Revista de Biología Tropical
versão On-line ISSN 0034-7744versão impressa ISSN 0034-7744
Rev. biol. trop vol.52 no.4 San José Dez. 2004
Akumal s reefs:Stony coral communities along the developing Mexican Caribbean coastline
Roshan E.Roy
Department of Integrative Biology,The University of Texas at Austin,Austin,TX 78712,USA.Fax:01 512 721-7363; roshanstrong@gmail.com
Received 17-III-2003. Corrected 25-V-2004. Accepted 28-V-2004.
Abstract:Fringing coral reefs along coastlines experiencing rapid development and human population growth have declined worldwide because of human activity and of natural causes.The "Mayan Riviera "in Quintana Roo,México,attracts large numbers of tourists in part because it still retains some of the natural diversity and it is important to obtain baseline information to monitor changes over time in the area.In this paper,the condition of the stony corals in the developing coastline of the Akumal-area fore reefs is characterized at the start of the new millennium at two depths,and along an inferred sedimentation gradient.Transect surveys were conducted in five fringing reefs starting at haphazardly chosen points.with respect to species composition,live cover,colony density,relative exposure to TAS mats and,for one species (Diploria strigosa ,Dana,1848),tissue regression rates in the presence of TAS mats.Fish population density and herbivory rates are also assessed.Data from line intercept transects (n=74)show that live stony coral cover,density and relative peripheral exposure of colonies to turf algal/sediment (TAS)mats were inversely related to an inferred sediment stress gradient at 13m.In 2000, live stony coral cover had decreased by 40-50%at two sites studied in 1990 by Muñoz-Chagín and de la Cruz- Agüero (1993).About half of this loss apparently occurred between 1998 and 2000 during an outbreak of white plague disease that mostly affected Montastraea faveolata ,and M.annularis .At a 13 m site,where inferred sedimentation rates are relatively high,time series photography of tagged Diploria strigosa ,(n=38)showed an average loss of 70 cm 2 of live tissue/coral/year to encroachment by TAS mats during the same period.Whereas densities of carnivorous fishes and herbivores (echinoids,scarids,acanthurids and Microspathodon chrysurus )in 2000 were low in belt transects at 10-19 m (n=106),turf-algal gardening pomacentrids were relatively common on these reefs.Rev.Biol.Trop.52(4):869-881.Epub 2005 Jun 24.
Key words:Stony coral declines,turf-algal/sediment mats,herbivory,disease,fore reefs
Fringing coral reefs along coastlines experiencing rapid development and human population growth have declined worldwide (Ginsburg and Glynn 1994,Wilkinson 2000).Regional die-offs of some key organisms (Diadema antillarum Phillipi, 1845, according to Lessios 1988,Acropora -Aronson and Precht 2000) and bleaching-associated mortality events (Wilkinson 2000)have further devastated coral reefs throughout the wider Caribbean (Jiménez 2001,Asch and Turgeon 2003,Banaszak et al. 2003,Borger 2003,Cróquer and Bone 2003, Croquer et al.2002,2003,García et al.2003, McGrath and Smith 2003,Miller et al.2003). The "Mayan Riviera "in Quintana Roo (QR), México attracts large numbers of tourists in part because it still retains some of the natural diversity that has been lost at other vacation destinations. Nevertheless, the disruptive ecological effects of inadequate environmental protection and poor enforcement of existing regulations are beginning to show,particularly in the Cancún-Tulum corridor in the northern region of the state (Jordán-Dahlgren 1993, Lang et al.1998).Cement is replacing mangroves,while public access to the shore has been privatized at huge resorts.At present,the only legal method for sewage disposal is in "deep-injection wells,"although the limestone bedrock is known for its world-class cenotes (cave systems)"carved out "by groundwater flowing into the sea.Turtle nesting beaches are disappearing and fishing has diminished the local invertebrate and fish populations. New pueblos supporting the tourism industry s workers generally lack the most basic sanitary facilities and conditions are hardly better in the longer-established villages.Development alone is not to blame for changes to the QR coastal ecosystems, however, as the Yucatán Peninsula is frequently exposed to hurricanes,of which the most destructive in recent decades were Gilbert in 1988 and Roxanne in 1995.
The fringing-barrier reefs along the QR coastline constitute the northernmost part of the Mesoamerican Barrier Reef System (MBRS). Detailed descriptions of these reefs can be found in Jordán –Dahlgren (1993),Gutierez-Carbonell et al.(1995)and other reports; Nolasco-Montero and Carranza-Edwards (1988)have characterized the associated sediments.The QR scleractinian coral fauna has been described most recently by Beltrán-Torres and Carricart-Ganivet (1999).
Akumal is a small resort about 105 km south of Cancún.It s fore reefs consist of loosely coalesced spurs and grooves on three narrow terraces (Muñoz-Chagín and de la Cruz-Agüero 1993).At depths of ~13 m,a sandy plain of about 10 m width separates low-relief (<3 m)spurs on the "inner lobes " (shallowest terrace)from the higher-relief (3-5 m)"middle-lobe " spurs (although the two lobes merge in some areas).A second sandy plain at ~19 m divides the middle-lobe spurs from the highest-relief (6-9 m) spurs of the "outer lobes." The outer lobes terminate at >30 m in a hard bottom which slopes off at about 65 m into the Yucatan Channel.
In 1990,area-based estimates of live stony coral cover on two fore reefs (Las Redes and Média Luna)at 6-18m that were spatially dominated (40-45%)by Montastraea annularis , (now species of the M.annularis complex, Weil and Knowlton, 1994)averaged 22-24% (Muñoz-Chagín and de la Cruz-Agüero 1993). As elsewhere in the MBRS (Kramer et al. 2000),the 1998 ENSO-related mass bleaching event and an outbreak of white plague (WP), a disease which here primarily affected the M.annularis complex,were major disturbance events.Linear estimates of live coral cover in March 1999 ranged between 13.5%and 21% at 11-18 m on six middle-lobe fore reefs near Akumal (Steneck and Lang 2003).
Coastal seawater temperatures in the northern QR varied from ~24-30 ºC between 1994 and 2000 (CARICOMP 1997,D.Linton pers. comm .).Nearshore surface currents generally flow to the south,but occasionally shift to the north.Bulk transport of sediment from the shallower terraces into deeper water can occur during the passage of hurricanes.Sediments are frequently shifted during tropical storms, with depths at known locations on the fore-reef sandy plains changing by as much as ± 0.3 m and,at a finer scale,sediment on the shallowest terrace is routinely suspended by surge. Razo Amoroz (1999)has measured relatively high (10-394 mg/cm2/day)sedimentation rates at 10 m in a fore reef (Shark Caves)off Akumal.
Sediment loading can be a determining factor in community structure.Acevedo and Morelock (1988),showed that communities which receive high rates of sediment input are characterized by lower diversity,of reduced light and high sediment tolerant species.Stony corals,are naturally exposed to resuspended sediments during storms and not entirely defenseless,possessing differing abilities at actively clearing their tissues of debris in one or more of four ways,ciliary action,tentacular utilization, stomodeal distension,and the secretion and shedding of mucus (Hubbard and Pocock 1972,Stafford-Smith 1993).Additional passive forms of debris removal are related to the morphology of the coral,which allow gravity and water-flow to assist the process (Lasker 1980).What is not generally recognized is that once sediment and debris reach the edge of a live stony coral s tissues,a potentially new problem occurs if algal turfs are growing in direct contact with its margin (McCook et al. 2001).The algal turfs can trap the sediment and form turf algal-sediment (TAS)mats.
TAS mats are also formed when turf algae (sensu Steneck 1988)flourish in areas of high sediment loading and resuspension.Turf algal filaments baffle water flow,causing some sediment to fall out of the water column and become trapped (Scoffin 1970,Carpenter 1986),thereby creating TAS mats. When TAS mats come into contact with stony corals,the marginal tissues of the latter may gradually die (Dustan 1977,1999).Herbivores (Diadema and other echinoids or fishes)that would graze algae are relatively scarce on the Akumal-area fore reefs,and preliminary estimates of fish are low (Steneck and Lang 2003).TAS mats are conspicuous,particularly on the inner-lobe reefs,where they are frequently in close physical contact with,and even appear to be over-growing,stony corals (pers.obs .).The possible expansion of TAS mats at the expense of the primary reef constructors adds a previously unstudied dimension to investigations of stony coral mortality in these reef systems.
Baseline information is extremely important in characterizing a community and analyzing measurable changes over time in systems like coral reefs (Loya 1976,Reichelt et al. 1986).Therefore the status of their coral reefs is vital for small communities like Akumal that are financially dependent upon the diving industry.In this paper,the condition of the stony corals in the Akumal-area fore reefs are characterized at the start of the new millennium at two depths,and along an inferred sedimentation gradient,with respect to species composition,live cover,colony density, relative exposure to TAS mats and, for one species (Diploria strigosa ,Dana,1848),tissue regression rates in the presence of TAS mats. Fish population density and herbivory rates are also assessed.
Materials and methods
Survey sites on five reefs were chosen near the seaward margins of the inner (at 13 m) or the middle (at 19 m)fore-reef lobe along a 5.5 km stretch of coastline between Bahia Akumal and Xaak (Fig.1,Table 1).Las Redes (sites LR13 and LR19)is off the 2 km long, <3 m deep Bahía Akumal,where high levels of small boat traffic frequently resuspend sediments (Muñoz-Chagín and de la Cruz-Agüero 1993)and is used as a training site for novice scuba divers.Morgan s Reef (MR13 and MR19)is off a rocky headland marking the northern end of Bahía Akumal and visited less frequently by recreational divers.Média Luna (ML13 and ML19)is off the middle to southern side of the small (0.5 km long),shallow (<2 m deep)Bahía de Média Luna.Yal Ku (YK13 and YK19)is directly in front of Caleta Yal Ku,a major surface outlet of groundwater into the Caribbean Sea.Both ML and YK are popular recreational dive sites.Xaak (XA13),near a headland on the southern side of a tiny, undeveloped embayment with a narrow beach,is seldom dived by tourists but is frequented by artisanal fishers (J.Lang pers.comm.).
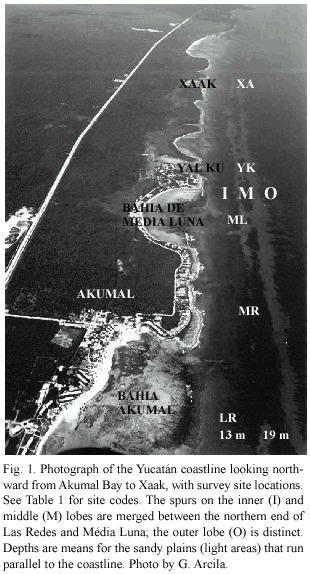
Reefs off large shallow sandy bays are considered to receive greater sediment loading than those off rocky headlands or narrow caletas (inlets).Surge decreases with depth, and suspended sediment over the spurs was markedly less conspicuous at 19 m than at 13 m on any given day.I ranked the reefs at 13 m by a gradient of expected sediment stress based on the above described assumptions of sediment re-suspension levels,as follows:LR>ML>MR>YK>XA.I expect to find an increase in coral cover and coral density,and a decrease in the frequency of corals in contact with TAS mats, as the sediment loading and resuspension rates decrease along this gradient.
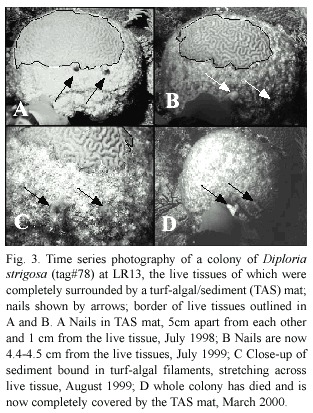
Line intercept techniques were used at LR13 in 1998-2000 (with a limited follow-up in March 2001 for Montastraea faveolata ,Ellis and Solander,1786,Diploria strigosa and Siderastrea siderea ,Ellis and Solander,1786),ML13 in 1999 and 2000 and at all remaining sites except YK19 (which was not surveyed for stony corals) in June-August 2000.Each of the 10m long transects began at a haphazardly chosen point on the top of a spur and was oriented parallel to its long axis.All stony corals under the line were identified to species and the intercept length of its live tissues was measured.Each colony was visually estimated as to whether its periphery was partially (~50 to 75%)or completely (~100%)surrounded by a TAS mat,since virtually none were in the >75 to <100% condition.Contact between TAS mats and live tissues in a total of 42 randomly chosen and tagged colonies of D.strigosa at LR13 were photographed once or twice each year in 1998 (when n=18 corals),1999 (when n=37 corals) and 2000-2001 (when n=38 corals).The slides were digitized and analyzed with the Scion Beta 4.0.2 version of NIH Image to extract temporal changes in surface area.Two nails emplaced ~5 cm apart in the TAS mat and,at the start of the experiment,~1 cm from the live coral tissues (R.Roy 2004)were used as fixed reference points and to set the scale of each image.Due to slight differences in the orientation of the slides,the unique ridge pattern of most colonies was used to draw an outline of the 2001 image over the initial image to calculate the net difference in surface area between the two time intervals;the results were standardized to cm of tissue change/year.
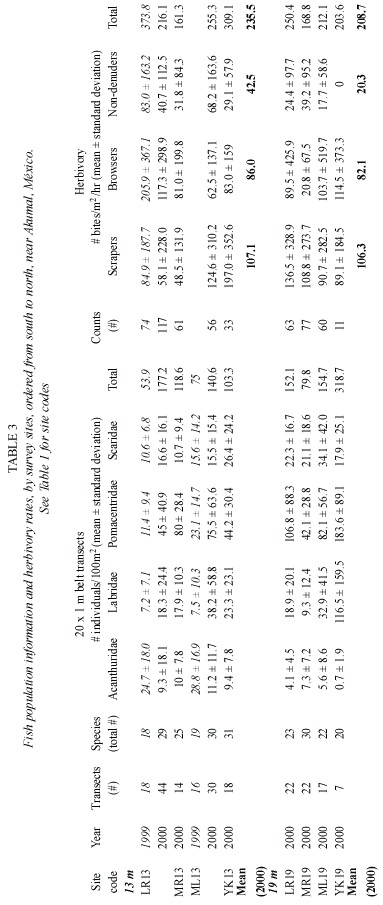
Fishes were surveyed at LR13 and ML13 in 1999 and at all other sites except XA in 2000 using a 20 m long x 1 m wide belt-transect method adapted from Fowler (1987).Starting from a haphazardly chosen position,transects were restricted to the top of a single spur.One diver remained stationary while spooling out a line attached to a second diver who counted fish while slowly swimming at a constant height of 3-5 m above the reef with a 1 m wide PVC T-bar for scale.Herbivory was estimated at LR13 in 1999,and at all other sites except XA in 2000,by a modification of Steneck s (1985)fish bite method.The total number of bites observed in a three-minute interval by scrapers (scarids),browsers (acanthurids and the pomacentrid Microspathodon chrysurus , Ginther,1862)and non-denuders (other herbivorous pomacentrids)was recorded.All fish observations were made from 11:45 a.m.-12:45 p.m.and 3:45-4:45 p.m.Due to their scarcity, echinoid populations were not surveyed.
Statistical analyses of the 2000 dataset were conducted with SAS version 8.1.Unless stated otherwise,data were tested with one-way weighted ANOVAs after normality was determined.When relevant,rather than testing for differences between specific sites,a simple linear regression (Motulsky 1995)was conducted as a post-hoc analysis to examine the relationship with the expected sediment- loading gradient.Data that could not be sufficiently normalized for a parametric test were analyzed with the Kruskal-Wallis,non-parametric ANOVA.
Results
A total of 26 species of stony corals (Table 2)was recorded at 13 m (range=14-19/site, n=5 sites)and similar numbers were found at 19 m (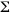

Live stony coral cover at LR13 (Table 1)declined significantly from 19%in 1998 to 10.5%in 2000 (ANOVA,p=<0.005).This change was largely due to a loss in cover of over 50%of the M.faveolata and about two- thirds of the S.siderea; however, both species appeared to be recovering in March 2001(Fig. 2).No significant changes in live cover were recorded at ML13 between 1999 and 2000. The between-site differences in live cover at 13m in 2000 (from 10.5%at LR to 24.5% at XA,Table 1)were significant (ANOVA, p<0.01).Mean stony coral densities,which showed no significant variation between 1998 and 2000 at LR13 or between 1999 and 2000 at ML13,did vary significantly (ANOVA, p<0.006)among the 13m sites in 2000 (from 0.6/m at LR and ML to 1/m at XA).Both live cover and density were inversely related to the inferred sediment stress gradient (r2 =0.16, p=0.0004 and r2 =0.12,p=0.0029,respectively).In 2000,live stony coral cover at the three 19m sites ranged from 10%-13.5%,and densities from 0.8-1/m.Although the between-depth differences in live coral cover in 2000 were only significant when including all sites (ANOVA,p<0.026),colony density showed a significant depth effect even among the three southernmost reefs (ANOVA,p<0.039).
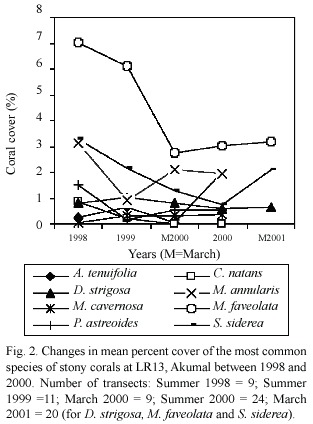
15-32%of the stony corals at the five13m sites,and 13-25%of those at the three 19m sites,had peripheries that were partially (>50%- 75%)in contact with TAS mats in 2000 (Table 1);these differences were not significant.At this time,the between-site differences at 13 m for stony corals that were completely surrounded by TAS mats (27%at XA-66%at LR)were significant (ANOVA, p<0.0004),and inversely related to the inferred sediment stress gradient (r2= 0.18, p=0.0001).Annual changes in the percent of corals that were completely surrounded by TAS mats were not significant at LR13 nor at ML13 for the years surveyed.Only 28-37%of the surveyed corals at 19 m were completely surrounded by TAS mats in 2000; the between-depth differences in 2000 for this parameter in the three southern reefs were significant (Kruskal-Wallis,p<0.0019)
At the level of species,all of the D. strigosa (n=5 corals),93%of the M.faveolata (n=14 corals)and a third of the S.siderea (n=15 corals)in the1998 line transects at LR13 were completely surrounded by TAS mats.In 2000,this parameter varied between XA13 and LR13 as follows:D.strigosa –50%(XA,n=2) and 92%(LR,n=13);M.faveolata –50%(XA, n=10)and 92.5%(LR,n=27);and S.siderea – 33%(XA,n=3)and 67%(LR,n=18).
Data for five of the photographed D. strigosa at LR13 are incomplete.Although all tagged corals may have grown vertically from 1998 -2001,none of the remaining 37 corals showed any lateral expansion in live surface area.These 37 corals did not maintain their original live tissue coverage either.Including three corals that died by 2001,these 37 corals lost an average of 70 ±13.2 cm2 of live tissues/ coral/year (Fig.3).
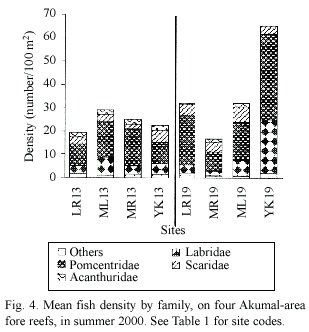
A sum of 20 to 31 fish species were recorded in each of the eight sites in 2000 (Table 3),most of which were common to all reefs. Total fish densities in 2000 varied from ~100- 175/100m2 at 13 m and from ~80-320/100m2 at 19 m (Table 3).Of the four most commonly observed families,pomacentrids (damselfish) were the most common at all sites,while acanthurids were generally least abundant,and with densities for scarids and labrids usually in the intermediate range (Fig.4).Herbivorous fishes (scarids,acanthurids,M.chrysurus and algal-gardening damselfishes),which overall averaged ~70/100m2 (n=8 sites),were about as common as all other fish guilds combined (~80/100m2 ).There were no significant spatial differences between the fish populations at LR13 and ML13 in either 1999 or 2000; however,fish densities at both sites were significantly higher in 2000 than 1999 (ANOVA for LR13,p<0.04,for ML13,p<0.0337). Also,no significant differences were found among any of the sites in 2000 (excluding the YK19 data,for which the number of replicate transects was low).Herbivory rates at 13 m (n=4 sites)averaged 107.1 bites/m2 /hour for scrapers,86 bites/m2 /hour for browsers and 42.5 bites m2 hour for non-denuders (Table 3).Corresponding rates at 19m were 106.3,82.1 and 20.5 bites/m2 /hour (n=4 sites), respectively.(Due to low replication,the fish bite data for YK19 is also suspect,especially for non-denuders,since pomacentrid density estimates were highest at this site.)No big serranids,lutjanids or other large-bodied carnivores such as Sphyraena barracuda, Walbaum, 1792,were encountered in the belt transects,although individuals were occasionally observed at other times.
Discussion
Between 1990 and 2000, live stony coral cover has clearly decreased in the Akumal-area reefs (compare Muñoz-Chagín and de la Cruz-Agüero 1993,with Steneck and Lang 2003 and this paper). Potential contributing factors inlcude; hurricanes,bleaching-related mortality,outbreaks of disease, overgrowth by macroalgae (Steneck and Lang 2003)or encroachment by TAS mats (this paper).Whereas hurricane Gilbert had destroyed much of the Acropora cervicornis on the Akumal-area fore reefs in 1988 (G. Arcila pers.comm.),Mitch in 1998 caused relatively little physical damage to this habitat (S.Slingsby pers. comm .).However,stresses associated with the 1998 mass bleaching event are thought to have increased the susceptibility of stony corals (in particular M.faveolata and M. annularis )to WP,which was wide-spread in the Akumal area in March 1999 (Steneck and Lang 2003).
The loss of nearly 50%of the live cover at LR13 between 1998 and 2000 can be attributed primarily to the effects of this epizootic in the M.annularis species complex.WP is also believed to have reduced the cover of these species at ML and on other reefs in the immediate Akumal area. WP was less conspicuous in March 2001 (when the live cover of M. faveolata at LR13 was about the same as in the previous summer), but was observed in ~2%of all colonies of the M.annularis complex during summer 2002 (J. Bruno pers.comm .).Yellow-blotch (also called yellow-band)disease (YBD),first noticed in a few colonies of Akumal-area M.faveolata in 1997 (Steneck and Lang 2003),was very conspicuous at LR13 in March 2000,yet it was less isible a year later (pers.obs .).J.Bruno (pers. comm.)has estimated that in summer 2002, less than 10%of the M.annularis complex in most areas of Akumal were infected with this disease.Signs of white-band disease (WBD) were seen in several colonies of Acropora palmata on the inner lobes at LR and XA in 1997 by J.Lang (pers.comm .).Although WBD was considered "fairly common "during 2000-2001 in A.palmata and in A.cervicornis,(which is currently rare,Table 2),no examples were seen in summer 2002 (J.Bruno pers.comm .).
Blooms of macroalgae were conspicuous in the months following the passage of Hurricane Mitch in 1998 after an initial reduction during the days immediately following the storm (S.Slingsby pers.comm.).The relative abundance of macroalgae on six middle-lobe fore reefs near Akumal was found to have increased from ~25%to 44%between 1997 and 1999 by Steneck and Lang (2003)who discussed increased colonization space on the skeletons of diseased stony corals,the possibity of increased nutrients in groundwater,and decreased herbivory rates as potential explanations for this change.Although Steneck and Lang (2003)noted a decline on the reef substratum in the relative abundance of algal turfs (from 62-~35%between 1997 and 1999)at 11- 18 m,I found no significant difference between the proportion of colonies completely surrounded by TAS mats at LR13 over the 1998- 2000 study period.Data from a separate study indicate that M.faveolata loses live tissues to marginal encroachment by TAS mats nearly as rapidly as documented above for D. strigosa, whereas S.siderea not only lost tissue at a lower rate,but some colonies have either maintained their original size or have expanded laterally into the mats (R.Roy 2004).
Chronic TAS mat encroachment is not as conspicuous as episodic natural disasters such as hurricanes or disease outbreaks.Nevertheless, it is important to ask why the mats are presently outcompeting what,until recently,have constituted several of the most important reef-framework builders on the Akumal-area fore reefs. Echinoids are important herbivores,potentially capable of removing TAS mats from the substratum (pers.obs.).Although there is a substantial population of D.antillarum on the patch reefs in Bahía Akumal,only in the summer of 2002 have their densities begun to increase on the fore reefs (Steneck pers.comm.).All else being equal, the relatively low densities of herbivore s in the Akumal-area reefs should facilitate the proliferation of benthic macroalgae.Their low rates of herbivory (overall averaging ~190 bites/m2/hour in 2000)are essentially identical to grazing rates recorded at 10 m in 1988 on the heavily over-fished north Jamaican reefs,where scrapers and browsers averaged 155 and 12.5 bites/m2/hour, respectively (Steneck 1993).
Territorial pomacentrids are known for their ability to cultivate and protect patches of turf algae from other herbivores (Vine 1974, Eakin 1988)unless overwhelmed by large schools of acanthurids and scarids (Ogden and Lobel 1978).When territorial damselfish are experimentally removed from reefs,other herbivores generally show a quick increase in abundance (e.g.,Hourigan 1986).At depths of 3-14 m in the Virgin Islands,Nemeth et al. (2003)found that the density of large-bodied herbivorous acanthurids and scarids was inversely related to the percentage of stony corals occupied by damselfish.This result may indicate some form of competitive exclusion of large herbivores by small territorial damselfish or,alternatively,is perhaps an artifact of the belt sampling method which underestimates large-bodied herbivores that school and move unpredictably (Nemeth et al.2003).
In the Akumal area,aggressively territorial pomacentrids (Stegastes fuscus,S.planifrons and S.partitus )averaged 23%and 28%of the total fish populations in 2000 at 13m and19m, respectively.Like acanthurids and scarids,dam-selfishes have doubtless experienced reduced predation from piscivores,especially groupers and snappers (Randall 1976);in contrast to the latter,they are unlikely to have been fished by humans.Their release from natural predation may have indirectly promoted the persistence of algal turfs in the Akumal area if aggressive pomacentrids are successfully excluding larger herbivores from the territories that they so vigorously defend.Moreover,sediment loading has likely increased along with the increase in human activity,and recent measurements of sedimentation rates near LR are rather high (Razo Amoroz 1999).Although TAS mats seldom develop when algal grazers are common, once established,the sediment that is bound by algal turfs may deter herbivorous fishes, Purcell (2000).
In conclusion,fringing reefs near Akumal have declined significantly in recent years. Between 1997 and 2000,M.faveolata incurred rapid losses from WP disease (data for LR13 demonstrate the value of repetitive monitoring before,during,and after major perturbations). In 2000,several attributes of fore-reef condition (for stony corals-live cover,density and relative exposure to TAS mats)were inversely related to an inferred sedimentation gradient at 13 m.These findings not only further support the known effects of sediment on coral communities but also reveal a relationship between sedimentation rates and the frequency of TAS mats in contact with stony corals.Although the displacement of D.strigosa and M.faveolata by TAS mats has been less obvious,its formation is currently facilitated by high sedimentation rates,a relative scarcity of herbivores (that is at least partially attributable to overfishing) and,possibly,by algal-gardening damselfish. Given the postulated relationships between large herbivorous fishes and territorial damselfish (see also Nemeth et al.2003),it would seem prudent to include the latter in the species lists for belt-transect rapid assessments (like the AGRRA method, http://coral.noaa.gov/agra/)to better document reef ecosystem dynamics.Community leaders in Akumal are already expending considerable effort to restrain environmentally destructive coastal development,support marine ecological research and initiate public outreach education.To reverse current declines,it may also be necessary to implement effective fishing bans,removal of territorial damselfish,and/or programs of artificial stock enhancement (for Diadema ,scarids or acanthurids).
Acknowledgments
Support for this project was kindly provided by the Raymond Foundation,Akumal Dive Shop,PADI Foundation,Sigma Xi,Seaspace, Zoology (now Integrative Biology)Department of the University of Texas at Austin,Centro Ecológico Akumal,and Great Outdoors Aqua Sports (Austin,Texas). J.Bibliowicz,J.Lang, C.Lawson,K.Ludlow,L.Milne,and L. Raymond are thanked for their assistance with the fieldwork.G.Arcila,D.Brewer,J. Bruno,J.Lang,D.Linton,S.Slingsby and R. Steneck contributed valuable information about Quintana Roo reefs.H.Moody is thanked for instruction in use of the Nikonos V.Thanks also to J.Lang for assistance with the preparation of this manuscript,and to other readers for their careful reviews.
Resumen
Para conocer el estado de las comunidades coralinas a lo largo de una costa en desarrollo,se realizó inventarios de transectos en cinco arrecifes de borde cerca de Akumal, México,con transectos de intersección (n=74).La cobertura de corales rocosos,la densidad y la exposición periférica relativa de las colonias y masas de algas-sedimentos son inversamente proporcionales a la gradiente de estrés de sedimentación inferida a 13m.En el 2000,la cobertura de corales rocosos vivos había decrecido un 40%en dos de los sitios estudiados por Muñoz-Chagín y de la Cruz-Agüero (1993).Alrededor de la mitad de esta pérdida aparentemente ocurrió entre 1998 y 2000 durante la epidemia de "blanqueo" que afectó principalmente a Montastraea faveolata y a M. annularis .En el sitio de 13m,donde las tasas de sedimentación inferida son relativamente altas,las fotografías en serie temporal de Diploria strigosa marcados (n=38)mostraron un promedio de pérdida de 70 cm2 de tejido vivo/coral/año y el cerco creciente de masas de algas durante el mismo periodo.Aunque las densidades de peces carnívoros y de organismos herbívoros (equinoideos, escáridos,acantúridos y Microspathodon chrysurus )en el 2000 fueron bajas en la franja a 10-19 m (n=106),se vio con frecuencia pomacéntridos que se alimentan de masas de algas.
Palabras clave:disminución de corales pétreos,masas de algas,herbivoría,enfermedad,arrecifes frontales.
References
Acevedo,R.&J.Morelock.1988.Effects of terrigenous sediment influx on coral reef zonation in southwestern Puerto Rico.Proc.8 th Int.Coral Reef Sym.2: 189-194. [ Links ]
Aronson,R.B.&W.F.Precht.2000.Herbivory and algal dynamics on the coral reef at Discovery Bay, Jamaica. Limnol.Oceanogr.45:251-255. [ Links ]
Asch,R.G.&D.D.Turgeon.2003.Detection of gaps in the spatial coverage of coral reef monitoring projects in the US Caribbean and Gulf of Mexico.Rev.Biol. Trop.(51 Supl.4):127-140. [ Links ]
Banaszak,A.T.,B.N.Ayala-Schiaffino,A.Rodríguez- Román,S.Enríquez &R.Iglesias-Prieto.2003. Response of Millepora alcicornis (Milleporina: Milleporidae)to two bleaching events at Puerto Morelos reef,Mexican Caribbean.Rev.Biol.Trop. 51(Supl.4):57-66. [ Links ]
Beltrán-Torres,A.U.&J.P.Carricart-Ganivet.1999. Lista revisitada y clave para los corales petreos zooxantelados (Hydrozoa:Milleporina;Anthozoa: Scleractinia)del Atlántico Mexicano.Rev.Biol. Trop.47:813-829. [ Links ]
Borger,J.L.2003.Three scleractinian coral diseases in Dominica,West Indies:distribution,infection patterns and contribution to coral tissue mortality.Rev. Biol.Trop.51 (Supl.4):25-38. [ Links ]
CARICOMP:Koltes,K.H.,F.R.Ruiz-Renteria,B. Kjerfve,S.R.Smith,G.Alleng,K.Bonair,D.Bone, K. Buchan, P. Bush, K. DeMeyer, J.Garzón-Ferreira, P.Gayle,J.Garcia,D.Gerace,E.Klein,R.Laydoo, H. Oxenford, C. Parker, A. Rodriguez, L. Sanchez, J.J,Tschirky &R.Varela.1997.Meteorological and oceanographic characterization of coral reef,seagrass and mangrove habitats in the wider Caribbean.Proc. 8 th Int.Coral Reef Sym.1:657-662. [ Links ]
Carpenter,R.C.1986.Partitioning herbivory and its effects on coral reef algal communities. Ecol. Mono. 56: 345-363. [ Links ]
Cróquer,A.&D.Bone.2003.Las enfermedades en corales escleractínidos:¿Un nuevo problema en el arrecife de Cayo Sombrero,Parque Nacional Morrocoy, Venezuela?Rev.Biol.Trop.51(Supl.4):167-172. [ Links ]
Cróquer,A.,E.Villamizar &N.Noriega.2002. Environmental factors affecting tissue regeneration of the reef-building coral Montastraea annularis (Faviidae)at Los Roques National Park,Venezuela. Rev. Biol. Trop.50:1055-1065. [ Links ]
Croquer,A.,S.M.Pauls &A.L.Zubillaga.2003.White plague disease outbreak in a coral reef at Los Roques National Park, Venezuela.Rev.Biol.Trop.51(Supl. 4):39-45. [ Links ]
Dustan,P.1977.Vitality of reef coral populations off Key Largo,Florida:Recruitment and mortality. Environ. Geol.2:51-58. [ Links ]
Dustan,P.1999.Coral reefs under stress:sources of mortality in the Florida Keys.Nat.Res.Forum.23: 147-155. [ Links ]
Eakin,C.M.1988.Avoidance of damselfish lawns by the sea urchin Diadema mexicanum at Uva Island, Panama.Proc.6 th Intl.Coral Reef Sym.2:21-26. [ Links ]
Fowler,A.J.1987.The development of sampling strategies for population studies of coral reef fishes.A case study.Coral Reefs.6:49-58. [ Links ]
García,A.,A.Cróquer &S.M.Pauls.2003.Estado actual de las enfermedades y otros signos de deterioro coralino en siete arrecifes del Parque Nacional Archipiélago de Los Roques,Venezuela.Rev.Biol. Trop.51(Supl.4):173-180. [ Links ]
Ginsburg,R.N.&P.W.Glynn.1994.Summary of the Colloquium and Forum on Global Aspects of Cora Reefs: Health,Hazards and History.p.220-226. In Ginsburg,R.N.(compiler),Proceedings of the Colloquium on Global Aspects of Coral Reefs: Health, Hazards,and History .Rosenstiel School of Marine and Atmospheric Science,University of Miami, Miami. [ Links ]
Gutiérrez-Carbonell,D.,M.Lara,C.Padilla,J.Pizaña, G.García,R.M.Loreto &T.Camarena.1995. Caracterización de los arrecifes coralinos en el corredor "Cancún-Tulum ",Quintana Roo,Mexico.Sian Ka an,Ser.Doc.4:3-39. [ Links ]
Hourigan,T.F.1986.An experimental removal of a territorial pomacentrid:effects on the occurrences and behavior competitors.Environ.Biol.Fish.15: 161-169. [ Links ]
Hubbard,J.A.E.B.&Y.P.Pocock.1972.Sediment rejection by recent Scleractinian corals;a key to paleoenvironmental reconstruction. Geol.Rundsch.61:598-626 [ Links ]
Jiménez,C.2001.Beaching and mortanity of ref organisms during a warning event in 1995 on the Caribbean coast of Costa Rica.Rev.Biol.Trop.49(Sup.2): 233-238. [ Links ]
Jordán Dhalgren,E.1993.Atlas de los arrecifes coralinos del Caribe Mexicano.Parte I.El Sistema Continetal. 110 p. [ Links ]
Kramer,P.,P.R.Kramer,E.Arias-Gonzalez &M.McField. 2000.Status of coral reefs of northern Central America: Mexico, Belize, Guatemala,Honduras, Nicaragua and El Salvador,p.287-313.In .C. Wilkinson (ed.).Status of Coral Reefs of the World –2000.Australian Institute of Marine Science. [ Links ]
Lang,J.,P.Alcolado,J.P.Carricart-Ganivet,M.Chiappone, A.Curran,P.Dustan,G.Gaudian,F.Geraldes,S. Gittings, R. Smith, W.Tunnell,&J.Weiner.1998. Status of coral reefs in the northern areas of the wider Caribbean,p.23-134. In .C.Wilkinson (ed.). Status of Coral Reefs of the World –1998.Australian Institute of Marine Science. [ Links ]
Lasker,H.R.1980.Sediment rejection by reef corals:The roles of behavior and morphology in Montastrea cavernosa (Linnaeus).J.Exp.Mar.Biol.Ecol.47: 77-87. [ Links ]
Lessios,H.R.1988.Mass Mortality Of Diadema antillarum In The Caribbean:What Have We Learned.Ann. Rev. Ecol. Syst. 19:371-393. [ Links ]
Loya,Y.1976.Recolonization of the Red Sea corals affected by natural catastrophes and man-made perturbations. Ecol. 57:278-289. [ Links ]
McGrath,T.A.&G.W.Smith.2003.Comparisons of the 1995 and 1998 coral bleaching events on the patch reefs of San Salvador Island,Bahamas.Rev.Biol. Trop.51 (Supl.4):67-75. [ Links ]
Miller,J.,C.Rogers &R.Waara.2003.Monitoring the coral disease,plague type II,on coral reefs in St. John,U.S.Virgin Islands. Rev.Biol.Trop.51(Supl. 4):47-55. [ Links ]
Motulsky,H.1995.Intuitive Biostatistics.Oxford University,New York.386 pp. [ Links ]
Muñoz-Chagin,R.F.&G.de la Cruz-Aguera.1993.Corales del Arrecife de Akumal,Quintana Roo.p.761-771. In Biodiversidad Marina y costera de Mexico.S.I. Salazar-Vallejo and N.E.Gonzalez (eds.).Com.Nal. Biodiversidad y CIQRO,Mexico. [ Links ]
Nemeth,R.S.,L.D.Whaylen &C.V.Pattengill-Semmens. 2003.Rapid assessment of coral reefs in the Virgin Islands (Part 2: Fishes).pp.566-589.In Lang,J.C. (ed.).Status of Coral Reefs in the western Atlantic: Results of initial Surveys,Atlantic and Gulf Rapid Reef Assessment (AGRRA)Program.Atoll Res. Bull.496. [ Links ]
Nolasco-Montero,E.&A.Carranza-Edwards.1988. Estudio sedimentologico regional de playas de Yucatan y Quintana Roo,Mexico.An.Inst.Cienc. Mar.Limnol.UNAM 15:49-66. [ Links ]
Ogden,J.C.&P.S.Lobel.1978.The role of herbivorous fish and urchins in coral reef communities. Env.Biol. Fish.3:49-63. [ Links ]
Purcell,S.W.2000 Association of epilithic algae with sediment distribution on a windward reef in the Northern Great Barrier Reef,Australia.Bull.Mar. Sci 66:199-214. [ Links ]
Randall,J.E.1976.Food habits of the reef fishes of the West Indes.Studies in Trop.Oceanogr.(Miami)5: 665-847. [ Links ]
Razo Amoroz,I.1999.El Sedimento como factor de mortalidad en corales escleractinios de un macizos arrecifal de la costa de Quintana Roo.Master s Thesis. Centro de Investigacion y de Estudios Avanzados del Instituto Politecnico Nacional Unidad Merida.69p. [ Links ]
Reichelt,R.E.,Y.Loya &R.H.Bradbury.1986.Patterns in the use of space by benthic communities on two coral reefs of the Great Barrier Reef Australia.Coral Reefs 5:73-80. [ Links ]
Roy,R.2004.Turf algal/sediment (TAS)mats:A chronic stressor in Scleractinian corals in Akumal, México. Ph. D. Dissertation, University of Texas at Austin, Austin,TX 171 p. [ Links ]
Scoffin,T.P.1970.The trapping and binding of sub-tidal carbonate sediments by marine vegetation in Bimini lagoon, Bahamas. J. Sed.Petrol.40:249-273. [ Links ]
Stafford-Smith,M.G.1993.Sediment-rejection efficiency of 22 species of Australian scleractinian corals.Mar. Biol. 115:229-243. [ Links ]
Steneck,R.S.1985.Adaptations of crustose coralline algae to herbivory:Patterns in space and time.p.352-366. In D.Toomy &M.Nitecki (eds.).Paleoalgology. Springer-Verlag,Berlin. [ Links ]
Steneck,R.S.1988.Herbivory on coral reefs:A synthesis. Proc.6th Intl.Coral Reef Sym.1:37-49. [ Links ]
Steneck,R.S.1993.Is herbivore loss more damaging to reefs than hurricanes?Case studies from two Caribbean reef systems (1978-1988).p.i-viii. In R.N.Ginsburg (compiler).Proceedings of the Colloquium on Global Aspects of Coral Reefs: Health,Hazards,and History .Rosenstiel School of Marine and Atmospheric Science,University of Miami,Miami. [ Links ]
Steneck,R.S.&J.C.Lang.2003.Rapid assessment of México s Yucatán reef in 1997 and 1999:pre- and post-1998 mass bleaching and Hurricane Mitch (stony corals,algae and fishes).pp.294-317.In J.C.Lang (ed.).Status of Coral Reefs in the western Atlantic:Results of initial Surveys,Atlantic and Gulf Rapid Reef Assessment (AGRRA)Program.Atoll Res.Bull.496. [ Links ]
Vine,P.J.1974.Effects of algal grazing and aggressive behaviour of the fishes Pomacentrus lividus and Acanthurus sohal on coral-reef ecology.Mar.Biol.24:131-136. [ Links ]
Weil,E.&N.Knowlton 1994.A multi-character analysis of the Caribbean coral Montastraea annularis (Ellis and Solander, 1786)and its two sibling species,M. faveolata (Ellis and Solander,1786)and M.franksi (Gregory, 1895). Bull.Mar.Sci.55:151-175. [ Links ]
Wilkinson,C.2000.Executive Summary,p.7-19.In C. Wilkinson (ed.).Status of Coral Reefs of the World –2000.Australian Institute of Marine Science. [ Links ]












 uBio
uBio