Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.57 no.3 San José sep. 2009
Diversity and distribution of vascular epiphytes in an insular Brazilian coastal forest
Rodrigo de Andrade Kersten1, Marília Borgo2 & Sandro Menezes Silva3
1. Pontifícia Universidade Católica do Paraná, CCBS, Herbário HUCP, Rua Imaculada Conceição 1155, CEP 80215901, Curitiba, Paraná, Brazil; kersten@pop.com.br
2. Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental (SPVS), R. Isaias Bevilacqua, 999, CEP 80430-040, Curitiba, Paraná, Brazil; maborgo@yahoo.com
3. Conservation International Brazil (CI), Rua Paraná, 32, CEP 79020-290, Campo Grande, Mato Grosso do Sul, Brazil; s.menezes@conservacao.org
Abstract: The study was carried out in a 3 000m2 area of coastal Atlantic rain forest at Ilha do Mel island (25o30S 48o23W), on 100 assorted trees separated into 2 meter-high strata starting from the ground. In each stratum all of the occurring epiphytic species were recorded. The sampled species were grouped into three categories: exclusive, preferential, and indifferent, according to their abundance in each strata, and selective, preferential and indifferent, according to abundance on the forophytes. Intermediate strata registered the highest diversity. Six species were considered exclusive to one or two strata, 15 were restricted to some strata and 5 presented a broad distribution. No epiphytic species showed uniform horizontal distribution on the area. The epiphyte richness in a host tree varied from zero to 30. Regarding to fidelity on host tree species, few selective or preferential, and mainly indifferent epiphyte species, were found. A total of 82 epiphyte species were sampled in the surveyed tree, and the Wittaker plot indicate a highly dominant assemblage. Rev. Biol. Trop. 57 (3): 749-759. Epub 2009 September 30.
Key words: epiphyte ecology, spatial distribution, host preference, coastal Atlantic forest.
Stratification in tropical forests is closely related to the vertical environmental variance found from the forest floor to the canopy (Johansson 1974). This variation influences not only the distribution of plant but also of birds, mammals and insects, some of them exclusive to the canopy (Nadkarni 1994). Like any other kind of life, epiphytes are influenced by the forest. Temperature, moisture and light incidence, for instance, are important factors in distribution patterns definition (Benzing 1995).
The distributions of epiphyte depend on the forophyte species, age and characteristics such as shape, bark texture, branching patterns and height (Freiberg 1996). Also crown shape, size of leaves and canopy density, affect the total rain retention and rearrangement, which also influences the distribution of epiphytes (Parker 1995). In addition to environmental characteristics, intra and interspecific interac tions can also control the distribution patterns of some species (Yeaton & Gladstone 1982, Hietz & Hietz-Seifert 1995).
Distribution of epiphytes among different host species is related to factors such as moisture retention, chemical composition and bark morphology, which can be decisive on the establishing and developing of some species (Benzing 1990). The objective of this study was to describe the vascular epiphyte community, its spatial distribution patterns and composition on different host tree species.
Materials and methods
Study Site: The research was carried out at Estação Ecológica da Ilha do Mel, Ilha do Mel Island, southern Brazil (25º 30"S 48º 23W). This protected area consists of 2.241ha of coastal plain forest. The sampled area (3 000 m2), had its tree community structure (Silva and Britez 2005) and vascular epiphyte checklist (Kersten & Silva 2006) previously analyzed. Its semi-hydromorphic soil is flooded during the rainy seasons, the forest contains three main strata: lower stratum contains herbaceous plants, leaf litter and shoots, middle stratum with bushes and samplings, and the upper stratum (canopy) with only few species trees, which may reach over 20 m (Silva & Britez 2005).
Sampling: Ten forophyte individuals from each of the ten most important species were previously chosen: Tapirira guianensis (Anacardiaceae), Calophyllum brasiliense (Clusiaceae), Faramea marginata (Rubiaceae), Myrcia racemosa, Myrcia insularis and Eugenia sulcata (Myrtaceae), Ocotea pulchella (Lauraceae), Alchornea triplinervia and Pera glabrata (Euphorbiaceae) and Schefflera angustissima (Araliaceae). Acronyms were constructed with the first letter of the genera and the two firsts of the epithet. Due to high mortality rate only 8 individuals from this last specie were sampled. A total of 98 host trees were then surveyed. Canopy access was realized with an aluminum leather (7.5 m) associated with modified mountaineering technique and manual climbing. The names of the species and authors were checked at TROPICOS (2008) and the synonyms at World Cheklist of Selected Plant Families (2008).
Host tree bark aspect was classified following Ribeiro et al. (1999). As epiphyte individuals are not always discernible, we use the Kersten & Silva (2001) system of 2-meter-vertical division (strata) from ground to canopies, were every observed epiphyte species was recorded, young individuals were not considered.
Statistical analyses: The abundance of the species was evaluated by its frequency on the strata and on the forophytes. The total number of species registered on each stratum was added-up to get the number of observation per tree, with was considered analogous to dominance. A rank-abundance plot (Wittaker Plot - Magurran 2004) for the epiphyte species was elaborated using a log10 scale. The spatial distribution was considered to be the vertical (on strata) and horizontal distribution (on individual forophytes and on forophyte species).
To analyze vertical distribution patterns tree α-diversity index (alpha-diversity, Chaos estimator of the absolute number of species, Margalefs α-diversity index) and two evenness measures (Simpsons index and Shannon evenness measure) were considered. According to species distribution, the epiphytes were divided into groups:
1. On the Strata (vertical distribution):
- Exclusive: species observed only in one or two successive strata;
- Restrict: species with more than 50% of frequency in one stratum or more than 60% in two or three successive strata;
- Broad: when the total frequency of any three successive strata is less than 60%.
Only species observed more than 10 times (in number of forest strata) or more than 5 times (when restricted to a single stratum) were considered in this analysis.
Shannons diversity index (H), based on Waechter (1998), was calculated using the species abundance (number of times observed) on host trees.
2. On host trees species:
- Specialist – when found in only one species of host tree;
- Selective – when frequently found on few species of host trees, and rarely on other species (more than 40% on one species, more than 60% on two species, or 100% on three species);
- Preferential – when found with higher frequency on some host trees, but also with relevant frequency on other species;
- Indifferent - when found with similar frequency on the host tree species.
Spearmans correlations between number of species, abundance and host tree perimeter and height were analyzed, numbers higher than 0.6 when significant (α = 0.05) were considered to indicate associations.
The similarity of epiphyte communities on host tree species was evaluated by Ellenbergs similarity index (Mueller-Dombois & Ellenberg 1974), that was used to make a dendrogram (Ward) and by correspondence analysis using forophyte specie as x-factor and epiphyte frequency of species as response.
Results
Vertical Distribution: Intermediate strata registered highest diversity (Fig. 1 and 2). Six species (7%) were considered exclusive, tree to one stratum and tree to two strata, 15 (18%) were restricted to some strata and 5 (6%) presented a broad distribution (Fig. 2); 56 (69%) low frequency species were not classified.

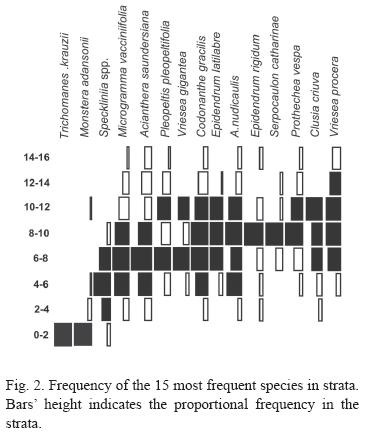
The species exclusive to one strata (Trichomanes krausii, Campyloneurum acrocarpon and Peperomia urocarpa) were all observed on the 0-2 m stratum and species exclusive to two strata (Vriesea gigantea, V. vagans and Pleopeltis hirsutissima) were found on the 6-8 m and 8-10 m strata. Four restrict species occurred more frequently in only one stratum: Monstera adansonii (0-2 m), Epidendrum rigidum and Serpocaulon catharinae (8-10 m), and Tillandsia tenuifolia (12-14 m). Eleven species (Microgramma vacciniifolia, Aechmea nudicaulis, Codonanthe devosiana, C. gracilis, Clusia criuva, Prosthechea vespa, Epidendrum latilabre, Pleurothallis spp, Acianthera saundersiana, Vriesea procera and Campyloneurum nitidum) were found mostly in two or three strata. Five species (19%) have broad distribution and occurred with similar frequency in many strata (Oncidium uniflorum, Peperomia glabella, Philodendron corcovadense and Scaphyglottis modesta).
Distribuition on forophytes: Vascular epiphytes were not regularly distributed on individuals or forophyte species. On individual host trees the number of epiphytes varied from zero to 30 epiphyte species (average of 6 ± 6.3). Highest richness was found at an Ocotea pulchella (30 species) and a Calophylum brasiliense (27 species). Other 17 individuals (six Calophyllum brasiliense, four Tapirira guianensis, two Ocotea pulchella, two Schefflera. angustissima, one Alchornea triplinervia, one Eugenia sulcata and one Pera glabrata) hosted 10 or more epiphytic species. In 11 host trees (five Myrcia insularis, two Pera glabrata, one Alchornea triplinervia, one Myrcia racemosa, one Faramea marginata and one Tapirira guianensis) no epiphyte were observed.
Different forophyte species also had different epiphyte richness and abundance. Calophyllum brasiliense supported the greater epiphyte community (55 species and 315 observations), followed by Ocotea pulchella (46 species and 193 observations), Tapirira guianensis (40 species and 127 observations) and Eugenia sulcata (37 species and 107 observations). Considering averages, the richness species were Calophyllum brasiliense (15 ± 7.3), Ocotea pulchella (9 ± 8.3), Tapirira guianensis (8 ± 4.5) and Eugenia sulcata (7 ± 5.4).
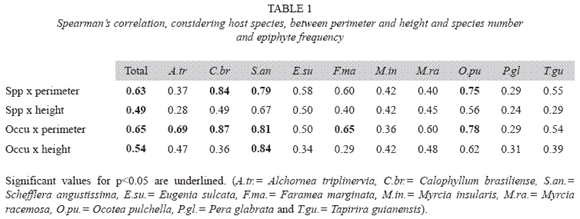
Epiphyte abundance was not always related to the size of the forophyte (Table 1). From 10 species, only Calophyllum. brasiliense, Schefflera angustissima and Ocotea pulchella the number of species and abundance were correlated to diameter; and in Alchornea triplinervia and Faramea marginata only the abundance. Considering host tree height only Schefflera angustissima had significant values.
Regarding fidelity, epiphytes were classified on three categories (selective, preferential and indifferents), none were classified as specialist, 58 low frequency species were not classified.
1) Selective –12 species in two subgroups:
a) On one forophyte species: Eight species, of which six were found more frequently on Calophyllum brasiliense: Clusia criuva, Acianthera saundersiana, Epidendrum rigidum, Tillandsia tenuifolia, Campyloneurum nitidum and Peperomia glabella (whith 80% of its abundance on this host tree species). Two other species (Scaphyglottis modesta and Vriesea vagans) were found more frequently on Tapirira guianensis.
b) On two forophyte species: Codonanthe gracilis and C. devosiana over Calophyllum brasiliense and Ocotea pulchella, and Pleurothallis spp over Eugenia sulcata and Myrcia racemosa.
2) Preferential - three species: Monstera adansonii, Pleopeltis pleopeltifolia and Trichomanes krauzii, all over Calophyllum brasiliense.
3) Indifferent - nine species: Microgramma vacciniifolia, Vriesea procera, Aechmea nudicaulis, Epidendrum latilabre, Prosthechea vespa, Serpocaulon catharinae, Vriesea gigatnea, Oncidium uniflorum and Pleopeltis hirsutissima.
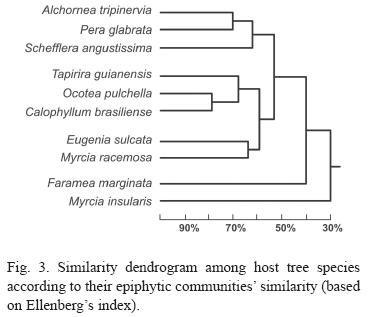
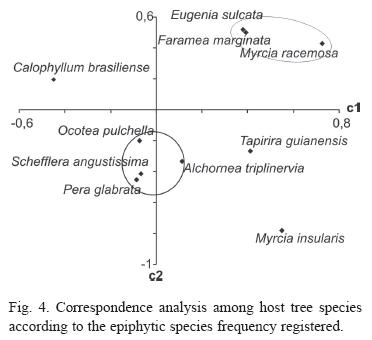
The similarities of the host tree species epiphyte communities are shown on figure 3. Clusters analysis formed three major groups. On the correspondence analysis (Fig. 4), the two fist exes explain only 38% of the variation, 95% of the variation is explained by the first 8 axes. Seven species formed two groups and tree species remained isolated.
Species Diversity and Abundance: We found 82 species in the 673 strata analyzed totalizing 995 registers. Shannons index (H) was 3.74, while equity (J) was 0.85. About 45% of the strata and 11% of host trees (11) showed no epiphyte.
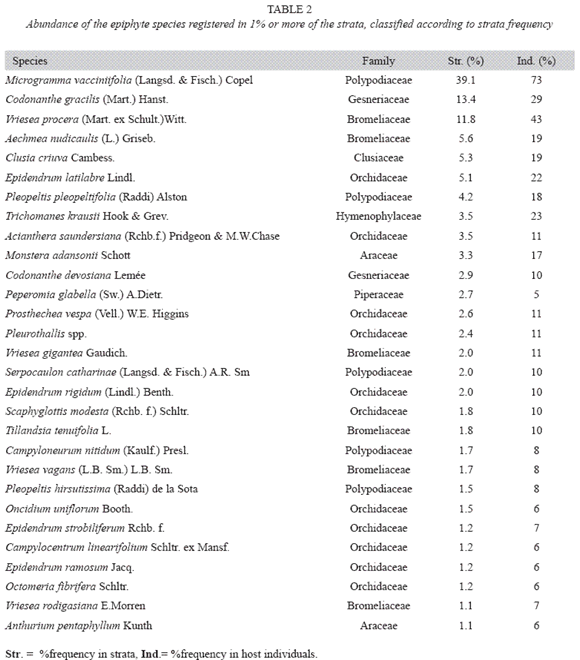
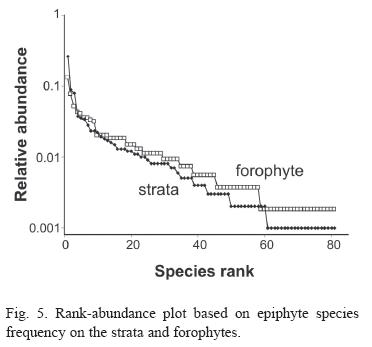
Considering species abundance (Table 2) only three species were registered to more than 10% of the strata and 18 to more than 10% of the forophyte. Thirty-three species were found only once or twice. The first four species (Microgramma vacciniifolia, Vriesea procera, Codonanthe gracilis and Epidendrum latilabre) are responsible for more than 30% of strata frequency; meanwhile the 35 least frequent species represent about 10% of total strata frequency. The morphospecie Pleurothallis spp. includes Pleurothallis seriata Lindl, Pleurothalli matinhensis Hoehne and Specklinia marginalis (Rchb.f.) F. Barros., which were very similar when not flowering. On the rank-abundance plot (Fig. 5), both slopes are indicative of log series distribution.
Discussion
Species Distribution: Unequal distribution along vertical column in forests (Figs. 1 and 2) is regularly explained by variation on moisture, luminosity, substrate availability substrate characteristics (Waechter 1980, 1992, Gill & Onyibe 1986, Steege & Cornelissen 1989, Zimmermman & Olmsted 1992, Kernan & Fowler 1995, Kersten & Silva 2001, 2002). Brown (1990) stated that variations of microclimate, and substrate diameter, are the main factors responsible for epiphyte distribution pattern. These studies also noticed highest epiphytic diversity in the intermediate strata. The exception is Kersten & Silva (2001) that registered the lower strata as the richest.
Taxonomic and ecological categories distribution is unequal along vegetal column (Fig. 2). While hemiepiphytes and facultative epiphytes occupy lower strata, holoepiphytes prefer intermediate and upper strata. Most pteridophyte families (Blechnaceae, Nephrolepidaceae, Hymenophyllaceae, i.e.) occurs more frequently close to the ground, while upper strata are occupied by angiosperms and two pteridophyte families – Polypodiaceae and Lycopodiaceae; curiously in evolutionary terms one is among the most derived families (Polypodiaceae) and the other among the most basal (Lycopodiaceae).
Gill & Onyibe (1986) considered ten important environmental factors to the establishment of epiphytic flora: host age, inclination, luminosity, humus deposition, host tree size, daily weather variation, seed dispersion, temperature, moisture and wind. Ingram & Nadkarni (1993) observed positive correlation between epiphytic biomass and branch size, and negative between biomass and branch inclination.
In this study, branch inclination and space availability along with luminosity and moisture explains the observed distribution pattern. Intermediate strata regularly have more branches and ramifications, and therefore larger areas available for colonization. At the same time, organic matter accumulation increase available water in places protected from direct sunrays incidence but with higher luminosity than forest floor.
In upper strata although the branche prufusion, they are thin and young. The sunlight level and stronger winds increase desiccation and difficult species development. In lower strata (0-6 m) moisture is sufficient, but lower light and strait vertical bark are straining. However, the 0-2m height stratum was richer than 2-4 strata (Fig. 1) due to accidental, facultative and hemiepiphytes species.
Two of the four species that preferred the 0-2 m stratum are facultative and another one is a primary hemiepiphyte closely related to soil. Though the T. krauzii is a typical holoepiphyte, its a Hymenophyllaceae, a very hydro-sensible family. Even though most species preferred intermediate strata, none or then were restrict to them, again indicating the space availability as an important aspect in epiphyte distribution. The one specie that really preferred higher strata (T. tenuifolia) is a well known atmospheric bromeliad, forming dense groups in unfavorable environments such as isolated trees and with high light exposition (Reitz 1983).
The broad distribution of some species usually comes along with significant morphological variation. In Orchidaceae there is a noticeable variation in succulence and size, in Bromeliaceae and Gesneriaceae, the individuals differ in color and size, the ones in higher strata are smaller, more succulent and present a reddish pigment. Also, stomata variations (Hietz & Briones 1998) are mentioned as related to habitat diversity in the same tree.
Maximum number of species over one single tree, although not exceptional (Johansson 1989, Waechter 1992, Freiberg 1996), can be considered high when compared to other field-works in similar areas. Kersten & Silva (2001) found 21 species over one tree. Waechter (1998; 1992) found 16 and 19 species respectively. The studied area, in spite of being humid, is a medium size forest (average height = 12m), and so is the epiphytic richness when compared to other forests. Johansson (1989) registered more than 50 species over one host tree that was higher than 30m. In the other hand, Kersten & Silva (2001) observed 21 species on a host tree that presented average height of 7.5m.
Distribuition on forophytes: The frequency of vascular epiphytes over different host trees can be explained by forophyte species traits, such as size, age, architecture, and bark type. Calophyllum brasiliense had the biggest individuals (180 cm of diameter and 20m high) has a deeply cracked bark. O. pulchella and T. guianensis has rough bark as well as favorable architecture for epiphyte fixation (great number of thick horizontal branches). Brown (1990) also observed that host trees with scabrous barks support more epiphytes. The forophyte species with the fourth highest epiphyte (E. sulcata) presents defoliating suber that do not totally detach from tree. Trunk and branches become recovered by many suber layers, which accumulate moisture and facilitate the fixation and development of epiphytes.
While these four species are suitable for epiphytic development, others like F. marginata and M. insularis are, otherwise, unfavorable for the development of this flora. Kernan & Fowler (1995), Zimmerman & Olmsted (1992) and Steege & Cornelissen (1989) presented similar results, indicating that the bark texture influences the epiphyte distribution, facilitating it (with rugged barks) or making it difficult (when totally loose). Zimmerman & Olmsted (1992) registered that 8% of host trees did not have any epiphyte (in contrast to 11% in this study). It is important to notice that this work was also realized in a seasonally flooded forest. In Brown (1990) 22% of trees did not have epiphytes, possibly due to seasonal climate.
Size and number of branches and ramifications influence epiphyte occurrence, as long as they increase the fixation area, host tree age is also important. As no relation between perimeter and quantity of epiphytes was observed to host tree species in this work, size is not a major factor influencing epiphyte distribution, though it can influences great part of the variation (Brown 1990, Zimmerman & Olmsted 1992, Ingram & Nadkarni 1993).
In opposition to Waechter (1980), the specificity analysis among epiphytes and host trees revealed no exclusive species. Steege & Cornelissen (1989) registered seven epiphyte species significantly more frequent on some host trees. Despite that, in this study, many epiphytes showed preference for some hosts, the greatest values were often registered to the same host species (C. brasiliense and O. pulchella), therefore little of epiphyte preference is due to internal traits, but hosts features (bark roughness, architecture, etc.) facilitate or not the establishment and development of this flora.
Two cases, however, deserve special attention: Peperomia glabella and Pleurothallis spp. In the first case, 78% of occurrences were registered on C. brasiliense. This may be explained by its facultative habit can being found either living on trees or the ground. Not being exclusively epiphytic it cannot grow in any substrate, but only in favorable trees.
In the second case, the group – Pleurothallis spp. – preferred Myrtaceae species with stripping bark (E. sulcata and M racemosa). Similar analysis can be realized in other areas to confirm if this is a real preference of the species for this type of bark or whether it is only a coincidence.
The analysis of epiphyte communities on host species was based on Ellenbergs index and correspondence. Both index indicated that relatively homogeneous groups exist (Fig. 3). The dendrogram group composed by C. brasiliense, O. puchella and T. guianensis was defined because these are the host tree species with more epiphyte. In the same way M. insularis and F. marginata were excluded from groups due to their low epiphytic level.
The two other groups perhaps are the most interesting. Presenting profuse abundance and richness, the group with defoliating suber (M. racemosa and E. sulcata) seems to indicate that this feature may be important to the epiphytes, influencing their distribution. The last group, specially the proximity between A. triplinervia and P. glabrata (71%), can also be explained by their bark similarity.
On the correspondences analysis the c1 axe does not represent the species diversity nor the abundance, even thought may seem to, due to Schefflera angustissima position next to Ocotea pulchella and Eugenia sulcata next to Faramea marginata and Myrcia racemosa. The two groups were formed by the proportional frequency of specific species on them and both support the dendrogram analysis.
Bennett (1986), with similar results, observed that epiphytic community was correlated to host tree species, but explained only 16% of variation, Dislich (1996) reported some epiphyte preferences for host species and Freiberg (1996) observed restricted epiphyte occurrences in some host species.
Species Diversity and Abundance: Considering the complete checklist (Kersten & Silva 2006) 80% of species 94% of genera and 94% of families were observed in the sampling. If we put together the 22 species observed only in the floristic survey and the 23 species registered once on this analysis, 45% of the species might be called rare. On the Wittaker Plot the log series distribution are indicative of high dominance assemblage with few species what seems to be the rule on atlantic forest epiphyte assemblage (Waechter 1992, 1998, Kersten & Silva 2001, Gonçalves & Waechter 2002, Giongo & Waechter 2004).
Microgramma vacciniifolia, was, by far, the most frequent one. Occurring widely in the Tropical America (Sehnem 1970) it was also abundant in other studies (Table 3). Therefore it can be considered the most abundant vascular epiphyte in coastal plain forests from south Brazil. Yet, on studies in seasonal forest (Dislich 1996) and Araucaria forest (subtropical rainforest) (Kersten & Silva 2002), another specie from the same genus (M. squamulosa) was the most frequent. This plasticity, showed by it geographic distribution, explains its occurrence in all host species, the abundance can be also explained by is creeping rhizomes that grows all over the host tree, increasing their frequency. At the same time, as a sporocoric fern it does not depend on mutualism to establish, as orchids do.
Codonanthe gracilis, has creeping or hanging stems what allows it to climb the host tree and also colonize lower branches and can be considered also typical of the Atlantic rain forest. According to Chautems (1997), this species is found in coastal regions from Bahia State (northeastern Brazil) to Rio Grande do Sul State (south Brazil), being mentioned in three other studies (Waechter 1986, Fontoura et al. 1997, Kersten & Silva 2001).
Resumen
El estudio se realizó en un área 3 000 m2 de bosque atlántico húmedo en una isla brasileña, Ilha do Mel (25o30 S 48o23 W). Seleccionamos 100 árboles de las 10 principales especies y las dividimos en estratos de dos metros a partir de la superficie del suelo. En cada estrato registramos todas las especies epífitas observadas. Las especies muestreadas fueron agrupadas en tres categorías: exclusivas, preferenciales, e indiferentes (según su abundancia en los estratos), y selectivas, preferenciales e indiferentes (según su abundancia en los árboles). Los estratos intermedios registraron las más altas diversidades. Seis especies fueron consideradas exclusivas a uno o dos estratos, 15 estuvieron limitadas a algunos estratos y 5 presentaron amplia distribución. Para ninguna especie epífita hubo distribución horizontal uniforme. La riqueza de las epífitas en un forofito varió de cero a 30 especies. Según la fidelidad de las epífitas en los árboles, registramos pocas especies selectivas o preferenciales y muchas indiferentes. En los 100 árboles examinados encontramos 82 especies de epífitas, y el diagrama del Wittaker indicó alta dominancia de unas pocas especies.
Palabras clave: ecología de epífitas, distribución espacial, bosque Atlántico costero.
Received 21-VIII-2008. Corrected 15-X-2008. Accepted 16-XI-2008.
References
Bennet B.C. 1986. Patchiness, diversity and abundance relationships of vascular epiphytes. Selbyana 9: 70-75. [ Links ]
Benzing D.H. 1990. Vascular epiphytes. Cambridge University Press. Cambridge. [ Links ]
Benzing D.H. 1995. The physical mosaic and plant variety in forest canopies. Selbyana 16: 159-168. [ Links ]
Brown D.A. 1990. El epifitismo en las selvas montanas del Parque Nacional "El Rey" Argentina: Composición florística y padrón de distribución. Rev. Biol.Trop. 38: 155-166. [ Links ]
Chautems A. 1997. New Gesneriaceae from São Paulo Brazil Candollea 52: 159-169. [ Links ]
Dislich R. 1996. Florística e estrutura do componente epifítico vascular na mata da reserva da Cidade Universitária "Armando de Salles Oliveira", São Paulo, SP. Dissertation, University of São Paulo. [ Links ]
Fischer E.A, Araujo A.C. 1995. Spatial organization of a bromeliads community in the Atlantic rainforest, southeastern Brazil. J. Trop. Ecol. 11: 559-567. [ Links ]
Fontoura T. 1995. Distribution patterns of five bromeliad genera in Atlantic rain forest, Rio de Janeiro State, Brazil. Selbyana 16: 79-93. [ Links ]
Fontoura T., Sylvestre L.S., Vaz A.M.S. & Vieira, C.M. 1997. Epífitas vasculares, hemiepífitas e hemiparasitas da Reserva Ecológica de Macaé de Cima, p. 89-101. In Lima H C, Guedes-Bruni R R (eds.) Serra de Macaé de Cima: diversidade florística e conservação da Mata Atlântica, Editora do Jardim Botânico do Rio de Janeiro. [ Links ]
Freiberg M. 1996. Spatial distribution of vascular epiphytes on three emergent canopy trees in French Guyana. Biotropica 28: 345-355. [ Links ]
Gentry A.H. & Dodson C.H. 1987. Diversity and biogeography of neotropical vascular epiphytes. An. Mo. Bot. Gard. 74: 205-233. [ Links ]
Gill L.S., Onyibe H.I. 1986. Phytossociological studies of epiphytic flora of oil palm (Elaeis guineensis Jack.) in Benin City, Nigeria. Feddes Repertorium 97: 691-695. [ Links ]
Giongo, C. & Waechter J.L. 2004. Composição florística e estrutura comunitária de epífitos vasculares em uma floresta de galeria na Depressão Central do Rio Grande do Sul. Ver. Brás. Bot. 27: 563-572. [ Links ]
Gonçalves C.N. & Waechter J.L. 2002. Epífitos vasculares sobre espécimes de Ficus organensis isolados no norte da planície costeira do Rio Grande do Sul: Padrões de abundância e distribuição. Acta bot. Bras. 16: 429-441. [ Links ]
Hazen W E 1966. Analysis of spatial patterns in epiphytes. Ecology 47: 634-635. [ Links ]
Hietz P., Briones O. 1998. Correlation between water relations and within-canopy distribution of epiphytic ferns in a Mexican Cloud Forest. Oecologia 114: 305-316. [ Links ]
Hietz P., Hietz-Seifert U. 1995. Intra and interspecific relations within an epiphyte community in a Mexican humid montane forest. Selbyana 16: 135-140. [ Links ]
Ingram S.W., Nadkarni N.M. 1993.Composition and distribution of epiphytic organic matter in a Neotropical cloud forest, Costa Rica. Biotropica 25: 370-383. [ Links ]
Johansson D.R. 1974. Ecology of vascular epiphytes in West African rain forest. Acta Phytogeographyca Suecica 59: 1-136. [ Links ]
Johansson D.R. 1989. Vascular epiphytes in Africa p. 183-194. In H. Lieth & M.J.A. Werger (eds.) Ecosystems of the world vol. 14b: Tropical Rain Forest Ecosystems. Amsterdam [ Links ]
Kelly D.L. 1985. Epiphytes and climbers of a Jamaican rain forest: vertical distributions, life forms and life history. J. Biogeog. 2: 233-243. [ Links ] Kernan C, Fowler N 1995, Different substrate use by epiphytes in Corcovado National Park, Costa Rica: a source of guild structure. J. Ecol. 83: 65-73. [ Links ]
Kersten R.A. 2006. Métodos quantitativos de estudo da flora epífita. In Os avanços da Botânica no início do séulo XXI ed. Mariath J.E.A. & Santos R.P. Sociedade Botânica do Brasil, Porto Alegre. [ Links ]
Kersten R.A., Silva S.M. 2001. Composição florística e distribuição espacial de epífitas vasculares em floresta da planície litorânea da Ilha do Mel, Paraná, Brazil. Rev. Bras. Bot. 24: 213-226. [ Links ]
Kersten R.A., Silva S.M. 2002. Florística e estrutura do componente epifítico vascular em floresta ombrófila mista aluvial do rio Barigüi, Paraná, Brasil. Rev. Bras. Bot. 25: 259-267. [ Links ]
Kersten R.A., SILVA S.M. 2005. Florística e estrutura de comunidades de epífitas vasculares da planície litorânea. In História natural e conservação da Ilha do Mel, eds. Marques M.C.M. & Britez R.M. Ed Universidade Federal do Paraná, Curitiba. [ Links ]
Kersten R.A., Silva S.M. 2006. The Floristic Compositions of Vascular Epiphytes of a Seasonally Inundated Forest on the Coastal Plain of Ilha do Mel Island, Brazil. Rev. Biol. Trop. 54: 935-942. [ Links ]
Kress W.J. 1986. The systematic distribution of vascular epiphytes: an update. Selbyana 9: 2-22. [ Links ]
Maddison M. 1977. Vascular epiphytes: their systematic occurrence and salient features. Selbyana 2: 1-13. [ Links ]
Mueller-Dombois E. & Ellenberg H. 1974. Aims and methods of vegetation ecology. Ed. John Wiley & Sons, Nova York. [ Links ]
Nadkarni N.M. 1994. Diversity of species and interaction in the upper tree canopy of forest ecosystems. Amer. Zool. 34: 70-78. [ Links ]
Parker G.G. 1995. Structure and microclimate of forest canopies p. 73-106. In M.D. Lowman & N.M. Nadkarni (eds.) Forest Canopies. Academic Press, San Diego. [ Links ]
Reitz R. 1983. Bromeliáceas e a Malária-bromélia endêmica -Flora Ilustrada Catarinense, Herbário Barbosa Rodrigues, Itajaí. [ Links ]
Ribeiro, J.E.L.S.; Hopkins, M.J.G.; Vicentine, A.;Sothers, C.A.; Costa, M.A.S.; Brito, J.M.; Sousa, M.A.D; Martins, L.H.P.; Cohmann, L.G.; Assunção, P.A.C.L.; Pereira, E.C.; Silva, C.F.; Mesquita, M.R. & Procópio, L.C. 1999. Flora da Reserva Ducke. INPA-DFID. Manaus. [ Links ]
Sehnem A. 1970. Polipodiáceas -Flora Ilustrada Catarinense, Herbário Barbosa Rodrigues, Itajaí. [ Links ]
Silva, S.M. & Britez, R.M. 2005. A vegetação da planície costeira. In História natural e conservação da Ilha do Mel -(M.C.M. Marques & R.M. Britez, orgs). Ed Universidade Federal do Paraná, Curitiba. Pp. 125-144. [ Links ]
Steege H., Cornelissen J.H.C. 1989. Distribution and ecology of vascular epiphytes in Lowland rain forest of Guyana. Biotropica 21: 331-339. [ Links ]
Waechter J.L. 1992. O epifitismo vascular na planície costeira do Rio Grande do Sul. PhD. Thesis Universidade Federal de São Carlos, São Carlos. [ Links ]
Waechter J.L. 1980. Estudo fitosociológico das orquidáceas epifíticas da mata paludosa do Faxinal, Torres, Rio Grande do Sul. Dissertation Universidade Federal do Rio Grande do Sul, Porto Alegre. [ Links ]
Waechter J.L. 1998. Epifitismo vascular em uma floresta de restinga do Brazil Subtropical. Revista Ciência e Natura 20: 43-66. [ Links ]
Yeaton R.I, Gladstone D.E. 1982. The patterns of colonization of epiphytes on calabash trees (Crescentia alata H.B.K.) in Guanacaste province, Costa Rica. Biotropica 14: 137-140. [ Links ]
Zimmerman J.K, Olmsted I.C. 1992. Host Tree utilization by vascular epiphytes in a seasonally innundated forest (Tintal) in Mexico. Biotropica 24: 402-407. [ Links ]
Internet references
Tropicos 2008 Tropicos.org. Missouri Botanical Garden. <http://www.tropicos.org>. accessed Jul. 2008. [ Links ]
World Checklist of Selected Plant Families. 2008. The Board of Trustees of the Royal Botanic Gardens, Kew. Published on the Internet; http://www.kew.org/ wcsp/ accessed Jul. 2008. [ Links ]












 uBio
uBio 

