Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.51 n.2 San José Jun. 2003
Gbemisola A. Akin-Oriola 1
Received 24-X-2001. Corrected 11-I-2002. Accepted 26-III-2002.
Abstract
Zooplankton abundance, composition and environmental parameters were monitored in two tropical rivers over a twenty month period. The data was subjected to cluster, factor and correlation analysis to determine the grouping pattern of zooplankton and their relationship to environmental parameters. Environmental factors in Ogunpa and Ona rivers -included buffering capacity, trace metal ions, pH-temperature/ transparency- were primarily influenced by rainfall. The dominance of the Rotifera in both rivers was attributed to their short developmental rate and fish predation on larger zooplankton. Two groups of associations were identified in each river – a commonly occurring species group exhibiting strong homogenous correlation with environmental factors and a predominant group exhibiting weak correlation with environmental factors and whose abundance / composition may be defined by biotic factors.
Key words: Zooplankton, environmental parameters, rivers, Nigeria, cluster, factor and correlation analysis.
In Nigeria the study of zooplankton has been developed mostly in large rivers and lakes. Such studies have focussed on descriptive ecology e.g. faunal composition, spatial and temporal distribution and plankton succession in newly-formed reservoirs (Green 1960, Clarke 1978, Egborge and Tawari 1987, Segers 1993). Less attention has been given to smaller rivers which are scattered all over the country and contain a significant proportion of the nations aquatic biodiversity. The aim of the present work was to provide information on the zooplankton communities of two rivers in relation to environmental conditons.
Materials and methods
Zooplankton were sampled with a 60 µm plankton net fortnightly between August 1992 and March 1994. Samples were preserved in 4% neutral formalin and identification was carried out using Ward and Whipple (1966), Jeje and Fernando (1986).
In situ measurements of temperature and transparency were taken while dissolved oxygen, carbon dioxide, alkalinity and ammonia-nitrogen were determined in the laboratory according to APHA (1985). Conductivity, pH and total dissolved solids were measured using ATC probes (Hanna Instruments, USA) while trace metal analysis was carried out by atomic absorption spectrophotometry.
Zooplankton abundance data were clustered using Euclidean distance and complete linkage method (Genstat 5, release 3.2). Principal component analysis of environmental parameters was carried out and the resulting factors were correlated with zooplankton associations.
Study area. The city of Ibadan in south western Nigeria (7º23 N, 3º5 E) is the largest urban centre in Africa south of the Sahara (Areola 1994). It is characterized by a West African monsoon type of climate with hot, dusty dry season (November–April) and cold, humid rainy season (May–October). The Ogunpa and Ona rivers are two major rivers draining the city. The markedly seasonal variations in temperature and rainfall result in intermittent flow in the Ogunpa with sections of the river becoming lentic in the dry season. The Ogunpa river system is a third-order stream with a channel length of 21.5 km and area 73.3 km2 draining the densely populated eastern part of Ibadan city. The Ona river has a length of 55 km and area of 81.0km2 and it flows through the low density western part. Five sites were sampled on each river.
Results
Species Composition. Forty-nine species were identified from both rivers (Table 1) and they were composed of Protozoa (6), Rotifera (32), Cladocera (6) and Copepoda (4). The zooplankton fauna of Ogunpa and Ona rivers were dominated by the Rotifera while the family Brachionidae recorded the highest species richness. Although, most of the species identified indicated a typical tropical assemblage, predominantly temperate genera like Synchaeta and Notholca were recorded. As in most tropical freshwaters, the cladoceran fauna included Diaphanosoma excisum and Ceriodaphnia cornuta however Moina micrura was not recorded. The genus Daphnia was absent and this is typical for tropical waters. Scapholeberis kingi was recorded only in Ona river. The Cyclopoda were the most abundant group of copepods in Ogunpa and Ona rivers with three species: Thermocyclops neglectus, Eucyclops serrulatus and Mesocyclops sp.

Cluster Analysis. The original zooplankton data consisted of forty–nine species however organisms with only one or two occurrences and < 2000 ind.l -1 throughout the study period were eliminated to facilitate cluster analysis. Hierarchical clustering (Figs. 1 and 2) suggests that the plankton in each river can be partitioned into distinct species associations or groups. These associations vary in size and exhibit heterogeneity in structure.
In Ogunpa river, at a level of 90%, two groups were formed G1 and G2 while Philodina sp. was ungrouped (Fig. 1). Group 1 was composed of three species (Paramecium, Lecane (L) luna and Mesocyclops) while Group 2 was composed of a mixed assemblage of zooplankton with common occurrence although E. senta, a typical predominant species was also included in this group.
The grouping pattern in Ona river was more distinct. At a level of 85%, a group G3 was formed made up of rare / commonly occurring rotifers and copepods. A second group G4 was formed at 80% level with predominant species such as B. c. anuraeiformis, Lecane luna, P. dolicoptera, F. opoliensis, Mesocyclops sp. and Difflugia sp. while Philodina sp. remained ungrouped.
The composition of zooplankton in groups G2 and G3 were similar and about 70% of the species are common.
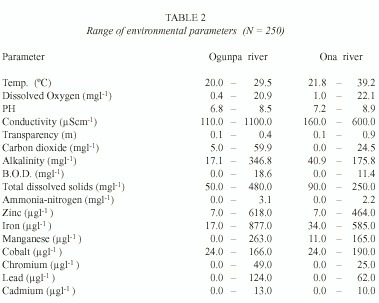
Environmental Factors. The data on environmental parameters presented in Table 2 showed wide variations in values. Zinc, iron and manganese recorded higher concentrations than other trace metals. Factor analysis (Tables 3 and 4) showed that buffering capacity was the single most important factor influencing conditions in both rivers.
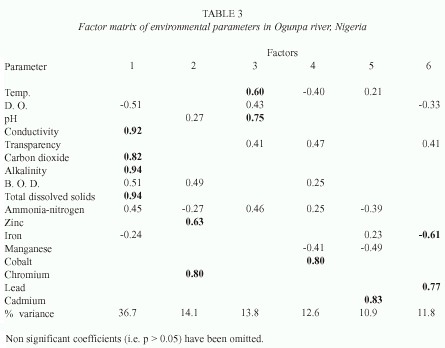
In Ogunpa river, buffering capacity (factor 1) accounted for 36.7% of variance while trace metals (factors 2, 4, 5 and 6) had a cumulative score of 49.4% (Table 3). Factor 3 which is pH-temperature accounted for 13.8% of variation.
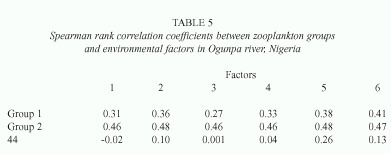
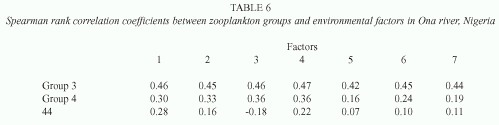
In Ona river, a similar result was obtained with buffering capacity (factor 1) accounting for 28.2% of variation while trace metals (factors 2, 4, 5, and 6) recorded a cumulative score of 48.2%. Factor 3 i.e. pH- transparency accounted for 12.0% while dissolved oxygen constituted an additional factor with a high negative loading on factor 7 (Table 4). Tables 5 and 6 show rank correlations between zooplankton groups and derived environmental factors. Groups 2 and 3 exhibited highest correlations with all factors while Philodina sp. (44) recorded consistently low or negative correlation with factors.
Discussion
Species Composition. The dominance of the Rotifera in Nigerian aquatic ecosystems has been documented by several authors (Green 1960, Jeje and Fernando 1986, Egborge and Tawari 1987). The predominant genus is usually Brachionus, Keratella or Lecane. The high population densities of rotifiers has been attributed to their parthenogenetic reproductive pattern and short developmental rates under favourable conditions (Herzig 1983, Pourriot et al. 1997).
The low species spectra of Cladocera recorded in this study was also observed by Jeje and Fernando (1992) in the Niger-Sokoto river. Although blue-green algae are abundant in Ogunpa and Ona in the dry season (Oduwole 1997), these cannot be effectively grazed upon by the filter-feeding Cladocera. Size-selective fish predation and the inadequacy of residence time of the organisms may also contribute significantly to their low numbers. In river environments due to the short residence time, only organisms with rapid growth and high renewal rate can increase their populations (Pourriot et al. 1997). However, the lack or paucity of planktonic Cladocera has also been reported from lakes in Cameroun, Uganda and South Africa (Green 1972, Burgis 1973, Fowles and Archibald 1987, Martin and Cyrus 1994) thus more information is needed on feeding guilds and size structure of zooplankton as well as the dominant fish assemblages in order to fully explain this phenomenon in African water bodies.
Environmental factors. The onset of the rains signals a radical change in physico-chemical characteristics of tropical rivers (Lowe-McConnell 1987, Chapman and Kramer 1991). The input of allochthonous organic materials from the catchment area during rainfall increases conductivity, pH, alkalinity, total dissolved solids and biochemical oxygen demand. The buffering capacity factor is thus a direct response to rainfall hence its high contribution (about 1/3) to variation in both rivers. Zooplankton abundance and population dynamics have been reported to be influenced by repeated environmental fluctuations of which rainfall is a primary steering factor (Kizito and Nauwerck 1995, Osore et al. 1997).
The higher levels of iron, zinc and manganese is expected since the basement complex of Ibadan is composed of preCambrian metamorphic rocks rich in zinc, iron and manganese ores (DHoore 1964). However, trace metal speciation is much more important in terms of biological availability and / or toxicity than total concentrations. Metal speciation is mediated by physico-chemical characteristics of the water, organic and inorganic complexation, adsorption and redox processes (Zhang and Huang 1993). Hence the contribution of trace metals to variance is coupled to environmental conditions.
Ecological implications of associations.
Associations are composed of species that have similar reactions to properties of the environment. This means that such species are responding in a related way to environmental changes (Legendre 1973). In order to identify cooccurring species, grouping was carried out and species groups or associations were correlated with environmental factors. In each river, two distinct groups were recognized: 1) groups 2 and 3 which consisted of commonly occurring species exhibiting homogenous correlation with all derived factors. 2) groups 1 and 4 comprising species which were predominant but showed weak correlation with most environmental factors. These results imply that for groups 2 and 3 environmental factors may play a key role in their abundance and community structure while biotic factors such as competition, predation and grazing patterns may define population dynamics in groups 1 and 4.
However, it is difficult to establish one-to-one causal relationships between zooplankton associations and factors without supporting experimental evidence.
Acknowledgments
This study was supported in part by the IDRC / AAU award for Environmental Research (1995/96).
Resumen
La abundancia, y composición del zooplancton, y parámetros ambientales fueron monitoreados en dos ríos tropicales durante un período de doce meses. Los datos fueron sometidos a análisis de agrupaciones, de factores y de correlación para determinar los patrones de agrupamiento del zooplancton y sus relaciones con parámetros ambientales. Los factores ambientales en los ríos Ogunpa y Ona, incluidos la capacidad tampón, iones metálicos traza, pH-temperatura/ transparencia, fueron influenciados principalmente por la lluvia. La dominancia de los Rotíferos en ambos ríos fue atribuida a su corta tasa de desarrollo y a la depredación del zooplancton grande por peces. Dos grupos de asociaciones fueron identificadas en cada río – la presencia de un grupo común de especies que exhiben una fuerte y homogénea correlación con factores ambientales y un grupo predominante que exhibe una débil correlación con factores ambientales y cuya abundancia / composición que podría estar definida por factores bióticos.
References
Anonymous. 1985. Standard methods for the examination of water and wastewater. APHA, Washington D. C. 1268. [ Links ]
Areola, O. 1994. The spatial growth of Ibadan city and its impact on the rural hinterland, p. 98-106. In M.O Filani, F.O. Akintola & C.O. Ikporukpo, (eds.). Ibadan Region. Rex Charles, Ibadan. [ Links ]
Burgis, M.J. 1973. Observations on the Cladocera of lake George, Uganda. J. Zool., (Lond.) 17: 339-349. [ Links ]
Chapman, L.J. & D.L. Kramer. 1991. Limnological observations of an intermittent tropical dry forest stream. Hydrobiol. 226: 153-166. [ Links ]
Clarke, N.V. 1978. A comparison of the zooplankton of lake Kainji and of the rivers Niger and Swashi. Hydrobiol. 58: 17-23. [ Links ]
DHoore, L.J. 1964. Soil map of Africa. C.C.T.A. Lagos, Nigeria. [ Links ]
Egborge, A.B.M. & P. Tawari. 1987. The rotifers of Warririver, Nigeria. J. Plankton Res. 9: 1-13. [ Links ]
Fowles, B.K. & C.G.M. Archibald. 1987. Zooplankton and estuarine relict fauna in lake Mzingazi, a fresh-water coastal lake of Natal. J. Limnol. Soc. S. Afr. 13: 66-74. [ Links ]
Green, J. 1960. Zooplankton of the river Sokoto. The Rotifera. Proc. Zool. Soc. Lond. Vol. 135: 491-523. [ Links ]
Green, J. 1972. Ecological studies on crater lakes in West Cameroun. Zooplankton of Barombi Mbo, Mboandongi, lake Kotto and lake Soden. J. Zool.166: 288-301. [ Links ]
Herzig, A. 1983. Comparative studies on the relationship between temperature and duration of embryonic development of rotifers. Hydrobiol. 104: 237-246. [ Links ]
Jeje, C.Y. & C.H. Fernando. 1986. A practical guide to the identification of Nigerian Zooplankton (Cladocera, Copepoda and Rotifera). Kainji Lake Res. Inst. 142. [ Links ]
Jeje, C.Y. & C.H. Fernando. 1992. Zooplankton associations in the middle Niger-Sokoto basin (Nigeria: West Africa). Int. Revue ges. Hydrobiol. 77 (2): 237-253. [ Links ]
Kizito, Y.S. & A. Nauwerck. 1995. Temporal and vertical distribution of planktonic rotifers in a meromictic crater lake, Lake Nyahirya (Western Uganda). Hydrobiol. 313/314: 303-312. [ Links ]
Legendre, L. 1973. Phytoplankton organisation in Baie des Chaleurs (Gulf of St. Lawrence). J. Ecol. Vol. 61 (1): 135-149. [ Links ]
Lowe-Connell, R.H. 1987. Ecological studies in fish communities. Cambridge. 3-33. [ Links ]
Martin, T.J. & D.P. Cyrus. 1994. Zooplankton in the open water of lake Cubhu, a freshwater coastal lake in Zululand, South Africa. Wat. S. Afr. 20: 107-112. [ Links ]
Oduwole, G.A. 1997. Indices of pollution in Ogunpa and Ona rivers, Ibadan: Physico-chemical, trace metal and plankton studies. Ph.D. Thesis, University of Ibadan, Nigeria. 236 p. [ Links ]
Osore, M.K., M.L. Tackx & M.H. Daro. 1997. The effect of rainfall and tidal rhythm on community structure and abundance of the zooplankton of Gazi bay, Kenya. Hydrobiol. 356: 117-126. [ Links ]
Pourriot, R., C. Rougier & A. Miquelis. 1997. Origin and development of river zooplankton: example of the Marne. Hydrobiol. 345: 143-148. [ Links ]
Segers, H. 1993. Rotifera of some lakes in the floodplain of the river Niger (Imo state, Nigeria) I. New species and other taxonomic considerations. Hydrobiol. 250: 39-61. [ Links ]
Ward, H.B. & G.C. Whipple. 1966. Freshwater biology. Wiley. 1248 p. [ Links ]
Zhang, J. & W.W. Huang. 1993. Dissolved trace metals in the Huanghe: the most turbid large river in the world. Wat. Res. 27(1): 1-8. [ Links ]
1 School of Life Sciences, Robert Gordon University. St Andrews Street, Aberdeen AB25 1HG; gakinoriola@yahoo.com ; ext.akinoriola@rgu.ac.uk