Revista de Biología Tropical
versión On-line ISSN 0034-7744versión impresa ISSN 0034-7744
Rev. biol. trop vol.48 no.4 San José dic. 2000
Elena Solana-Arellano, Hector Echavarria-Heras and Olga Flores-Uzeta1
Received: 15-II-00 Corrected: 6-V-00 Accepted: 16-VI-00
Abstract
This paper presents a new method for the recovery of marked seagrass blades. The introduction of a plastic belt surrounding the marked shoot at a sediment level provided a relocation arrangement which was unloosed by drag forces or grazing. The relocation method was tested on Zostera marina L. It proved to have the advantage of increasing dramatically the number of marked shoots recovered up to 100% while reducing the cost of the procedure to a minimum. An allometric model indicated that the introduced relocation method has no impact on the development of the plant.
Key words
marking technique, vegetative shoots, seagrass production.
The assessment of seagrass production has stimulated the development of several leaf-marking techniques aimed to monitor temporal increases in plant biomass or plant elongation. One important step in these procedures relates to the recovery of the marked shoots. Most of the these seagrass production assesment techniques are modifications of the Zieman (1968) marking method. Modifications to this technique had been made as an attempt to reduce damage to the leaves and to improve the ratio of recovered to marked shoots. One of the most used shoot recovering technique, is the Kentula and McIntare (1986) in which a.5 mm stake of PVC rod bent to a ring at one end is inserted into de sediment next to a vegetative shoot and the base of the shoot is isolated within the ring. It has been observed that the effect of drag forces can liberate the bundle of marked leaves from the ring. This effect induces a high loss of marked shoots (Kentula and McIntire 1986). On the other hand, the possible alterations induced by this technique to growth, production or any other physiological process of the seagrass under study have not been evaluated.
The problem of the loss of marked shoots has been constantly addressed. For instance Ibarra-Obando (1985) reports a 33.3% of lost shoots in Zostera marina study in San Quintin Bay, México, while using the Ziemans (1968) technique. For the same area and using the Kentula & McIntire method Ibarra-Obando and Huerta-Tamayo (1987) reported a lost of 14.5% of the marked shoots and Melling-Lopez (1995) lost between 9% and 13.3% of marked shoots. Patriquin (1973) reports that while using the Zieman (1968) technique recovered only 60% of marked shoots. An alternative to this procedure was due to Zieman (1974) who improved the technique described by Odum (1959) and stapled Thalassia but he did not proved if this stapling technique had a deleterious effect on leaf production.
In any kind of marking technique there are several problems to be addressed. Here we will consider the following; relocation of marked shoots, cost of the device to mark them and possible impact of these devices on growth and production of the plant. The present propose an easy and inexpensive technique which allowed to relocate and recuperate 100% of previously marked shoots. Using an allometric model as a comparison device, was also demonstrated that the method has no impact on the biological development of the plant.
The Punta Banda estuary (31 40N-31
48N, 116
40W) is located on the West Coast of northern Baja California México; it comprises a total area of 21
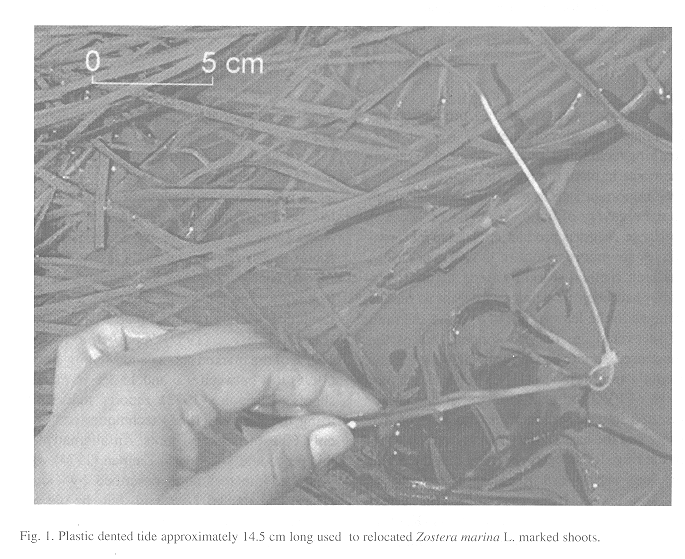
The study area is a transect of approximately 50 meters long at a 0.38m MLLW. In a first sampling experiment, using a systematic random sampling technique we marked 12 shoots biweekly form January to February 1998 utilizing the Kentula and McIntire technique (1986). Then we placed each shoot into a welding rod bent to a ring with a flag on the top of the rod. While using this procedure we had high difficulties to relocate the marked shoots and lost in average 20% of marked shoots. By virtue of this, we did some modifications to the original method. We marked again 12 shoots biweekly, from March to April marking the bundle of leaves of each shoots in the same way but we introduced a 14.5 cm plastic dentate tie painted on the tip with red nail polish (Fig. 1). The tie was placed around the marked shoot (as a belt) at a level of the sediment. The dentation of the tie permitted us to adjust the belt in such a way that the tie was securely attached to the marked shoot. We made sure that the belt did not press the shoot. This procedure was able to prevent the tie to be loosed by drag forces To relocate exactly the place were the shoot was marked we placed a colored flag next to each belted and marked shoot. Most of the time, the tip of the belt showed off by itself and when the Zostera marina L. was partially covered the flag led us to the marked and belted shoots so we relocated it easily.
Following an allometric law for leaf growth, Solana-Arellano et al.(1998) showed that for Zostera marina L. leaf dry weight could be expressed in terms of length and width through the relationship:
- w = Kl
In order to demonstrate that the plastic belt had no impact on shoot development we used the following procedure. Since Zostera m. has an annual pattern for production and considering the consistency of the Solana-Arellano et al. (1998) allometric model to estimate the involved parameters, we proceeded to fit equation (1) to data collected biweekly during March and April 1998 using the belt modification. Similarly we fitted the allometric model separately for data collected without the belt during the months of January and February. Finally we compared statistically the results to explore possible differences.
Using our belt technique we recuperated 100% of marked shoots with reduced cost. When fitting the allometric equations for both sets of data we found that the coefficient of determination was of 0.89 for both cases. Also no statistical differences between the obtained parameters with an F=0.78 at a significance level of 95% were found (see Table I). The standard error of the estimation was of 0.36 for the case where we did not use the belt technique and 0.38 for the belt technique. The residual Analysis in each case gave no indications of abnormality in the distribution of the residuals. Moreover, the mean number of leaves per shoot, the means for length, weight and width obtained with the modified relocation technique are in accordance with those reported in the literature for the study site.
It is important to point out that Solana-Arellano et al. (1998) found no statistical differences in the parameters obtained by fitting equation (1) monthly for a whole year period in a Zostera marina meadow. Therefore as a result of the allometric comparisons based on the statistical analysis we can conclude that the belt technique is appropriate for the efficient recovery of marked Zostera marina shoots and furthermore we have no indications that it produces alterations in growth and production patterns of Zostera marina.
We believe that the efficiency of our relocation technique was due to the fact that the marked shoot dentate plastic belt system was able to move as a whole. Hence drag forces were unable to separate the marked-shoot from the relocation device as it occurs in the conventional techniques were the shoot moves outside the relocation ring by drag forces even though the ring remains attached to the sediment. Moreover, our technique have the advantages of being easy to manipulate, easy to relocate and has a very small monetary cost. It also permits to reduce the uncertainty on growth and production estimations due to the avoidance of lost marked shoots.
This paper is part of the research on eelgrass production partially funded by CONACYT by grant 26665-B. We thank Jose Ma. Dominguez and Francisco Ponce for the technical assistance.
References
Ibarra-Obando S.E. 1985. Introducción al método de marcado para medir la produccón de pastos marinos. Universidad Autónoma de Baja California, México. 30p. [ Links ]
Ibarra-Obando S.E & R. Huerta-Tamayo 1987. Blade production of ZosteramarinaL. during the summer-autumn period on the Pacific Coast of México. Aquat. Bot. 28: 301-315. [ Links ]
Kentula, ME. & C.D. McInitire 1986. The autoecology and production dynamics of eelgrass (Zosteramarina L.) in Netarts Bay, Oregon. Estuaries 9: 188-199. [ Links ]
Meling-Lopez, A. E. 1995. Comparación y análisis de dos métodos que miden el crecimiento y la producción foliar de ZosteramarinaL. Tésis de Maestría CICESE, Ensenada Baja California, México 81 p. [ Links ]
Odum, E.O. 1959. Fundamentals of ecology. W.B. Sanders, Philadelphia. 546 p. [ Links ]
Patriquin, D. 1973. Estimation of growth rate, production and age of the marine angiosperm Thalassia testudinum Koing. Carib. J. Sci. 13: pp. 111-123. [ Links ]
Solana-Arellano E., D.J Borbon-Gonzalez & H. Echavarria-Heras. 1998. A general allometric model for blade production in Zostera narina L. Bull. South. Cal. Acad. Sci. 97: 39-49. [ Links ]
Zieman, J.C. 1968. A study of the growth and decomposition of the sea-grass, Thalassia testudinum. MSc thesis University of Miami, Miami Florida. 50 p. [ Links ]
Zieman J.C. 1974. Methods for the study of the growth and production of turtle grass, Thalassia testudinum Koing. Aquaculture. 4: 139-143. [ Links ]
Zieman, J.C. & R.G Wetzel. 1980. Productivity in seagrasses: Methods and rates. p. 87-118. In D.C. Phillips & C.P. McRoy (eds). Handbook of Seagrasses Biology: An Ecosystem perspective. Garland STPM, New York. [ Links ]
Table 1
Estimations of the parameters of equation (1) for both
techniques with standard error and level of significance.
| Marking technique | Parameter | Std. error | p-level |
| k=0.000005 | 0.3 | p<0.00 | |
| Without belt | 0.06 | p<0.00 | |
| 0.21 | p<0.01 | ||
| k=0.000004 | 0.3 | p<0.00 | |
| With belt | 0.06 | p<0.00 | |
| 0.18 | p<0.05 |
1Centro de Investigación Científica y de Educación Superior de Ensenada B. C., México.
Mailing address: P.O. Box 434844, San Diego, CA. 92143-4844, U.S.A
Fax: (617)45154 & 44880; email: esolana@cicese.mx












 uBio
uBio