Revista de Biología Tropical
versão On-line ISSN 0034-7744versão impressa ISSN 0034-7744
Rev. biol. trop vol.57 no.1-2 San José Mar./Jun. 2009
Scarlet Macaw (Ara macao) (Psitaciformes: Psittacidae) Parental Nest Visitation in Costa Rica: Implications for Research and Conservation
Christopher Vaughan1,2,3, Mark Bremer3,4 & Fiona Dear5
1. Department of Forest and Wildlife Ecology, University of Wisconsin-Madison, Madison, WI 53706 Tel. 608-2632071; cvaughan@wisc.co.edu
2. International Institute for Wildlife Conservation and Management, Universidad Nacional, Heredia, Costa Rica.
3. Associated Colleges of the Midwest, Apartado 10265, San Jose, Costa Rica.
4. State University of New York-Institute of Technology & Onondaga Community College Syracuse, NY 13215.
5. School of Biological Sciences, University of East Anglia, Norwich, NR4 7TJ, UK.
Abstract: We studied temporal parental visitation of Scarlet Macaws (Ara macao) to six active nests in the Central Pacific Conservation Region of Costa Rica. Total parental time in the nest decreased significantly as the nestlings aged. Results provide guidelines to scientists for planning invasive activities to nestlings, such as placement of radio collars, or biological sample collection. These activities should be performed close to the end of the nesting period for minimal disturbance of parents and nestlings. Our results also provide information to aid wildlife guards in protecting active nests from poachers when chicks are close to fledging. Rev. Biol. Trop. 57 (1-2): 395-400. Epub 2009 June 30.
Key words: nesting; nestling, parental visit; Psittacine, scarlet macaw.
The Scarlet Macaw (Ara macao) is the most widely distributed (Mexico to Brazil) of the 17 extant macaw species (Wiedenfeld 1994). Throughout its range, it is endangered due to habitat modification and the pet trade (Iñigo-Elías 1996, Wright et al. 2001, Vaughan 2002). Costa Rica has two "viable" Scarlet Macaw populations: approximately 700 individuals in the Osa Conservation Area (ACOSA), and approximately 430 individuals in the Central Pacific Conservation Area (ACOPAC)(Marineros and Vaughan 1995, Vaughan 2002, Vaughan et al. 2005). Limited research has been conducted on its nesting ecology (Marineros and Vaughan 1995, Inigo- Elias 1996, Vaughan 2002, Vaughan et al. 2003). The Scarlet Macaw is an asynchronous egg layer (Beissinger and Bucher 1992), laying one to four eggs in tree cavities which average 20 m high (Vaughan et al. 2003) at three-day intervals between December and February. Eggs hatch about 22 days after eclosion and chicks fledge after approximately 75 days. Asynchronous nesting results in chicks fledging over a 45 day period (middle April-late May; Vaughan 2002). This makes nest protection difficult, and also complicates research efforts because it is difficult to estimate the developmental stages of nestlings at any given time without climbing trees to directly examine each nestling (Vaughan et al. 2003, Myers and Vaughan 2004). Thus, it is important to determine less invasive methods to age chicks in ways that facilitate their protection and data collection for research.
A non-invasive technique for estimation of chick age could be based on observing how changes in frequency and duration of parental nest visitation relate to chick development. Lack (1968) observed that parental nest visitation decreases with nestling development in most bird groups. Parental presence at nests for the Green-rumped Parrotlet (Forpus passerinus) decreased and nestlings spent longer periods between feedings from days 9 to 43 days post-hatch because parents spent more time away from the nest to gather food for their rapidly growing nestlings (Grenier and Beissinger 1999). Other studies have examined factors that relate feeding rates, food intake, and brood size in birds (Royama 1966, Wiggins 1990, McCarthy 2002). To test the hypothesis that the duration of parental nest visits correlates with nestling age, we studied parental visits throughout the breeding season at active Scarlet Macaw nests in the Central Pacific Conservation Area.
Materials and methods
This research was conducted on the Hacienda Quebrada Bonita (90º47 N, 85º37 W), a 650-ha dairy farm located between Carara National Park, an important feeding site for macaws, and Guacalillo Mangrove Reserve, the primary roosting site for the Central Pacific Conservation Area (Costa Rica) Scarlet Macaw population. Hacienda Quebrada Bonita consists of cattle pastures with scattered large trees, living fencerows, and scattered woodlots.
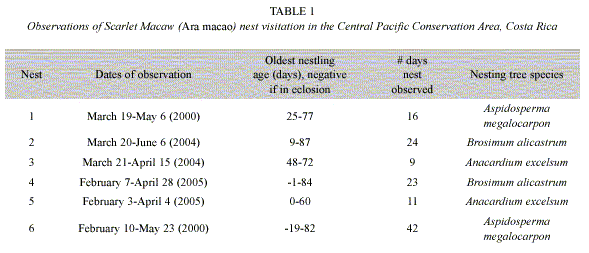
We studied the timing and duration of Scarlet Macaw parental visits at six nests during the breeding season between January-June (Table 1). To calculate total parent visitation time, an observer sat 20-30 m from the nest between 0600-1200 h or a total of 360 minutes and recorded the number of adult visits and minutes spent per visit at each nest. The total number of minutes for the morning was totaled, which was a maximum of 360 minutes if the parents were in the nest the entire time. When a nest was chosen, it was visited using a similar climbing system as outlined by Munn (1991). Chicks were observed and their age estimated based on weight and body development comparisons from data on chicks from nests in the study area followed from eclosion to fledging (Vaughan, unpublished data). Nestlings from each of the six nests were weighed and body parameters measured weekly. We modeled parental time at the nest (visit) as a linear function of age and included nest as a fixed-factor in analyses to examine potential differences between breeding pairs (Maindonald and Braun 2007).
Results
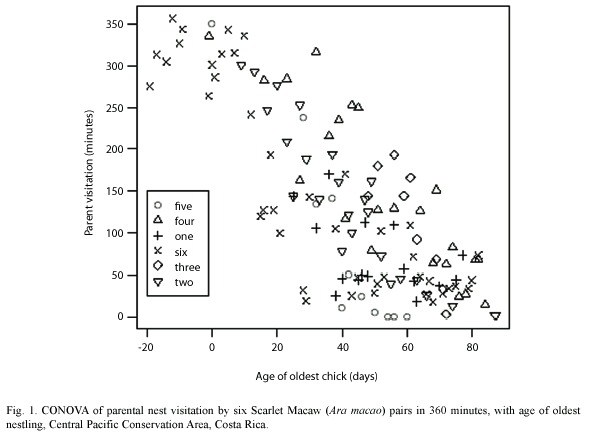
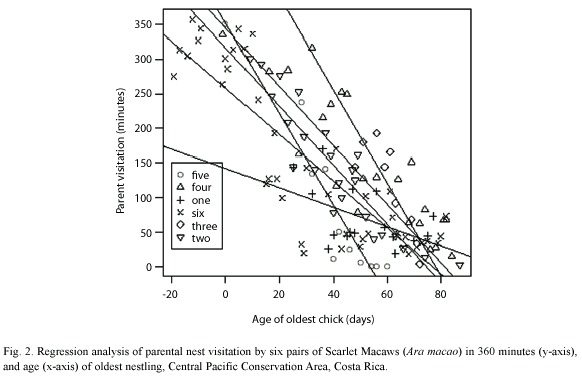
Macaw pairs were observed a total of 125 times visiting six nests in 2000, 2004 and 2005 (Table 1). One nest was monitored since the eggs were laid, about 20 days before hatching. As nestlings aged, parental time in nests decreased from 70% when chicks were 10-20 days old to about 20% when nestlings were 60 days old. Relationships between nestling age and parental time in the nest were significantly negatively correlated (Fig. 1, Table 2). An analysis of variance demonstrated (Table 2) that nestling age influences parental time at the nest, but also that each nest is slightly different in this regard as they do not have the same intercept or slope (Fig. 2).
Discussion
One can easily differentiate between active and inactive nests by observing whether adult macaws are entering or not. In our study of active nests, Scarlet Macaw adults spent less time at their nests as nestlings grew (Table 2, Fig. 1). This was probably related to the need to search for increasing quantities of food for rapidly growing chicks (Wiggins 1990, Grenier and Beissinger 1999, McCarthy 2002), as well as the increased ability of the chicks to thermoregulate as body size and feathering increases. The model that relates parental time at each nest (visit) as a linear function of age (Fig. 2) indicated that each nest had a unique intercept and/or slope, although some were similar (e.g. nests 2 and 6). Slope differences may signify behavioral differences among macaw pairs in caring for their chicks, including (1) the possibility that some parents spent more time in the nest as their chicks aged while others spent less time, (2) differences in number of chicks in nest, (3) less food in the nest vicinity and therefore more time spent searching for food, and/or other reasons (Vaughan et al. 2006). Differences in intercepts could relate to individual parental care, but also if older chicks either fledged or were poached (nest 5) and we were unaware they were gone (nest 2, 4) (Table 1, Figure 2). However, if one chick was poached, it seems likely that siblings would also be poached at the same time.
It is important to develop non-invasive techniques to determine the age of chicks for several reasons. First, for research purposes (placing radio-collars and sampling for health, genetics and/or disease studies), it is safest to handle chicks that are closer to fledging age. Second and related to the first, climbing trees to observe chicks in nests is difficult, timeconsuming and dangerous, so time should not be wasted by climbing inactive nests or those in which chicks are very immature. Third, to maximize limited resources for chick protection, wildlife guards must be able to determine which nests are active, especially those with nestlings close to fledging age. This information is vital because poaching for the pet trade poses the principal threat to psittacines in the Central Pacific Conservation Region (Vaughan 2002, Vaughan et al. 2003) and elsewhere (Wright et al. 2001).
The present study demonstrates that by observing parental visits, one can reasonably estimate the developmental stage of nestlings without disturbing nesting activity. This facilitates both research and protection. However, because of the variation in reproduction onset (Table 1) and parental visitation patterns (Fig. 2), in addition to the observation that ACOPAC chicks are sometimes stolen from nests at several weeks of age (Vaughan, pers. obs.), nest protection should begin early and continue until confirmation that all chicks have fledged. In addition, the observation of Scarlet Macaw adults visiting a nest is also suggestive that the nest is likely active. If the nesting attempt fails, it will also be apparent from the type of monitoring used in this study. Nest predators, such as the Black Iguana (Ctenosaura similis), the common opossum, (Didelphis marsupialis), and white-faced monkey (Cebus capucinus) (Vaughan et al., 2003) can also be discouraged from predating macaw nests if human observers are nearby. We believe that one of the major advantages of estimating nestling ages with the parental visitation method is providing the researcher a general idea of when to climb the nest to more safely study its nestlings.
Acknowledgments
We thank Idea Wild, Wildlife Trust International, The Parrot Society-UK, the Regional Wildlife Management Program, Paradise Wildlife Park UK and the C. James Cadbury Charitable Trust UK for equipment and logistical support. Also we thank Hernan Vargas for allowing us to work on his land and Jerimias Sirio for assistance in protecting the macaw nests from poaching. Dan Storm assisted in data analysis and Mark Myers and Nicole Nemeth provided valuable manuscript suggestions. Finally, we thank Carrie Kowalski for assisting in data collection.
Resumen
Estudiamos las visitas temporales de padres de la lapa roja (Ara macao) a seis nidos activos en el Área de Conservación del Pacifico Central (Costa Rica). El tiempo total que pasaron los padres en los nidos bajó en forma significativa en relación con el crecimiento de los pichones. Estos resultados pueden ayudar a los científicos a planear sus actividades "invasivas", tales como colocación de radio collares o colección de muestras biológicas. Las actividades deben coincidir con el final de la época de anidación para molestar menos a las aves. Nuestros resultados proveen información que puede ayudar a los guardaparques a proteger nidos activos cuando los pichones estan casi listos para salir.
Palabras claves: anidación, pichón, visita de padre, Psittacine, lapa roja
References
Beissinger, S. R,. & E. H. Bucher. 1992. Sustainable harvesting of parrots for conservation, pp. 73-115. In S. Beissinger & N. Snyder (eds.). New world parrots in crisis: solutions from conservation biology. Smithsonian Institution, Washington, D.C., USA. [ Links ]
Grenier, J. L., & S. R. Beissinger. 1999. Variation in the onset of incubation in a neotropical parrot. Condor 101: 752-761. [ Links ]
Inigo-Elías, E. 1996. Ecology and breeding biology of the Scarlet macaw (Ara macao) in the Usumacinta drainage basin of Mexico and Guatemala, Ph.D. Thesis, University of Florida, Gainesville, Florida., USA. [ Links ]
Lack, D. 1968. Ecological adaptation for breeding in birds. Metheun, London, United Kingdom. [ Links ]
Maindonald, J. & J. Braun. 2007. Data analysis and graphics using R: An example-based approach. Cambridge University, Cambridge, United Kingdom. [ Links ]
Marineros, L., & C. Vaughan. 1995. Scarlet macaws of Carara, p. 445-467. In J. Abramson, B. Speer & J. Thomsen (eds.). The large macaws: their care, breeding and conservation. Raintree Publications, California, USA. [ Links ]
McCarthy, J. 2002. The number of visits to the nest by parents is an accurate measure of food delivered to nestlings in tree swallows. J. Field Ornithol. 73: 9-14. [ Links ]
Munn, C. 1991. Tropical canopy netting and shooting lines over tall trees. J. Field Ornithol. 62: 454-463. [ Links ]
Myers, M., & C. Vaughan. 2004. Movement and behavior of scarlet macaws (Ara macao) during the postfledgling dependence period: implications for in situ versus ex situ management. Biol. Conserv. 118: 411-420. [ Links ]
Royama, T. 1966. Factors governing feeding rates, food requirement and brood size of nestling great tits Parus major. Ibis 108: 313-347. [ Links ]
Vaughan, C., N. Nemeth, L. Marineros. 2006. Observations of Scarlet Macaw (Ara macao) diet in Central Pacific Costa Rica. Int. J. Trop. Biol. Conser. 54(3): 919-926. [ Links ]
Vaughan, C., N. Nemeth, J. Cary & S. Temple. 2005. Response of a Scarlet Macaw (Ara macao) population to conservation measures. Birdlife Internat. 15: 119-130. [ Links ]
Vaughan, C., N. Nemeth & L. Marineros. 2003. Ecology and management of natural and artificial Scarlet Macaw (Ara macao) nest cavities in Costa Rica. Ornit. Neotrop. 14: 381-396. [ Links ]
Vaughan, C. 2002. Conservation strategies for a Scarlet Macaw (Ara macao) population in Costa Rica. Ph.D. thesis, Univ. of Wisconsin, Madison, Wisconsin, USA. [ Links ]
Wiedenfeld, D. 1994. A new subspecies of scarlet macaw (Ara macao) and its status and conservation. Ornitología Neotrop. 5: 99-104. [ Links ]
Wiggins, D. 1990. Food availability, growth and heritability of body size in nestling tree swallows (Tachycineta bicolor). Canadian J. Zool. 68: 1292-1296. [ Links ]
Wright, T., C. Toft,, E. Enkerlin-Hoeflich, J. Gonzalez-Elizondo, M. Albornoz, A. Rodriguez-Ferraro, F. Rojas-Suarez, V. Sanz, A. Trujillo, S. Beissinger, V. Berovides, X. Galvez, A. Brice, K. Joyner, J. Eberhard, J. Gilardi, S. Koenig, S. Stoleson, P. Martuscelli, J. Meyers, K. Renton, A. Rodriguez, Sosa-Asanza, F. Villela, & J. Wiley. 2001. Nest poaching in Neotropical parrots. Conserv. Biol. 15: 710–720. [ Links ]












 uBio
uBio 

