Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.57 no.3 San José sep. 2009
It takes two to tango: self incompatibility in the bromeliad Tillandsia streptophylla (Bromeliaceae) in Mexico
Ivón M. Ramírez Morillo, Francisco Chi May, Germán Carnevali & Filogonio May Pat
Centro de Investigación Científica de Yucatán, A.C., Unidad de Recursos Naturales-Herbario CICY, Calle 43#130, Colonia Chuburná de Hidalgo, Mérida 97200, Yucatán, México; ramirez@cicy.mx; frankcm@cicy.mx; carneval@cicy.mx; mamu@cicy.mx
Abstract: Floral phenology and breeding system of Tillandsia streptophylla (Bromeliaceae) were studied in a low inundated forest in Yucatan, Mexico. During the flowering season, from March to August, terminal scapose 1-branched, paniculate inflorescences are produced with one flower per branch opening per day, over a period of 11-29 days. Flowers are tubular, light violet, with the stigma placed below the anthers, both protruding above the corolla. Flowers are protandrous, with anthers releasing pollen from 0500 hours and stigma becoming receptive around 0900 hours. Controlled experimental crosses suggest that Tillandsia streptophylla is self incompatible and therefore, pollinator-dependent. Rev. Biol. Trop. 57 (3): 761-770. Epub 2009 September 30.
Key words: breeding systems, Bromeliaceae, conservation, Yucatan Peninsula.
Bromeliaceae is the largest endemic family to the Neotropics (albeit there is a Pitcairnia LHér species in W. Africa), with 56 genera and approximately 3 086 species (Luther 2006). Mexico has 18 genera and 342 species of Bromeliaceae (Espejo-Serna et al. 2004), with two endemic genera (Ursulaea Read & Baensch) and Viridantha Espejo, an adiditional genus which is almost restricted to its territory, Hechtia Klotzsch, and centers of diversity for Tillandsia L. subgenus Tillandsia, and for Pitcairnia.
Bromeliaceae constitutes a clade composed almost entirely of hermaphroditic species. Most of the flowers of the species in the family are bisexual; unisexual flowers have been reported for the Mesoamerican and Mexican species of Hechtia Klotzsch, in some species of Catopsis Griseb., for the single species of Androlepis Brongn. ex Houllet, and in one species in the polyphyletic genus Aechmea Ruiz & Pav. (de Faria et al. 2004). Members of Dyckia Schult. f. (Benzing et al. 2000), Cottendorfia Schult. f. (Smith 1986), and Cryptanthus Otto & A. Dietr. spp. (Ramírez-Morillo 1996) have unisexual flowers as well, representing the only groups in the family with this unusual sex distribution south of the Equator (Benzing et al. 2000).
Preliminary hypotheses of mating systems are based on flower sex and their distribution within and among individuals in the population, but few controlled pollination experiments have been carried out to test such hypotheses. Most of the reports, based on experimental crosses, suggest self compatibility and spontaneous self-pollination as a rule in hermaphroditic species, such as in Pitcairnia species and some of its natural hybrids in Brazil (Wendt et al. 2001) while probable self-incompatibility has been found in Aechmea distichantha Lem. var. distichantha, A. fasciata (Lindl.) Baker var. fasciata, Billbergia pyramidalis (Sims) Lindl. var. pyramidalis, Quesnelia arvensis (Vell.) Mez, Q. lateralis Wawra, and Q. marmorata (Lem.) Read (Martinelli 1994).
Gardner (1982) reported protogyny for almost all the 150 species of Tillandsia subgenus Tillandsia, including T. streptophylla Scheidw. On the other hand, Martinelli (1994) reported protandry for all of the seventeen bat-pollinated, unrelated Vriesea species inhabiting the rain forest in Eastern Brazil. Bush and Beach (1995) suggested that epiphytism generally favors autogamy, as well as Madison (1979) who suggested that ant-garden dwelling epiphytes usually set self-seeds, which is probably true in some species of Aechmea (A. mertensii (G. Mey.) Schult. & Schult. f., A. tillandsioides (Mart. ex Schult. & Schult. f.) Baker, A. bracteata (Sw.) Griseb., A. brevicollis L.B. Sm., etc.). However, it is well documented that many orchid and also several bromeliad species, feature mechanisms promoting cross-pollination.
Andromonoecy has been reported for Cryptanthus subgenus Cryptanthus (Ramírez-Morillo 1996), a Southeastern Brazilian group of terrestrial species; dioecy has been reported for the genus Hechtia (however, there is a poligamomonoecious species), for the only species of Androlepis Brongn ex Houllet (A. skinneri (K. Koch) Brongniart), for Aechmea Ruiz & Pav. (A. maria-reginae H. Wendland), and in several species and Mexican populations of Catopsis (Palací 1997). Thus dioecy exists then in the three subfamilies (Tillandsioideae, Bromelioideae and Pitcairnioideae) and in terrestrial as well as in epiphytic plants, suggesting several independent evolutionary events in this family.
Predominantly dioecious clades of Bromeliaceae concentrate in Mexico and Mesoamerica. Cleistogamy has been reported for Tillandsia capillaris Ruiz & Pav. (Gilmartin & Brown 1985), a species in subgenus Diaphoranthema from Argentina, Bolivia, Chile, Uruguay, and Peru, based on studies of floral biology, anther morphology, and pollen germination on the stigma.
Self compatibility in the Bromeliaceae was first suggested by Ule (1896, 1898) when describing corollas that remained closed in flowers of Aechmea, Nidularium Lem. and Quesnelia Gaudich. It was eventually discovered that these closed flowers were not cleistogamous but rather cross-pollinated by insects that penetrate the floral tissue (Gilmartin & Brown 1985). It is also well known that many bromeliads produce fruits under cultivation without any assistance by pollinators, a clear evidence of self-compatible species.
Self incompatibility in the family has been reported for Ananas comosus (L.) Merrill and the proposed mechanism is homomorphic gametophytic self incompatibility (Brewbaker and Gorrez 1987). It has also been reported for species of Billbergia Thunb. subgenus Billbergia based on the fact that they do not produce self-seeds (Benzing et al. 2000). Canela-Ferreira and Sazima (2003) reported self-incompatibility for Aechmea pectinata Baker, a hermaphroditic homogamic species, inhabitant of the rainforests in southeastern Brazil.
The aim of this contribution is to study the floral morphology and biology, and mating system of Tillandsia streptophylla, a bromeliad with high horticultural value. An objective of this research is to test the hypothesis put forward by Gardner (1986) suggesting that the species has protogynous flowers, with nocturnal or crepuscular anthesis and with flowers maturing before dawn (Gardner 1982).
Tillandsia streptophylla is known to occur in southern Mexico (Campeche, Chiapas, Oaxaca, Quintana Roo, Veracruz, Tabasco, and Yucatan) ranging southward into Belize and Nicaragua, also in Jamaica. It grows in a variety of vegetation types, such as low inundated forests, tall evergreen forests, low caducifolious forests, and mangroves.
Plants are epiphytic and usually produce small pups at the base of the flowering rosette but rarely form large clumps in the wild as compared to the large specimens frequently seen when under cultivation. Leaves are covered both sides with a dense, white indument. The foliar sheaths overlap while the laminae curl forming a pseudobulbose rosette inhabited by several ant species (Suárez 2005). Inflorescence is central, a 1-pinnate erect panicle, with pink-white lepidote bracts, flowers have a light purple or lilac corolla while the stamens and stigma are exerted. It is well known that, as opposed to many other species in subgenus Tillandsia, isolated plants of this species do not produce fruits in cultivation.
Materials and methods
Study site: We studied the floral morphology and biology and evaluated the breeding systems of a population of T. streptophylla in a natural habitat, at the Ría Celestún Biosphre Reserve. Voucher specimens are deposited at CICY (Ramírez-Morillo et al. 817A). The research was conducted during the peak of two flowering seasons in May 2000 and May 2001. The area is located in the northwestern section of the Yucatan Peninsula, between the States of Campeche and Yucatan, about 80 km SSW of the city of Merida (20°48-20°58 N, 90°1590°25 W, 2 masl). The Yucatan Peninsula is geologically a giant slab of Quaternary limestone substrate that imposes a carstic hydrography and topography. The climate is tropical hot and subhumid, with most precipitation occurring during summer for an annual total of ca. 760-800 mm. Annual mean temperature averages ca. 26 °C (Balam Ku et al. 1999). The dry season occurs between March and mid May while the rainy season is from late May to mid October. Between the rainy and the dry season (October to February) there is a cooler period called "nortes". These "nortes" often reach ca. 80 km/h and as they bring rains, they make an important contribution to the average annual precipitation (Gómez-Pompa et al. 1995, Orellana et al. 1999).
At the Ría Celestún Biosphere Reserve there are at least eight vegetation types. These include low caducifolious forest, low inundated forest, mangrove, coastal shrubland, inundated grassland, marine grassland, floating and emergent hydrophyte community, and "petén" [CONANP-SEMARNAT 2000]. Here, T. streptophylla populations occur in low inundated forests and transition to mangrove associations, with an arboreal substrate composed by species such as Ehretia tinifolia L. (Boraginaceae), Gymnopodium floribundum Rolfe (Polygonaceae), Metopium brownei (Jacq.) Urb. (Anacardiaceae), Manilkara zapota (L.) P. Royen (Sapotaceae), Bursera simaruba (L.) Sarg. (Burseraceae), Ceiba aesculifolia (Kunth) Britten & Baker f. (Bombacaceae), Cochlospermum vitifolium (Willd.) Spreng. (Cochlospermaceae), Conocarpus erectus L. ("mangle botoncillo", Combretaceae), and shrubs such as Dalbergia glabra (Mill.) Standl. (Fabaceae), Bonellia macrocarpa (Cav.) B. Ståhl & Källersjö (Theophrastaceae), Bravaisia berladieriana (Nees) T.F. Daniel (Acanthaceae), Helicteres baruensis Jacq. and Malvaviscus arboreus Cav. (Malvaceae), Randia longiloba Hemsl., R. aculeata L. (Rubiaceae), and Croton spp. (Euphorbiaceae). The height of the trees is 5-7 m and shrubs reach ca. 2 m high close to the mangrove. Low inundated forests are unique vegetation types restricted to the Yucatan Peninsula and not found elsewhere in Mexico.
Epiphytes in these communities include Catasetum integerrimum Hook., Encyclia alata (Bateman) Schltr., E. guatemalensis (Klotzsch) Dressler & G.E. Pollard, E. nematocaulon (A. Rich.) Acuña, Laelia rubescens Lindl., Cohniella cebolleta (Jacq.) Christenson, Lophiaris andrewsiae R. Jiménez & Carnevali, Myrmecophila christinae Carnevali & Gómez-Juárez, Notylia orbicularis A. Rich & Galeotti ssp. orbicularis (Orchidaceae), Aechmea bracteata, Tillandsia balbisiana Schult. f., T. dasyliriifolia Baker, and T. brachycaulos Schltdl. (Bromeliaceae)
Procedure: Flowering phenology of the species was first assessed analyzing information at vouchers at herbarium CICY, and then it was evaluated in the study site (during 2000 and 2001). In situ, marked reproductive plants (n= 83) were observed every two months, recording floral buds, open flowers, fruits, and seed release as well as the non reproductive phase, for a total of 12 mo recorded.
Number of flowers per inflorescence, number of flowers open per day per inflorescence and inflorescence dimensions were evaluated in situ (n= 12 plants). Floral morphology was evaluated in fixed (ethanol 70% and 5% glycerin) flowers (n= 30) collected in the field and measured in the lab with the aid of a stereoscopic microscope Nikon SMZ800 at different magnifications.
Floral biology was studied also in situ during two flowering periods; anthesis was recorded by observation of (n= 30) open flowers in (n= 8) plants every 30 min for 24 hours during each flowering season, to determine the times when stigma was receptive (secretion of sticky to substance) and pollen was mature (sticks to the fingers) and morphological changes associated with anthesis. Floral longevity, i.e., time a flower remains open, was estimated for three days each flowering season for 24 hours by direct observations of flowers (n= 30) and considered to start when the corolla opens and finish when it gets flaccid, anthers retract and stigma becomes flaccid as well. Floral fragrance emission and nectar volume were estimated in bagged flowers (n= 15); nectar volume was quantified in flowers (n= 15) at sunset by micropipetes and total sugar concentration was measured with a hand refractometer (Atago N-1E, Brix 0~32%).
Pollen viability was estimated in the lab assessing the percentage of germinated pollen grains in a media prepared with sacarose (15 g), H3Bo3 (0.010 g), Ca(NO3)2-4H20 (0.0086 g), MgSO47H2O (0.004 g), KNO3 (0.002 g) and agar (1 g), in 100 ml of distilled water. In this media fresh pollen from six anthers from different flowers was dispersed and then incubated in an oven at 30 °C for 24 hours. Then, the preparation was colored with 0.5 ml of dye (ethyl alcohol 25%, 0.03 g of aniline blue and 0.02 g of basic fuchsine), counting germinated pollen grains out of 100 randomly selected.
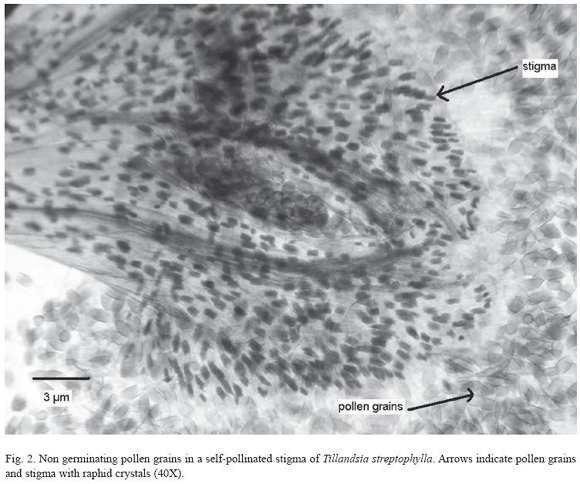
As a further test of compatibility, pollen tube germination on the stigma of self pollinated and cross pollinated flowers was analyzed. This was performed by collecting manual self and cross pollinated flowers at the greenhouses and fixing them for 12 hours after pollination in a solution of 95% ethanol lactic acid, staining with anyline blue 1% in lactophenol (DSouza 1972). Then, we assessed pollen germination on stigmas under a Nikon steresocope SMZ800; digital photographs were taken with a Samsung Camera at 40x magnification.
Mating system was investigated using the methodology of Ramírez and Seres (1994) performing controlled experimental crosses in the field. We used a total of 1 205 flowers on 83 plants to carry out all controlled crosses. A total of five different crosses were performed to detect (AG) agamospermy, (SP) manual selfing, (SS) spontaneous selfing (SS), cross pollination (CP), and (NP) natural pollination. Also, we included a setting to detect anemophily (AN) by emasculating flowers in bud and leaving them covered with a bag that allowed the passage of air and pollen but not insects nor birds. All fruit production was evaluated every two weeks for 1-3 months after pollinations until fruits were mature and beginning to open. Mature fruits were collected to quantify seeds and to carry out seed germination tests. To test seed germination and evaluate seed viability and abortion, seeds were germinated in closed Petri dishes with humid filter paper on bottom; 100 seeds randomly selected from each treatment were placed in the dishes; dishes then were incubated under controlled temperature and humidity (32 °C, light cycle 12x12 and 70% of relative humidity) for two months. Then, germinated seeds were recorded calculating the percentage of germination.
Results were analyzed using a xi2 in order to assess whether there were differences between the amounts of viable seeds among controlled crosses. In order to test which cross(es) produced the largest amount of viable seeds (or the minimum number of aborted seeds), a LSD test of media comparison was performed.
Reproductive strategy was determined following the indices described by Ramírez and Brito (1990). Self-compatibility index (SCI) was estimated by dividing the number of fruits or seeds produced in self-pollinated (SP) flowers by the number of fruits or seeds from cross-pollinated (CP) flowers. Values between 0.30–1 are considered as self-compatibility of partially self-compatibility, values below 0.30 are considered self-incompatibility. The self fertility index (Lloyd and Shoen 1992) was calculated by dividing the amount of fruits or viable seeds produced in spontaneous selfing (SS) by those in cross pollination (CP). Thus, it measures the ability of flowers to self seeds in the absence of pollinators. Values between 0.30-1 were considered as self fertility of partially self fertility and below 0.30 were considered as self-infertility. Indices calculated by using both fruits and fertile seeds could indicate inbreeding effects at different levels in the hierarchy of reproductive output (Jacquemart 1996).
Pollinator efficiency was estimated by dividing the number of fruits or viable seeds on natural pollination by the same parameters on manipulated cross pollination. Thus, it measures if there is pollinator shortage or poor pollinator activity in the natural population.
Results
Plant species: T. streptophylla is an epiphytic herb, growing at low and medium elevations (2-20 m) above the forest floor. Plants are rosetofilous, with sympodial growth with each ramet monocarpic, forming small to medium-sized clumps of several rosettes, on tree branches. The base of the rosette is formed by inflated leaf sheaths and partially by the bases of the foliar blades, forming several cavities (domacia) inhabited by several species of ants (Suárez 2005). The foliar blades are twisted and more or less recurved, giving the plants an overall aspect of silvery octopuses (Fig. 1). The leaves and floral parts are all covered by a dense white trichome layer, and the plant looks silvery or sometimes whitish, especially when it is dry. Population density is usually low (ca. 200 individuals per ha) and individuals are found at different maturity stages, such that we find seedlings and mature blooming plants in the same population.

Populations in the study site initiate blooming in March and continue until August with a flowering peak during May with some individuals blooming in November. Flowers are hermaphroditic and borne in a paniculate (1-divided) inflorescence, rarely erect, usually arching to pendulous especially when the plant is growing upside down, with a conspicuous scape (18.17±1.94 cm long), usually red or pink, densely pubescent and concealed by pink bracts. The inflorescence has a fertile portion (14.77 ±4.75 cm long, 82.83 ±35.2 flowers per inflorescence; 2.83 ±1.7 flowers open per day/inflorescence). Flowers are actinomorphic, with a tubular light violet corolla (4.16 ±0.02 cm long), three free petals and sepals, petals 2.5 ±0.1 cm longer than sepals, corolla remains straight during anthesis, with exerted sexual organs, six stamens in two series (4.3 ±0.38 cm the shortest and 4.8 ±0.2 cm the largest one), concolorous with the petals, a white, conduplicate spiral stigma in an elongated, white style 6.04 ±0.06 cm long.
Each branch of the inflorescence has 3-4 basal sterile floral bracts. The flowers protrude from the floral bract and open the night before around 1700-1900 hours; anthers release pollen from 0500 hours until 1200 hours and stigma becomes receptive around 0900 hours until 1900 hours, with some receptive almost until 2100 hours, thus stigma and anthers on a same flower are simultaneously in anthesis for at least ca. 3 hours. However, flowers are herkogamous, since the stigma is located above the anthers and separated by 1.32 ±0.03 cm. Nectar is produced inside the corolla tube and volume varies between 8-15 µl with a total sugar concentration of 25±1.66 °Brix. Pollen viability resulted of 48.5 ±1.51%.
Pollinators (hummingbirds) were observed visiting flowers during anthesis time but were not captured. In open flowers of T. streptophylla, only male individuals of the hummingbird Amazilia yucatanensis were observed, visiting all open flowers in an inflorescence and traveling to the next plant in flower.
Fruits were only produced on manual cross and natural pollinations, indicating that the species reproduce by cross pollination only since the manual, spontaneous selfing, agamospermy, and anemophily treatments did not yield any fruits (Table 1). Statistical analysis show that there is no difference between the amount of fruits produced by natural pollination and manual cross-pollination (F(1.175)= 0.08; p< 0.7827) as well as no differences between these same crosses regarding the amount of viable seeds.
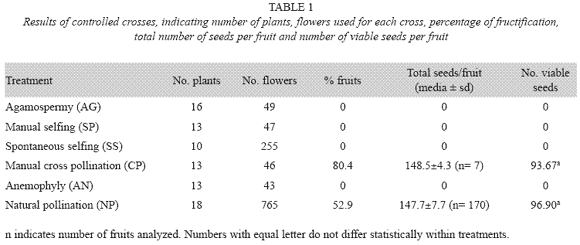
Self compatibility (SP/CP) and self fertility indexes (SS/CP) for fruits as well as for viable seeds were equal to zero indicating that the species is self incompatible. Pollinator efficiency calculated by fruit percentage in natural pollination is equal to 0.65 indicating pollinator limitations; but when calculated with total amount of seeds and with viable seeds resulted in 0.99 and 1.03 respectively, indicating good pollen transfer, fertilization, seed production, and seed viability.
A further test for compatibility was carried out by observation of pollen germination (tube formation) in stigmas of hand self pollinated flowers and hand cross pollinated flowers. Pollen did not germinate on stigmas of the same flowers (Fig. 2) but external pollen (in the hand cross-pollination treatments) germinates on the stigma of a different flower.
Discussion
T. streptophylla belongs to the subfamily Tillandsioideae and subgenus Tillandsia, characterized by hermaphrodite flowers with sexual organs protruding above the corolla tube and erect petals. The floral morphology and biology of T. streptophylla suggest pollination by hummingbirds, as it was also indicated by the presence of males of Amazilia yucatanensis visiting flowers of the species in the study site during the day. Gardner (1982) also reported hummingbirds visiting flowers of the species in Mexico.
Gardner (1982) suggested that protogyny occurs almost without exception in Tillandsia subgenus Tillandsia, including T. streptophylla. We observed flowers of the species opening ca. 0300 hours and remaining so until dawn, with anthers starting to deliver pollen at ca. 0500 hours while the stigma becomes receptive ca. 0900 hours until 1900 hours (partial dichogamy) with both structures well separated. Gardner (1982) proposed that anthers eventually reach the stigma by elongation of the filaments, so by the end of the day self-pollination occurs, however the lack of fruit production in our crosses of spontaneous-selfing does not support Gardners proposal.
Gardner (1984) reported pollen fertility data for ca. 46 species in Tillandsia with values ranging between 80-90% for most of them, although a few species featured values as low as 7.6% (e.g. Tillandsia erubescens Schltdl.) or even zero for some putative hybrids. Pollen viability in our populations of T. streptophylla resulted on 48.5 ± 1.51% as compared with the values published by Gardner (1984) for the same species (96.3%). This suggests variation among populations within the species probably involving the use of different reproductive strategies under different conditions. Tests of self pollen on stigmas to test self-incompatibility (no germination of pollen) is a case of sporophytic self incompatibility where pollen and stigma cells interact and inhibit the growth of pollen tube avoiding self fertilization. The second type of self incompatibility (homomorphic gametophytic) has been reported only for Ananas comosus (Brewbaker & Gorrez 1987).
Results of our studies indicate that this hermaphroditic species is self incompatible and only produces fruits by cross pollination, being individuals of the hummingbird A. yucatanensis the most likely pollen vectors.
A comparison between the results of manual cross-pollination and natural pollination indicates that the populations of T. streptophylla in our study site suffer of pollinator limitation, since more fruits were produced by manual cross-pollination than by natural pollination. In fact, despite the relatively high density of plants in bloom, there was only one recorded event of a pollinator visiting flowers of T. streptophylla during two days of work, accounting for almost 12 hours of observations. Pollen viability is relatively low (ca. 49%) for T. streptophylla which is probably standard for self incompatible species. On the other hand, self compatible species such as several Pitcairnia spp. (Wendt et al. 2001) feature percentages between 63-90% of pollen viability.
Variable floral morphology in Bromeliaceae suggests the presence of different breeding systems. Nevertheless, most of the reported breeding systems in the family correspond to hermaphroditic species with self compatible systems, suggested by the fact that sexual organs become in contact with changes in corolla shape during the flower maturation process (non dikogamous, not herkogamous species). Self incompatibility has only been reported for A. comosus (Brewbaker & Gorrez 1987), and suggested for Tillandsia deppeana Steud. (García-Franco 1990) a conclusion based on the fact that no fruits were produced in non-assisted selfing while there was only 7.89% of fruit set in geitonogamy experiments. However, García-Franco suggested that the low fruit production could be the result of immature sexual organs (anthers without pollen available and non receptive stigma) during manipulation. Here we suggest that it could also be a case of herkogamy or dichogamy, preventing selfing. Ramírez-Morillo et al. (2004) reported that T. dasyliriifolia Baker, set fruits by unassisted selfing and cross pollination, indicating that some species relay upon more that one breeding system.
Gardner´s (1986) classification of most species of subgenus Tillandsia divides it in five groups, placing T. streptophylla in group I, the largest one with ca. 60 taxa, which are supposedly mostly self-pollinated since the filaments often elongate after the anthers mature thus contacting the stigma. Gardner also suggested hummingbirds as the most likely pollinators. Within group I, Gardner recognized eight subgroups, and T. streptophylla is placed in subgroup 5 along with species characterized by presenting features of xeric species such as coriaceous leaves, foliar sheaths usually inflated and forming partially hollow pseudobulbs with ants often inhabiting them. McWilliams (1974) suggested that several of the species that Gardner later placed in subgroup 5 (T. butzii Mez, T. caput-medusae E. Morren) were self-fertile, a conclusion arrived at based only on floral morphology without further tests. Our study strongly indicates that flowers of T. streptophylla do not self pollinate, since bagged flowers (for spontaneous or unassisted selfing) did not yield fruits. Likewise, fruits were not produced on manipulated selfed flowers, a strong indication of self incompatibility.
The occurrence of self incompatibility in T. streptophylla does not support Gardners hypothesis (which was based only on floral morphology) suggesting that there are factors other than those related to floral morphology, such as floral biology involved in the breeding system.
T. streptophylla is a very peculiar species. While placed in subgenus Tillandsia or Group I subgroup 5 by Gardner (1986), it seems that the species does not have any closely related known taxon. The plant architecture, the thick and conspicuous white indumentum, the inflorescence of red rachis and pale pink primary bracts with green floral bracts, the light purple color of the corolla with concolorous filaments, all make this species unique. Furthermore, the presence of self incompatibility is for the first time reported for the genus. Nevertheless, Garth (1964) reported that in bagged flowers of Tilllandsia usneoides (L.) L. there were no developing pollen tubes on four out of eight flowers examined with self pollen, but fruit developing was very poor.
The results of this study raise an important point about the conservation status of the species, an over collected one and restricted to few habitats at low population densities. T. streptophylla is a very conspicuous and interesting species by its ornamental value. In the study site, it grows at low elevation from the floor level in several tree species, such as C. erectus, A. germinans (L.) L., Ehretia tinifolia and G. floribundum, and it takes many years to reach reproductive size. Under high humidity conditions, the genet can be conformed by several rosettes or ramets. The species is locally known as "x-mulix" (Mayan, meaning twisted or curled) or by "x-jolom wool" (empty and round).
The species is locally known by its use on traditional medicine (to treat colds and headaches) and it has been a species heavily collected for its ornamental value. This entire situation, added to the fact that the species grows at low populations densities, with a very slow growth rate as many epiphytic bromeliads with CAM metabolism, and the self incompatibility factor linked to the pollinator scarcity, place this species is high risk.
Acknowledgments
We thank Anahí López for her help in staining stigma of pollinated flowers; and Lilia Can for her help in searching the herbarium data-base.
Resumen
Estudiamos la fenología floral y el sistema de cruzamiento de la bromelia Tillandsia streptophylla (Bromeliaceae) en una selva baja inundable en Yucatán, México. Durante la estación de floración (marzo a agosto), las plantas producen una inflorescencia terminal, escaposa, paniculada, 1-dividida, con una flor abriendo por rama por día para un período de floración de 11-29 días por inflorescencia. Las flores son tubulares, de corola violeta claro, con el estigma y anteras exertos, pero las anteras más largas que el estigma en antesis. Las flores son protandras, con las anteras liberando el polen desde las 0500 horas y la receptividad del estigma comenzando a las 0900 horas. Los cruces experimentales controlados sugieren que Tillansdia streptophylla es auto incompatible y por ende, dependiente de los polinizadores.
Palabras clave: Bromeliaceae, conservación, sistema de cruzamiento, Península de Yucatán.
Received 20-XI-2008. Corrected 22-II-2009. Accepted 25-III-2009.
References
Balam Ku, M., I. Bañuelos Robles, E. García de Miranda, J. A. González-Iturbe, F. Herrera Cetina, R. Orellana Lanza & J. Vidal López. 1999. Evaluación Climática, p. 163-182. In A. García de Fuentes, J. Córdoba y Ordóñez, J. Chico Ponce de León (eds.). Atlas de Procesos Territoriales de Yucatán. Facultad de Arquitectura, Universidad Autónoma de Yucatán-CONACYT, Mexico. [ Links ]
Benzing, D.H., H. Luther & B. Bennet. 2000. Reproduction and life history, p. 245-328. In D.H. Benzing (ed.). Bromeliaceae: Profile of an adaptive radiation. Cambridge University, London, UK. [ Links ]
Brewbaker, J.L. & D.D. Gorrez. 1987. Genetics of self-incompatibility in the monocot genera Ananas (pineapple) and Gasteria. Am. J. Bot. 54: 611-616. [ Links ]
Bush, S.P. & J.H. Beach. 1995. Breeding systems of epiphytes in tropical montane wet forest. Selbyana 16: 155-158. [ Links ]
Canela-Ferreira, M.B. & M. Sazima. 2003. Aechmea pectinata: a hummingbird-dependent bromeliad with inconspicuous flowers from the rainforest in South-Eastern Brazil. Ann. Bot. 92: 731-737. [ Links ]
CONANP-SEMARNAT. 2000. Programa de manejo Reserva de la Biosfera Ría Celestún, México. Consejo Nacional de Áreas Naturales Protegidas, México. [ Links ]
DSouza, I. 1972. Staining pollen tubes in the styles of cereals with cotton blue: fixation by ethanol-lactic acid for an enhanced differentiation. Stain Technol. 47: 107-108. [ Links ]
De Faria, A.P.G., T. Wendt & G.K. Brown. 2004. Cladistic relationships of Aechmea (Bromeliaceae, Bromelioideae) and allied genera. Ann. Mo. Bot. Gard. 91: 303-319. [ Links ]
Espejo-Serna, A., A.R. López-Ferrari, I. Ramírez-Morillo, B.K. Holst, H. Luther & W. Till. 2004. Checklist of Mexican Bromeliaceae with notes on species distribution and levels of endemism. Selbyana 25: 33-86. [ Links ]
García-Franco, J.G. 1990. Biología reproductiva de Tillandsia deppeana Steudel (Bromeliaceae). M. Sc. Thesis, Universidad Nacional Autónoma de México, Mexico D.F, Mexico. [ Links ]
Gardner, C.S. 1982. A systematic study of Tillandsia subgenus Tillandsia. Ph.D. Thesis, College Station, Texas A & M University, Texas, USA. [ Links ]
Gardner, C.S. 1984. Natural hybridization in Tillandsia subgenus Tillandsia. Selbyana 7: 380-393. [ Links ]
Gardner, C.S. 1986. Inferences about pollination in Tillandsia (Bromeliaceae). Selbyana 9: 76-87. [ Links ]
Garth, R.E. 1964. The ecology of the Spanish moss (Tillandsia usneoides), its growth and distribution. Ecology 45: 470-481. [ Links ]
Gilmartin, A.J. & G.K. Brown. 1985. Cleistogamy in Tillandsia capillaris (Bromeliaceae). Biotropica 17: 256-259. [ Links ]
Gómez-Pompa, A., R. Dirzo, B.H. Fernández & E.G. Becerra. 1995. Las Reservas de la Biósfera y otras Áreas Naturales Protegidas de México. SEMARNAP, INE. CONABIO, Mexico. [ Links ]
Jacquemart, A.L. 1996. Selfing in Narthecium ossifragum (Melianthaceae). Plant Syst. Evol. 203: 99-110. [ Links ]
Lloyd, D.G. & D.J. Shoen. 1992. Self- and cross-fertilization in plants. I. Functional dimensions. Int. J. Plant Sci. 153: 358-369. [ Links ]
Luther, H.E. 2006. An alphabetical list of bromeliad binomials. The Bromeliad Society Internacional, Florida, USA. [ Links ]
Madison, M. 1979. Additional observation of ant-gardens in Amazonas. Selbyana 5: 107-115. [ Links ]
Martinelli, G. 1994. Reproductive biology of Bromeliaceae in the Atlantic rain forest of Southeastern Brazil. Ph.D.Thesis, University of St. Andrews, Scotland. [ Links ]
McWilliams, E.L. 1974. Evolutionary ecology. In L.B. Smith & R.J. Downs (eds.). Bromeliaceae (Pitcairnioideae). Flora Neotropica Monograph No. 14. Hafner, New York, USA. [ Links ]
Palací, C.A. 1997. A systematic revision of the genus Catopsis (Bromeliaceae). Ph.D. Thesis, University of Wyoming, Laramie, Wyoming, USA. [ Links ]
Ramírez-Morillo, I. 1996. Systematics, phylogeny, and chromosomal evolution of Cryptanthus (Bromeliaceae). Ph.D. Thesis, University of Missouri-St. Louis. St. Louis, Missouri, USA. [ Links ]
Ramírez-Morillo, I., G. Carnevali Fernández-Concha & F. Chi-May. 2004. Portraits of Bromeliaceae from the Mexican Yucatan Peninsula-IV: Tillandsia dasyliriifolia Baker: Taxonomy and reproductive biology. J. Brom. Soc. 54: 112-121. [ Links ]
Ramírez, N. & Y. Brito. 1990. Reproductive biology of tropical palm swamp community in the Venezuelan Llanos. Am. J. Bot. 77: 1260-1271. [ Links ]
Ramírez, N. & A. Seres. 1994. Plant reproductive biology of herbaceous monocots in a Venezuelan tropical cloud forest. Plant Syst. Evol. 190: 129-142. [ Links ]
Smith, L.B. 1986. Revision of the Guayana Highland Bromeliaceae. Ann. Mo. Bot. Gard. 73: 689-721. [ Links ]
Suárez, H.D. 2005. Forrajeo y anidamiento de hormigas (Hymenoptera: Formicidae) en epífitas mirmecófilas de Rio Hondo, México. Tesis de Licenciatura, Instituto Tecnológico de Chetumal, Quintana Roo, Mexico. [ Links ]
Ule, E. 1896. Ueber Bluthenverschluss bei Bromeliaceen mit berucksichtigung der Blutheneinrichtungen der ganzen familien. Berichte der Deutschen Botanischen Gessellchaft 14: 407-422. [ Links ]
Ule, E. 1898. Weiteres uber Bromeliaceen mit Bluthenverschluss und Bluthenein-richtungen dieser Familie. Berichte der Deutschen Botanischen Gessellchaft 16: 346-362. [ Links ]
Wendt, T., M.B. Ferreira Canela, A.P. Gelli de Faria & R. Iglesias Ríos. 2001. Reproductive biology and natural hybridization between two endemic species of Pitcairnia (Bromeliaceae). Am. J. Bot. 88: 1760-1767. [ Links ]












 uBio
uBio 

