Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.49 no.2 San José jun. 2001
(Cucurbitaceae) extracts on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae
Fernando Mancebo1Luko Hilje2* Gerardo A. Mora3 Víctor H. Castro3 Rodolfo Salazar4
Received 7-VI-2000. Con-ected 27-X-2000. Accepted 13-XI-2000.
Abstract
Biological activity of a plant extract (common rue, Ruta chalepensis) and a semi purifíed fraction (from "tacaco cimarrón", Sechium pittieri) on mahogany shootborer larvas (Hypsipyla grandella) was studied. A randomized complete block design, with four replications, was used. H. grandella third instar larvas were exposed for 24 h to Cedrela odorata leaf discs dipped in several treatment dissolutions of each extract (0.1, 0.32, 1.0, 3.20, and 10%); afterwards, each larva was transferred to a flask containing an artificial diet and was allowed to complete its development. Variables measured included food consumption (foliar area eaten in 24 h), mortality, and developmental effects (developmental time for each larval instar and the pupa, and pupal weight). The common rue extract showed a clear antifeedant activity at a concentration as low as 0.32%, whereas the "tacaco cimarrón" fraction caused toxicity, especially at the two highest concentrations (3.20 and 10%).
Key words
Hypsipyla grandella, mahogany shootborer, Meliaceae, plant extracts, common rue, "tacaco cimarrón", antifeedant, mortality.
The mahogany shootborer, Hypsipylagrandella (Zeller) (Lepidoptera: Pyralidae), is a key Neotropical forest pest, because it causes harm to precious wood trees of the Meliaceae family, such as mahoganies (Swietenia spp.) and cedars (Cedrela spp.) (Schabel el al. 1999). This pest bores into terminal shoots of young host plants, breaking the apical dominance, which causes forking of the stems and excessive production of lateral branches, rendering trees unmarketable. Therefore, it has precluded attempts to establish commercial plantations of mahoganies and cedars in Latin America and the Caribbean (Grijpma and Ramalho 1973).
In spite of a considerable amount of research aimed at managing H. grandella (Grijpma 1973, Whitmore 1976a, b, Newton et al. 1993, Mayhew and Newton 1998), management practices have not been feasible. This is so because a single borer larva can destroy the apical meristem, giving rise lo trees of poor form for utilization as timber. Thus, this low damage threshold calls for a preventive approach, in which either deterrents or repellents could play an important role, along with the planting of tolerant genotypes (Mesén 1999) and other biointensive practices (Newton et al. 1993, Speight 1997).
As a result of insect-plant coevolution, substances acting as insect deterrents and repellents are expected to be more common in nature than those causing acute mortality (Isman 1999). In fact an antifeedant effect on H. grandella larvae has been shown to occur with both wood and leaf extracts of the bitterwood tree (Quassia amara L. ex Blom, Simaroubaceae) (Mancebo et al. 2000a). Since H. grandella is specific to Meliaceae, It is very likely that chemical principles present in plants belonging to other families may act as deterrents, repellents, growth disrupters or insecticides (Mancebo et al. 2000b). In preliminary trials, methanolic extracts of foliage of common rue (Ruta chalepensis L. Rutaceae) and fruits of "tacaco cimarrón" (Sechium pittieri (Cogn.) C. Jeffrey, Cucurbitaceae) reduced feeding by H. grandella larvae (Mancebo et al. 2000b), but it was unknown if this happened because of deterrence or toxicity. Therefore, the objective of this research was to determine, through laboratory bioassays, possible antifeedant or insecticidal effects of both extracts, as well as minimum concentrations causing these effects, on H. grandella larvae.
Materials and Methods
Plant material included common rue (R. chalepensis) foliage, collected from Turrialba, and fruits of "tacaco cimarrón" (S. pittieri), from Orosi, both locations in Cartago, Costa Rica. Methanolic extracts of common rue were prepared along with other plant extracts to be tested against H. grandella (Mancebo et al. 2000b), whereas the semi purified fraction of "tacaco cimarrón" was already prepared for other kind of experiments (Castro et al. 1997)
Common rue extracts were prepared at CIPRONA (Research Center on Natural Products), as follows: Plant material was dried in an oven at 40°C, ground and placed in 70% methanol in a suitable flask for 24 h; the solvent was drained and the residue was treated again with methanol for 24 h. The pooled extracts were filtered through a Whatman No. 4 filter paper, and concentrated at 40°C using a rotary evaporator. The final residue was freeze-dried to eliminate any water remaining in the crude extract. In the case of "tacaco cimarrón", the fresh fruits were extracted with methanol and the extract was concentrated in vacuo to give an aqueous suspension which was passed through a column of MCI gel Diaion HP-20. The column was washed with water, 50% methanol/water, methanol and ethyl acetate. The third fraction was concentrated by evaporation in vacuo.
Laboratory bioassays were carried out at CATIE, in Turrialba, Costa Rica, in two environmental chambers (Percival I-35L) set at 22°C, 80-90% RH, and 12: 12 (L: D) photoperiod.
Hypsipyla grandella larvae were taken from colonies maintained at CATIE, where they were initially reared on tender foliage of Spanish cedar (Cedrela odorata) and later transferred to an artificial diet (Vargas and Shannon, unpublished). Third-instar larvae, which had been fed exclusively on cedar foliage, were selected because their size allowed easy handling.
Bioassays included treatment with both plant extracts, at five increasing concentrations of each extract (0.1, 0.316, 1.0, 3.162, and 10%) mixed with a surfactant (Nu film 17, at 0.03%). They were compared to two relative controls (70% methanol, and Nu film 17 at 0.03%), and an absolute control treatment (distilled water). All dissolutions were prepared just before the experiment was set up, with distilled water as a carrier.
Disks of Spanish cedar tender foliage (2.3 cm in diameter) were cut with a cork-borer, dipped in the selected treatment for 10 sec, and allowed to dry for 30 min. Treated disks were placed individually in 30 ml glass flasks, along with a third-instar H. grandella larva which had been deprived of food for 3 h. A piece of paper towel was fastened with the lid of each flask and was moistened periodically, in order to retain leaf turgor.
A randomized complete block design, with four replications, was used. The experimental unit consistes of seven larvae, except in the control (14 larvae). Blocks were represented by plastic trays, and flasks representing each treatment were randomized within each tray.
After being exposed to the treatment for 24 h, each larva was transferred to a flask containing about 6 ml of artificial diet, where it was allowed to complete its development; larvae were transferred to other flasks in cases where it was judged that the diet was not suitable for their development.
Three types of variables were measured in response to plant extract treatinents: food consumption, mortality, and developmental effects. Food consumption was assessed for each disk, by recording the percentage of foliar area that was consumed in 24 h. This was done by means of a visual scale of the program Distrain 1.0 (Tomerlin and Howell 1988). Mortality was determined for each larva every 24 h, and the instar at which mortality occurred was recorded; cessation of movement and color change to black were the criteria used for judging mortality. Developmental effects included developmental time for each larval instar and the pupa, as well as pupal weight on the day after pupation; dates for larval moulting, moulting into pupae and adult emergence were recorded.
Data were analyzed by means of ANOVA procedures, and means were compared by the Tukey's test, at a significance level of a= 0.05. For leaf consumption, analyses were performed with the original data, since even after transfortnation with various approaches (the arcsin, logarithmic, and square root methods), the distribution of the data did show significant deviations from normality. In addition, regression analyses were performed for leaf consumption in response to plant extract concentrations.
Results
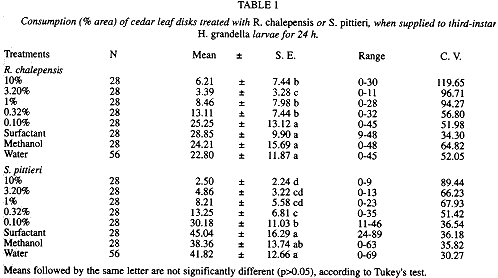
Food consumption: In terms of leaf disk consumption by H. grandella larvae, there were very large differences between treatments for both common rue (F= 26.23, d.f= 7, 21, p < 0.0001) and "tacaco cimarrón" (F= 82.95. d.f= 7, 21, p < 0.0001).
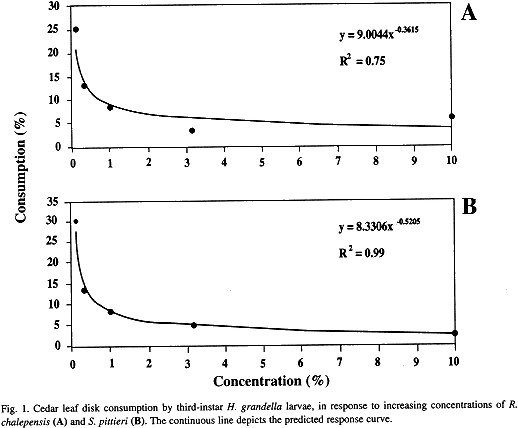
For the common rue extract, there were three groups of treatments, wiúi the lower consumption averages attained at the 3.16%, followed by 0.32, 1.0 and 10% concentrations, which did not differ among themselves (Table 1). The 0.1% concentration did not differ from the control treatments. The response curve for the extract concentrations and leaf disk consumption was best fitted by a potencial model (Fig. 1A). For the "tacaco cimarrón" fraction, all treatments differed from the control treatments, excepting the 0.1% concentration, which did not differ from the methanol treatment (Table 1). Leaf consumption averages decreased as concentrations increased, with the response curve being best fitted by a potential model (Fig. 1B).
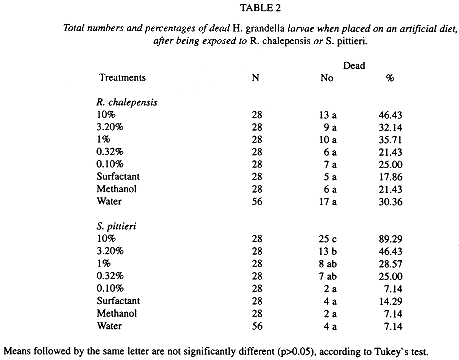
Larval mortality: In terms of larval mortality, there were no differences between treatments for the common rue extract (F= 1.14, d.f= 7, 21, p > 0.05). Mortality values ranged from 21-46% of the exposed larvae to common rue treatments (Table 2), with death occurring by the first day after exposure.
On the contrary, for the "tacaco cimarrón" fraction, differences between treatments were very large (F= 29.21, d.f= 7, 21, p < 0.0001) (Table 2). Mortality ranged from 7-89% of the exposed larvae, with the higher values (89 and 46%) occurring at the highest 10 and 3.16% concentradons, respectively. Its value at the 10% concentration differed from the other concentrations, whereas that at 3.16% did not differ from the two subsequent lower concentrations (1 and 0.32%).
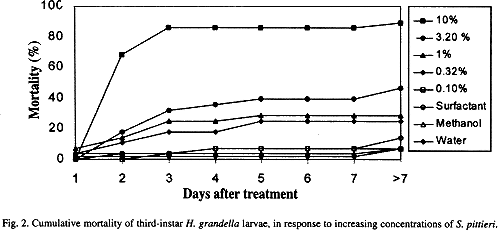
Even trough larval mortality occurred at different intervals, for die 10% concentration 68% of the larvae were dead one day after exposure, and 85% of mortality was attained by day 3 (Fig. 2). For the following three concentrations, mortality values as high as 32,25 and 17% were achieved by day 3 (at 3.16, 1.0 and 0.32%, respectively) and mortality remained quite stable from then on. In the control treatments, as well as at the 0. 1 % concentration, mortality was very low, with a maximum of 14%.
Developmental effects: For the common rue extract, developmental times for both 4th (F= 0.65, d.f= 7, 21, p > 0.05) and 5th larval instars (F= 0.77, d.f.= 7, 21, p > 0.05), as well as for pupae (F= 1.07, d.f.= 7, 21, p > 0.05), did not differ between treatments. Pupal weight, which ranged from 0. 115-0.133 g, did
not differ between treatments (F= 0.35, d.f.= 7, 21, p > 0.05). These trends held for the "tacaco cimarrón" fraction, with the following values: 4th instar (F= 0.45, d.f.= 7, 21, p > 0.05), 5th instar (F= 1.04, d.f= 7, 21, p > 0.05), pupae (F= 1.27, d.f.= 7, 21, p > 0.05), and pupal weight (F= 1.73, d.f.= 7, 21, p > 0.05); pupal weight values ranged from 0. 172-0.189 g.
Discussion
The selected methodological approach allowed to clearly discriminate between antifeedant and insecticidal effects in response to plant extracts, both of which were demonstrated.
Substances present in the common rue extract showed antifeedant activity against H. grandella larvae, at the highest four concentrations of this extract. Evidence of antifeedant was that at a concentration as high as 0.32% of the extract, larvae barely consumed treated leaf disks and showed low mortality once they were transferred to artificial diet. This was also shown for wood and leaf extracts of bitterwood (Quassia amara, Simaroubaceae) in parallel experiments (Mancebo et al. 2000a). H. grandella larvae possess deterrent receptors in a sensilla styloconica located on the maxillae (Schoonhoven 1980).
Antifeedant effect by common rue extracts was also demonstrated for the Colorado potato beetle, Leptinotarsa decemlineata (Coleoptera: Coccinellidae) 4th instar larvae and adults (Hough-Goldstein 1990), and repellency was shown for the cat flea, Ctenocephalides canis (Siphonaptera: Pulicidae) (Cox 1980). Common rue foliage contains a number of chemicals, such as fenilpropanoids (anethol glycol), benzenoids (anisic acid), quinoline and acridone alkaloids, terpenoids (bergamoten derivatives) and coumarins (bergapten) (Torres 1950, Vasudevan and Lukner 1968, Kong et al. 1984), but it remains unknown if any of them are responsible for causing either antifeedant or repellent activities.
Concernig other effects, none of the concentrations tested affected neither development of immature stages nor pupal weight, and there was no evidence of toxicity. Richter et al. (1990) showed that ethanolic extracts of common rue foliage affect molting duration in the last nymphal instar of the American cockroach Periplaneta americana (Blattaria: Blattidae). Also, Sasanelli (1997) tested aqueous extracts of common rue foliage and roots on several nematode species and found that the former highly reduced hatching in Meloidogyne spp.
In regards to the "tacaco cimarrón" fraction, its effect in reducing leaf disk consumption at the highest concentrations was due not to antifeedant activity, but to larval mortality. Larval death occurred quickly at the highest concentration (10%), whereas at the other concentrations mortality was lower and delayed, possibly due to consumption of smaller quantities of foliage that were treated with lower concentrations of the fraction. Plant chemicals responsable for causing mortality remain unknown, although probably they are a series of glycosides known as tacacosides, which are very bitter and irritating. Six of these bayogenin saponins have been isolated from fruits and aerial parts of S. pittieri and S. talamancense (Castro et al. 1997), in an effort to look for antiproliferative principies in neotropical plants. Cucurbitacins, which have severas kinds of activities, including toxicity and feeding deterrence (Mabry and Gill 1979), were not found in this fraction nor in the plant.
In summary, these findings substantiate the presence of antifeedant or toxic principles against H. grandella larvae, in plants belonging to families taxonomically unrelated to Meliaceae, such as Rutaceae and Cucurbitaceae, as well as in Simaroubaceae (Mancebo et al. 2000a). Nonedieless, even pantropical Meliaceae, such as the neem tree (Azadirachta indica A. Juss.) harbor either growth disrupting or toxic principles against such neotropical pest (Mancebo et al. 2000c). H. grandella is a rather monophagous insect, which is restricted to at least 17 neotropical Meliaceae species (Becker 1976).
Those toxic principles, as well as others present in other tropical plant species (Mancebo et al. 2000b), could be a source of promising chemicals to be used either as crude extracts by resource-poor growers, or as more refined formulations. Nevertheless, the role of plant extracts in integrated pest management (IPM) programs for H. grandella would make sense as long as they could contribute to developing preventative management schemes for this insect. For a pest with a very stringent damage threshold (i.e. one larva per tree), like H. grandella, convencional chemical control is limited because of the high cost of repeated applications through many years, as well as some operational factors, such as inaccessibility of the larvae, high rainfall, and application methods (Newton et al.1993). Therefore, novel systemic substances preventing newly emerged larvae from entering the shoots and causing irreversible damage would be preferable.
Thus, if their systemic effect is demonstrated, either antifeedant or toxic substances present in common rue and "tacaco cimarrón", respecfively, could be formulated as controlled-release materials. Some systemic insecticides, such as methomyl and carbofuran, were applied in pellet form, at planting of Spanish cedar trees, and provided complete control of H. grandella for several months (Allan et al. , 1973; Wilkins et al. , 1976). Currently, there are semi-rustic methods for manufacturing these types of formulations (Richard M. Wilkins 1998, Newcastle University, England, pers. comm.), which could allow lo make low cost formulations, especially suited for small farmers in the tropics.
Protection from H. grandella is especially important during the first 5-8 years of tree development, depending on the region (Cibrián et al. 1995). Thus, in order to protect trees during this critical period of susceptibility, fomulated plant extracts could be applied at transplantation, in complementarity with other IPM preventative approaches, such as the deployment of tolerant genotypes, silvicultural practices, and biological control (Newton et al. 1993, Speight 1997, Mesén 1999).
Acknowledgements
This paper is a partial result of a CATIE's M.Sc. thesis by the first author, which was funded by Fundatrópicos and IICA (Instituto Interamericano de Cooperación para la Agricultura). Thanks to Bernal Valverde, Phillip J. Shannon, Carlos Vargas, Johnny Pérez, Francisco López, Douglas Cubillo, Guido Sanabria, Arturo Ramírez, Claudio Arroyo, Alfonso González, Marvin Hemández, Manuel Carballo and Eduardo Molina (CATIE), as well as to Juan Carlos Brenes (CEPRONA), for their intellectual or logistical support.
Resumen
Se estudió la actividad biológica de un extracto de follaje de ruda (Ruta chalepensis) y de una fracción semipurificada de "tacaco cimarrón" (Sechium pittieri) sobre las larvas del gusano barrenador de las meliáceas (Hypsipyla grandella). Se utilizó un diseño de bloques completos al azar, con cuatro repeticiones. Durante 24 h se expusieron larvas de tercer estadio de H. grandella a discos de follaje de Cedrela odorata impregnados con cada tratamiento. Estos consistieron en disoluciones de cada extracto (0.1, 0.32, 1.0, 3.20 y 10%); posteriormente cada larva se transfirió a un frasco que contenía dieta artificial, donde se le permitió completar su desarrollo. Las variables de respuesta fueron el consumo de alimento (área foliar comida en 24 h), la mortalidad y efectos sobre el desarrollo (tiempo de desarrollo de cada estadio larval y de la pupa, y el peso de la pupa). El extracto de ruda causó fagodisuasión a una concentración de apenas 0.32%, mientras que la fracción de "tacaco cimarrón" provocó toxicidad especialmente a las dos mayores concentraciones (3.20 y 10%).
References
Allan, G.G., R.I. Gara & R.M. Wilkins. 1973. The evaluation of some systemic insecticides for the control
of larvae in Cedrela odorata L., p. 40-48. In P. Grijpma (ed.). Studies on the shootborer Hypsipyla
grandella (Zeller). Lep. Pyralidae. Vol I. IICA Misc. Publ. No. 101. IICA, Turrialba, Costa Rica.
Beeker, V.O. 1976. Microlepidópteros asociados con Carapa, Cedrela y Swietenia en Costa Rica, p. 75-101. In J.L. Whitmore (ed.). Studies on the shootborer Hypsipyla grandella (Zeller). Lep. Pyralidae. Vol II. IICA Misc. Pub¡. No. 101
Castro, V., E. Ramírez, G. Mora, Y. Iwase, T. Nagao, H. Okabe, H. Matsunaga, M. Katano & M. Mori. Structures and antiproliferative activity of saponins from Sechium pittieri and S. talamancense. Chem. & Pharm. Bull. 45: 349-358. [ Links ]
Cibrián, D., J.T. Méndez, R. Campos, H.0. Yates III & J.E. Flores. 1995. Insectos forestales de México.
Universidad Autónoma de Chapingo-Comisión Forestal de América del Norte (COFAN). Publ. No. 6.
México D.F., México. 453 p.
Cox, N.D. 1980. Flea treatment composition for animals. Patent-US-4,193,986 (USA). 3 p. [ Links ]
Grijpma, P. (ed.). 1973. Studies on the shootborer Hypsipyla grandella (Zeller). Lep., Pyralidae. Vol. I.
IICA Misc. Publ. No. 101. 91 p.
Grijpma, P. & R. Ramalho. 1973. Toona spp., posibles alternativas para el problema del barrenador
Hypsipyla grandella de las Meliaceae en América Latina, p. 3-17. In P. Grijpma (ed.). Studies on the
shootborer Hypsipyla grandella (Zeller). Lep. Pyralidae. Vol I. IICA Misc. Publ. No. 101. IICA, Turrialba,
Costa Rica.
Hough-Goldstein, J.A. 1990. Antifeedant effects of common herbs on the colorado potato beetle
(Coleoptera: Chrysomelidae). Environmental Entomology 19: 234-238.
Isman, M.B. 1999. Neem and related natural products. p. 139-153. In FR. Hall & JJ Menn (eds.).
Biopesticides: Use and delivery. New Jersey, Humana Press.
Kong, Y.C., C. Lau, P.P.H. But, K.F. Cheng & R.C. Cambie. 1984. Quinoline alkaloids from Ruta
chalepensis. Fitoterapia 55: 67-71.
Mabry, T.M. & J.E. Gill. 1979. Sesquiterpenene lactones and other terpenoids. p. 501-537. In G.A.
Rosenthal & D.H. Janzen (eds.). Herbivores: Their interaction with secondary plant metabolites. New
York, Academic Press.
Mancebo, E, L. Hilje, G. Mora & R. Salazar. 2000a. Antifeedant activity of Quassia amara (Simaroubaceae) extracts on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae. Crop Protection 19: 301-305. [ Links ]
Mancebo, F., L. Hilje, G. Mora & R. Salazar. 2000b. Efecto de extractos vegetales sobre larvas de
Hypsipyla grandella. Manejo Integrado de Plagas (Costa Rica) 55: 12-23.
Mancebo, F., L. Hilje, G. Mora & R. Salazar. 2000c. Biological activity of two neem (Azadirachta indica
A. Juss., Meliaceae) products on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae Crop Protection.
(Submitted).
Mayhew, J.E. & A.C. Newton. 1998. The silvicultura of mahogany. CABI Publ. Wallingford, UK. 226 p. [ Links ]
Mesén, E 1999. Avances en el mejoramiento genético de meliáceas con referencia especial a América
Central. In Consulta Mesoarnericana sobre Conservación de Recursos Genéticos Forestales, con Enfasis
Especial en Meliáceas. REDCA. San Salvador, El Salvador. 19 p. (Manuscript).
Newton, A.C., P Baker, S. Ramnarine, J.F. Mesén & R.R.B. Leakey. 1993. The mahogany shoot borer:
Prospects for control. Forest Ecol. Manag. 57: 301-328.
Richter, K., R. Nothlich, A. Stacke & H. Penzlin. 1990. Moulting inhibiting effect of extracts from
Rutaceac in the cockroach Periplaneta americana. Zool. Jahr., Abt. Allgem. Zool. & Physiol. Tiere 94:
63-72.
Sasanelli, N. 1997. Perspectives for the use of nematicidal plants. Rev. Suisse Agric. 29: 157-158. [ Links ]
Schabel, H., L. Hilje, K.S.S. Nair & R.V. Varma. 1999. Economic entomology in tropical forest
plantations: An update. Jour. Trop. For. Sci. 11: 303-315.
Schoonhoven, L.M. 1982. Biological aspects of antifeedants. Entomol. exp. et applic. 31: 57-69. [ Links ]
Schoonhoven, L.M. 1982. Perception of azadirachtin by some tepidopterous larvae. p. 105-108. In H.
Schmutterer, K.R.S. Ascher & H. Rembold, (eds.). Natural pesticides from the neem tree (Azadirachta
indica A. Juss). GTZ. Eschborn, Germany.
Speight, M.R. 1997. Forest pests in the tropics: Current status and future threats. p. 207-227. In
A.D.Watt, N.E. Stork & M.D. Hunter (eds.). Forest and insects. London., Chapman & Hall.
Tomerlin, J.R. & T. Howell. 1988. DISTRAIN: A computer program for training people to estimate disease
severity on cereal leaves. Plant Dis. 72: 455 459.
Torres, J.C. 1950. Pharmacognostic study of rue leaf- Its principle component rutoside and essence.
Farmacognosia 10: 275-361.
Vasudevan, T.N. & M. Lukner. 1968. Alkaloids from Ruta angustifolia, Ruta chalepensis and Ruta
Montana. Pharmazie 23: 520-526.
Whitmore, J. L. (ed.). 1976a. Studies on the shootborer Hypsipyla Arandella (Zeller). Lep., Pyralidac. Vol.
Il. IICA Misc. Publ. No. 101. 139 p.
Whitmore, J. L. (ed.). 1976b. Studies on the shootborer Hypsipyla grandella (Zeller). Lep., Pyralidae.
Vol. III. IICA Misc. Publ. No. 101. 116 p.
Wilkins, R.M., G.G. Allan & R.I. Gara. 1976. Protection of Spanish cedar with controlled release
insecticidas. p. 63-70. In J.L. Whitmore (ed.). Studies on the shootborer Hypsipyla grandella (Zeller). Lep.
Pyralidae. Vol III. IICA Misc. Publ. No. 1. IICA, Turrialba, Costa Rica.
1 Fertilizantes Químicos Dominicanos (FERQUIDO). Santo Domingo, Dominican Republic.
2 Unidad de Fitoprotección, CATIE. Turrialba, Costa Rica.
3 Centro de Investigación en Productos Naturales (CIPRONA). Universidad de Costa Rica.
4 Proyecto de Semillas Forestales (PROSEFOR). CATIE. Turrialba, Costa Rica.
Address for correspondence: Unidad de Fitoprotección, CATIE. Turrialba, Costa Rica.
Fax (506) 556-0606, lhilje@catie.ac.cr












 uBio
uBio