stony coral Madracis mirabilis. Unlike other coral habitats along the West coast, deposits of white calcareous sands distinguished these sites. The majority of the vast west coast sandy habitats were black and of volcanic origin. Small areas of reef accretion were also observed at Tarou Point and Cachacrou (Fig. 2a West of the village of Scott’s Head), and Toucari and Douglas Bay (Fig. 2c). Assemblages of sessile organisms with the biotic attributes examined here (e.g. calcareous rhodophytes, sponges, stony corals etc.) were observed on most rocky substrates, but were best developed at locations most exposed to ocean currents and furthest away from rivers (e.g. northern side of Soufriere Bay). The epibenthic cover was higher on the north facing sides of outcrops or peninsulas such as Tarou Point (Fig. 2a) and Cabrits (Fig. 2c).
Northern region: The northern region (Fig. 2d) of Dominica included large rocky areas, in particular between Capucin and Au Parc, but also the largest fringing reef system (33ha), the second largest area of seagrass beds (17.7ha), and a close to even mix of black and white sandy areas. The fringing reefs were characterized by frameworks of the branching scleractinian coral Acropora palmata and lined with the massive (Siderastrea siderea, Montastraea faveolata) and encrusting (Diploria clivosa, D. strigosa) species. However, live coral cover was less than 1% in most locations. The reef flats and back reef areas provide the necessary habitat for the north coast seagrass beds, which remain in shallow waters (0-6m). Thalassia testudinum was only found in this region (and a single location in the East) where it was a common to dominant seagrass species. S. filiforme was also common and well intermixed with T. testudinum, while H. wrightii occasionally occurred in protected areas along seagrass bed margins. The Halophila species were not seen in this region.
Eastern and southern regions: Dominica’s eastern (Fig. 2e-2f) and southern (Fig. 2g) regions were similar in habitat composition and dominated by rocky environments. Fringing reefs, covering a total of approximately 10ha lined the north facing shores of Middle Bay (East of Marigot), Anse Quaneri and Saint Sauveur, and approximately 2ha of fringing reef were identified along the south facing shore by Dubuc (East of Grand Bay). Despite the minimal live coral cover (<0.5%), A. palmata frameworks were clearly recognizable as a principal architectural scleractinian component. The only other true coral reefs identified in this region were small patch reefs (approx. 0.04ha) including M. mirabilis assemblages located 75m off the southwestern shore of Carib Point (Roche Cassé). Seagrasses were only observed at Middle Bay (East of Marigot) in the form of small patches of T. testudinum totaling less than 0.01ha.
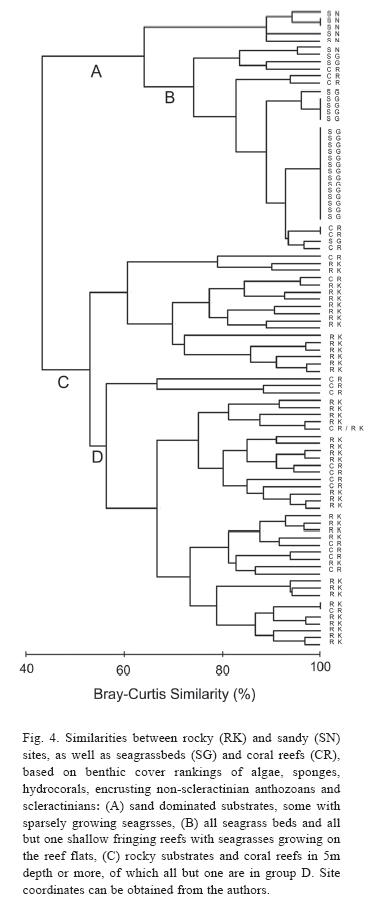
Comparisons in benthic composition: The comparison of habitats, based on the benthic cover rankings of biotic attributes (Fig. 4), shows sandy environments (some of which include areas of sparse seagrass growth but no beds) and seagrass beds forming a distinct group (Fig. 4, group A) with a pronounced similarity among most seagrass beds. Within this group are also all but one of the shallow water fringing reefs in less than 5m depth (Fig. 4, group B). The reef flats of shallow water fringing reefs (exclusively from the North coast) provide the habitat for seagrasses and thus have a similar benthic composition with the latter. Deeper coral reefs in more than 5m depth formed a much more heterogeneous group with all rocky habitats (Fig. 4, group C), and all reefs but one were in one of two subgroups (Fig. 4, group D). The stable substrate provided by rocky habitats support communities of algae, sponges, hydrocorals, encrusting non-scleractinian anthozoans and scleractinians (as ranked in this study) similar to those of coral reefs. Therefore, coral reefs in Dominica do not have a distinctive "signature" which clearly separates them from other habitat types when comparing the biotic attributes chosen here.
Discussion
This first large-scale marine habitat survey of Dominica covered 90% of the shoreline and has provided a composite picture of the island’s benthic environments. Previous and future studies can now be viewed within an island-wide context. It is now evident that the marine plants of sandy substrates, in the form of seagrass beds, represent the island’s largest epibenthic habitat type. Prior to this study, the geographic distribution, overall size and species composition of Dominica’s seagrasses were undetermined. Although the distribution of seagrasses around Dominica has been illustrated in a report by the Government of Dominica (2005), the information provided could not be corroborated by this survey and does not appear to be based on field surveys. Only minor additions, if any, to the geographic distribution of Dominica’s seagrass beds established by this study may be expected from additional studies, as the areas which remain unsurveyed do not include conditions suitable for seagrass beds. In contrast, the benthic cover of seagrass beds recorded here (265ha) is a conservative estimate because the deep margins of some beds were outside of the offshore boundary of survey sites, and because the depth limit of Halophila spp is beyond the depth limit (30m) of this study. Furthermore, the shallow margins of Dominica’s seagrass beds periodically recede due to their uprooting or burial during storms, and expand during prolonged periods of calm sea conditions (pers. observ.). The total benthic cover of this habitat type must thus be expected to vary seasonally.
Seagrass beds have many ecological and economic values which include their role as a nursery for juvenile fish and invertebrates (Randall 1965, Peterson & Heck 2001) and shelter for adult organisms (Boström & Mattila 1999, Danovaro & Gambi 2002), as well as their functions to stabilize shorelines through wave attenuation (Fonseca & Fisher 1986, Fonseca & Cahalan 1992, Grizzle et al. 1996), decrease suspended material in the water column (Agawin & Duarte 2002, Marba et al. 2006), recycle nutrients and increase oxygen loads (Lee & Dunton 1999, Gacia et al. 2005), and ranking as one of the most productive ecosystems on the planet in terms of biomass and diversity (Constanza et al. 1998, Duarte & Chiscano 1999, Duarte 2002). Therefore, the findings presented here justify a closer look at the condition of this resource, the potential needs for conservation, and mitigation measures, in particular as they relate to the presence of the invasive H. stipulacea as first reported by Willette & Ambrose (2009) and its extensive geographic distribution in Dominica (Steiner et al. unpubl. data).
Dominica’s leeward and windward seagrass beds displayed differences in species composition and depth range. Leeward beds were dominated by S. filiforme, and were typically found between 2 and 18m, in otherwise sandy regions. The narrow and steep shelf of the West coast limits the habitat available for light dependent seagrasses. Near-shore turbulence creates unstable conditions for seagrasses and therefore Dominica’s West coast communities start growing at depths no less than 2m, commonly at 5m, and extend towards their physiological light limits, which in Dominica seem to be at 18m for S. filiforme. Although T. testudinum has lower mean minimum light requirements than S. filiforme at various Caribbean locations (Lee et al. 2007), self-shading by T. testudinum with blade-like leaves may be a disadvantage along the West coast setting, possibly explaining its absence in this region during the survey. A single small mixed patch (<0.05ha) of sparsely growing T. testudinum and S. filiforme, was observed between 2001 and 2007 in a sheltered location near the southern village of Scott’s Head. It was destroyed during Hurricane Dean in August 2007 (pers. observ.).
In contrast, windward beds were characterized by T. testudinum and were growing in depths of 0-4m in back reef areas or behind other sheltering features such as islands. T. testudinum has a the deepest live rhizome mats of the 5 species mentioned (Duarte 1991, den Hartog 1970) as well as unbranched roots with substantial root hairs that are adapted for a range of sediment types (Kuo & den Hartog 2006) which may provide the necessary stability in the sand and coral rubble substrates along the turbulent North coast. The similarities between seagrass beds and sandy areas (West coast) and shallow reefs (North coast), both spatially and based on the biotic attributes surveyed here, indicate that successful protection of seagrasses in Dominica is linked to the protection of the contiguous sandy environments and coral reefs.
To date, most studies on Dominica’s benthic communities focused on coral reefs and assemblages, yet 10.3ha of the reefs surveyed here had previously not been recorded. It has been clear that coral reefs are marginal communities on the narrow shelf of Dominica (Steiner 2003), but just how marginal is now apparent considering that they cover only approximately 72.2ha. This finding does not support two previous estimates of 7 000ha and 4 700ha of coral reefs for Dominica, based on Caribbean wide assessments and listed in Bruke & Maidens (2004). Even when considering the coral assemblages that are found in some of Dominica’s rocky environments, characterized by a lack of reef accretion, as "coral reefs", the benthic cover of "coral reefs" would be far below 1 000ha.
Distinguishing between coral reefs sensu stricto and coral assemblages may be unnecessary as both habitat types harbor and attract organisms supporting the fisheries and tourism interests. However, true coral reefs are bioherms marked by long-term development, growth, and in Dominica by greater coral diversity than rocky habitats (Steiner 2003). Their loss has different consequences than the loss of other weedier coral assemblages growing on rocky substrates without building reefs. In light of poorly mitigated local stresses (e.g. sediment runoff, fishing pressure), regional disturbances (e.g. elevated surface temperatures and coral bleaching events), and the demise of particular framework-building species such as A. palmata (Patterson et al. 2002, Precht et al. 2002), also evident along the island’s northern shores, Dominica’s reef resources are facing continuous deterioration.
With this study the first composite picture of Dominica’s benthic near shore environments has been constructed, thus correcting our understanding of their distribution, size and composition. Seagrasses beds and coral reefs are the only epibenthic habitat types in Dominica formed by organisms. Besides their intrinsic value, they provide numerous ecological and socio-economic benefits. Both of these ecosystems are threatened by similar disturbances and while we have a good understanding of the (marginal) coral reefs of Dominica and the plight they face, little is known about Dominica’s largest resource, the seagrass beds. Furthermore, the preservation of these habitats cannot be addressed individually, as they share spatial limitations, as well as local distribution and composition patterns. This favors conservation initiatives focusing on island regions over habitat-specific ones. Many habitat assessment approaches exist (Diaz et al. 2004.), which can provide the necessary environmental baseline for such conservation initiatives, but most require technical and financial support rarely available or prioritized in small developing nations. This study demonstrates that low-tech field methods, local knowledge, and the systematic compilation of published and unpublished field observations, can be applied on an island-wide scale to build a sound inventory of marine resources. Without such fundamental efforts, the adequacy of conservation measures, local or initiated via international conventions, will remain questionable.
Acknowledgments
We are very grateful for the assistance provided by K. Macfarlane, L. Price, N. Wallover and K. Wilson during the 2007 field surveys. Logistic and financial support for this project was provided by the Institute for Tropical Marine Ecology Inc. (ITME RPfa07/spsufa08).
References
Agawin, N.S.R. & C.M. Duarte. 2002. Evidence of direct particle trapping by a tropical seagrass meadow. Estuaries 25: 1205-1209. [ Links ]
Borger, J.L. 2003. Three scleractinian coral disease in Dominica, West Indies: Distribution, infection patterns and contribution to coral tissue mortality. Rev. Biol. Trop. 81: 25-38. [ Links ]
Borger, J.L. 2005. Dark Spots Syndrome: A scleractinian coral disease or a general stress response? Coral Reefs 24: 139-144. [ Links ]
Borger, J.L. & S.C.C. Steiner. 2005. The spatial and temporal dynamics of coral diseases in Dominica, West Indies. Bull. Mar. Sci. 77: 137-154. [ Links ]
Boström, C. & J. Mattila. 1999. The relative importance of food and shelter for seagrass-associated invertebrates: a latitudinal comparison of habitat choice by isopod grazers. Oecologia 120: 162-170. [ Links ]
Bruke, L. & J. Maidens. 2004. Reefs at risk in the Caribbean. World Resources Institute, Washington D.C., USA. [ Links ]
Byrd, K., M. Jordan, M. Klarman, A. Lowe, J. McNeal, N. Wallover & R. Zuercher. 2005. Atlantic and Gulf Rapid Reef Assessment (AGRRA) protocol v.4.0: First Implementation in the Commonwealth of Dominica, October-November 2005. ITME Research Reports 23: 1-91. [ Links ]
Clarke, K.R. & R.V. Groley. 2001. Primer v5:User Manual/ Tutorial. PrimerE Ltd., Plymouth, UK. [ Links ]
Constanza, R., R. d’Arge, R. De Groot, S. Farber, M. Grasso, B. Hanon, K. Limburg, S. Naeem, R.V. O’Neill, J. Paruelo, R.G. Raskin, P. Sutton & M. van den Belt. 1998. The value of the world’s ecosystem services and natural capital. Ecol. Econ. 25: 3-15. [ Links ]
Danovaro, R. & C. Gambi. 2002. Biodiversity and trophic structure of nematode assemblages in seagrass systems: evidence for a coupling with changes in food availability. Mar. Biol. 141: 667-677. [ Links ]
Davis, C., S. Laffan, J. Sabattis & J. Weems. 2006. Atlantic and Gulf Rapid Reed Assessment (AGRRA) protocol v.4.0, October - November 2006: Quantifying the Impact of the 2005 Bleaching Episode in Dominica. ITME Research Reports 24: 1-40. [ Links ]
Den Hartog, C. 1970. The sea-grasses of the world. North- Holland, Amsterdam, Netherlands. [ Links ]
Diamond, A. 2003. Identification and Assessment of Scleractinians at Tarou Point, Dominica, West Indies. Coast. Manag. 31: 409-421. [ Links ]
Diaz, R.J., M. Sloan & R.M. Valente. 2004. A review of approaches for classifying benthic habitats and evaluating habitat quality. J. Environ. Manage. 73: 165-181. [ Links ]
Duarte, C.M. 1991. Allometric scaling of seagrass form and productivity. Mar. Ecol. Prog. Ser. 77: 289-300. [ Links ]
Duarte, C.M. 2002. The future of seagrass meadows. Environ. Conserv. 29: 192-206. [ Links ]
Duarte, C.M. & C.L. Chiscano. 1999. Seagrass biomass and production: a reassessment. Aquatic. Bot. 65: 159-174. [ Links ]
Fonseca, M.S. & J.S. Fisher. 1986. A comparison of canopy friction and sediment movement between four species of seagrass with reference to their ecology and restoration. Mar. Ecol. Prog. Ser. 29: 15-22. [ Links ]
Fonseca, M.S. & J.A. Cahalan. 1992. A preliminary evaluation of wave attenuation by four species of seagrass. Est. Coast. Shelf Sci. 35: 565-576. [ Links ]
Gacia, E., H. Kennedy, C.M. Duarte, J. Terrados, N. Marba, S. Papadimitriou & M. Fortes. 2005. Light-dependence of the metabolic balance of a highly productive
Philippine seagrass community. J. Exp. Mar. Biol. Ecol. 316: 55-67.
GESAMP. 1990. The state of the marine environment. GESAMP Reports and Studies 39: 1-111. [ Links ]
Grizzle, R.E., F.T. Short, C.R. Newell, H. Hoven & L. Kindblom. 1996. Hydrodynamically induced synchronous waving of seagrasses: "monamis" and its possible effects on larval mussel settlement. J. Exp. Mar. Biol. Ecol. 206: 165-177. [ Links ]
Green, D. 2003. Dominica Reef Fish Status 2002: An Assessment of the Abundance and Species Composition of Dominican Reef Fishes. ITME Research Reports 13: 1-4. [ Links ]
Government of Dominica. 2005. Dominica’s Biodiversity Strategy Action Plan, Government of Dominica, Roseau, Commonwealth of Dominica. [ Links ]
Ishikawa, M., J. Kerr & K. Alfsnes. 2004. Preliminary Surveys of Northern Reefs of Dominica, West Indies. ITME Research Reports 21: 1-26. [ Links ]
Judge, J.G., A. Madisetti & B.M.H. Walter. 1987. Dominica Expedition 1987. [ Links ]
Knuth, J.K. 2003. Scleractinian Monitoring in Dominica, West Indies: Species Richness, Diversity and Live Cover. ITME Research Reports 12: 3-10. [ Links ]
Kristeuer, E. 1967. Bredin-Archbold-Smithsonian Biological Survey of Dominica. 3. Marine archiannelids from Dominica. Proc. U.S. Nat. Mus. 123: 1-6. [ Links ]
Kuo, J. & C. den Hartog. 2006. Seagrass morphology, anatomy, and ultrastructure, p. 51-87. In A.W.D. Larkum, R.J. Orth, C.M. Duarte (eds.). Seagrasses: Biology, Ecology and Conservation. Springer, Netherlands. [ Links ]
Lee, K. & K.H. Dunton. 1999. Inorganic nitrogen acquisition in the seagrass Thalassia testudinum: Development of a whole-plant nitrogen budget. Limnol. Oceanogr. 44: 1204-1215. [ Links ]
Lee, K.S., S.R. Park & Y.K. Kim. 2007. Effects of irradiance, temperature and nutrients on growth dynamics of seagrasses: A review. J. Exp. Mar. Biol. Ecol. 355:144-175. [ Links ]
Lucas, D., W. Lehman & A. Diamond. 2001. The Marine Environment of Tarou Point, Commonwealth of Dominica. ITME Research Reports 6: 1-3. [ Links ]
Marbà, N., M. Holmer, E. Gacia & C. Barron. 2006. Seagrass beds and coastal biogeochemistry, p.135- 157. In A.W.D. Larkum, R.J. Orth, C.M. Duarte, (eds.). Seagrasses: Biology, Ecology and Conservation. Springer, Netherlands. [ Links ]
McDonald, K.L. 2003. The Abundance of Herbivorous and Predatory Fishes in Relation to Diadema antillarum along the West Coast of Dominica. ITME Research Reports 19: 11-21. [ Links ]
Petterson, B.J. & K.L. Heck. 2001. Positive interactions between suspension-feeding bivalves and seagrass–a facultative mutualism. Mar. Ecol. Prog. Ser. 213: 143-155. [ Links ]
Patterson, K.L., J.W. Porter, K.B. Ritchie, S.W. Polson, E. Mueller, E.C. Peters, D.L. Santavy & G.W. Smith. 2002. The eitology of white pox, a lethal disease of the Caribbean elkhorn coral (Acropora palmata). PNAS 99: 8725-8730. [ Links ]
Porter, K.M. 1966. Bredin-Archbold-Smithsonian Biological Survey of Dominica. 1. The Echinoids of Dominica. Proc. U.S. Nat. Mus. 121: 1-10. [ Links ]
Precht, A., A.W. Bruckner, R.B Aronson & R.J. Bruckner. 2002. Endangered Acroporid corals of the Caribbean. Coral Reefs 21: 41-42. [ Links ]
Randall, J.E. 1965. Grazing effect on sea grasses by herbivorous reef fishes in the West Indies. Ecology 46: 255-260. [ Links ]
Randolph, T. & C. Rhyne. 1970. Marine algae of Dominica. Smithson. Contrib. Bot. 3: 1-16. [ Links ]
Raymond, M.B. 1970. Mithrax (Mithraculus) commensalis, a new West Indian spider crab (Decapoda, Majidae) commensal with a sea anemone. Crustaceana 19: 157-160. [ Links ]
Ross, A. 1968. Bredin-Archbold-Smithsonian Biological Survey of Dominica. 8. The intertidal balanomorph Cirripedia. Proc. U.S. Nat. Mus. 125: 1-23. [ Links ]
Rützler, K. 1971. Bredin-Archbold-Smithsonian Biological Survey of Dominica; Borrowing sponges, genus Siphonodictyon Bergquist, from the Caribbean. Smithson. Contrib. Zool. 77: 1-37. [ Links ]
Steiner, S.C.C. 2003. Stony Corals and Reefs of Dominica (Lesser Antilles). Atoll Res. Bull. 498. [ Links ]
Steiner, S.C.C. & J.M. Kerr. 2008. Stony corals in Dominica during the 2005 bleaching episode and one year later. Rev. Biol. Trop. 56: 139-148. [ Links ]
Steiner, S.C.C. & S.M. Williams. 2006a. The Density as Size Distribution of Diadema antillarum of Dominica (Lesser Antilles): 2001-2004. Mar. Biol. 149: 1071-1078. [ Links ]
Steiner, S.C.C. & S.M. Williams. 2006b. A Recent Increase in the Density of the Echinoid Diadema antillarum in Dominica (Lesser Antilles): 2001-2005. Rev. Biol. Trop. 54: 97-103. [ Links ]
Smith, Q., J. McKinney, L. Komoroske & L. Pettersen. 2002. Dominica Coral Reef Status 2002: Assessment of the Sea Urchin Diadema antillarum, Reef Fishes, and Algae. ITME Research Reports 12: 1-25. [ Links ]
Weyerman, M., N. Rogers, I. Williams & G. Goodman. 1996. Rapid assessment of Soufriere and Scott’s Head. Dominica Ecotourism Project, EC Project No: B7-5040-24. [ Links ]
Willette, D.A. & R.F. Ambrose. 2009. The distribution and expansion of the invasive seagrass Halophila stipulacea in Dominica, West Indies, with a preliminary report from St. Lucia. Aquatic Bot. 91: 137-142. [ Links ]
Williams, S.M., E. Mohan, D.A. Willette & N. Lestrade. 2001. Dominica Coral Reef Status 2001: Assessment of the Sea Urchin Diadema antillarum, Reef Fishes, Algae and Sponges. ITME Research Reports 9: 1-30. [ Links ]