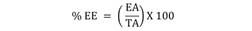Introduction
Parasitosis is a problem that affects a huge number of the human population; likewise, their transcendence is linked to secondary diseases, there are different types of parasites that affect humans, which are closely linked to poverty, socio-geographic and the lack of funding for their care (Theel & Pritt, 2016). Among the most common parasitosis are amebiasis caused mainly by Entamoeba histolytica and trichomoniasis caused by Trichomonas vaginalis. E. histolytica is a human pathogen that mainly colonizes the large intestine through fecal-oral contamination and sex ual transmission through anal-oral contact has also been described (Theel & Pritt, 2016). Its prevalence increases in geographical areas with inadequate environmental sanitation, as occurs in developing countries and it is estimated that 10 % of the world’s population is infected with approximately 100 000 deaths per year (Turkeltaub et al., 2015). There are 500 million new infections per year, of which 50 million suffer from invasive amebiasis, meaning that the manifestation is intestinal and extraintestinal (Kantor et al., 2018). T. vaginalis is one of the most prevalent non-viral sexually-transmitted pathogens in the world; in 2016, World Health Organization (WHO) estimated 156 million cases of trichomoniasis around the world (WHO, 2018). After colonization, this parasite causes vaginitis, urethritis, and prostatitis (Cudmore et al., 2004). Also, the pathogen has been associated with other serious consequences such as adverse pregnancy outcomes and preterm delivery, infertility, predisposition to cervical cancer, and pelvic inflammatory disease. Evidence has demonstrated the potential of T. vaginalis as a cofactor for increased HIV transmission (Wendel & Workowski, 2007).
For decades, the treatment for parasitic protozoal infections has been metronidazole among other nitroimidazoles, however, it is now well established that extensive use of this mono-therapeutic treatment regimen fosters drug resistance, leading to unsuccessful treatment and relapse of the disease (Dingsdag & Hunter, 2018). Resistance to metronidazole has been observed in T. vaginalis and has been suggested in E. histolytica due to the increased frequency of unsuccessful treatments, likewise, the use of this drug frequently presents side effects that force the patient to abandon the treatment, due to this, the need to identify new compounds as antiparasitic drugs, natural sources can be an alternative for the treatment of parasitic infections (Davoodi & Abbasi-Maleki, 2018; Elizondo-Luévano et al., 2021c).
Plants represent an important source and diversity of biomolecules with unique properties and many of these compounds are attractive candidates for the development of new antiparasitic agents in addition some plant extracts have shown interesting antiparasitic properties without the serious side effects of cytotoxic agents (Elizondo-Luevano et al., 2020a; Molina-Garza et al., 2014). Among the most relevant plants are Curcuma longa (Zingiberaceae), a tuberous herbaceous perennial plant member of the ginger family that grows in tropical climates (Prasad et al., 2014), whose main ingredient is the lipophilic polyphenolic substance curcumin (Cur), which is a valuable compound in the food and pharmaceutical industries due to its wide range of beneficial health effects with mainly antioxidant, antimicrobial, antiparasitic, anti-inflammatory, and antiangiogenic properties (Cárdenas Garza et al., 2021; Kocaadam & Şanlier, 2017; Kumar et al., 2018). Berberis vulgaris (Berberidaceae) is another plant that is currently relevant due to its various health-promoting properties (Raju et al., 2019). The isoquinoline alkaloid, i.e. berberine (Ber), is the main compound of B. vulgaris, whose pharmacological properties include anticonvulsant, antidepressant, antiarrhythmic, anti-inflammatory, antiparasitic, antiviral, antibacterial, antineoplastic and antidiabetic (Verma & Sharma, 2018). In general, the application of natural products can be problematic because they can be deteriorated by different factors such as light, pH, oxygen, and enzymes (Barrera-Ruiz et al., 2020). In addition to presenting low solubility in water as in the case of Cur and Ber (Fig. 1), which hinders their bioavailability, these characteristics limit their potential pharmaceutical uses (Mehra et al., 2016). Therefore, to overcome those effects nano-encapsulation into polymeric nanoparticles (NPs) has become an alternative (Pina-Barrera et al., 2019).
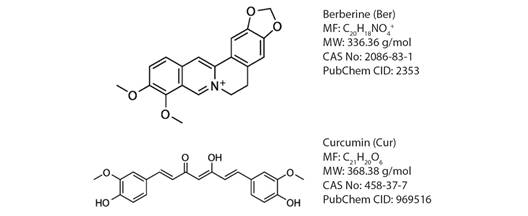
Fig. 1 Chemical structures, molecular formula (MF), molecular weight (MW), CAS number (CAS No.), and PubChem compound identifier (PubChem CID) of Ber: Berberine and Cur: Curcumin.
Among the nano-carriers used, the synthetic polymers Eudragit® (polymethacrylates by Evonik Industries AG, Germany) are widely used for the preparation of oral drug delivery systems designed for controlled-release and delivery of drugs to specific sites in the gastrointestinal tract distinguished by their versatility and ease of handling (Chong-Cerda et al., 2020). Particularly, the polymer used in this research was the pH-sensitive cationic polymer Eudragit® EPO (cationic terpolymer of N,N-dimethyl amino ethyl methacrylate with methylmethacrylate and butylmethacrylate), which has been used for the preparation of hydrophobic drug dispersions that have a dissolution rate-dependent bioavailability (Khachane et al., 2011).
In order to introduce a novel strategy for the administration of natural products such as extracts and metabolites. In this study, the activity of the extracts of C. longa (ExtCl), B. vulgaris (ExtBv), and the main components Ber and Cur in free form as well as encapsulated into NPs against trophozoites of E. histolytica and T. vaginalis was investigated in in vitro assays.
Materials and methods
2.1. Chemicals: Berberine (Ber), curcumin (Cur), absolute ethanol (EtOH), absolute methanol (MeOH), ethylenediaminetetraacetic acid (EDTA), and phosphate buffer solution were purchased from Sigma-Aldrich (Merck KGaA, Germany). Decomplemented Adult Bovine Serum (DABS) was donated by the Center for Biomedical Research of the Northeast (C.B.R.N.) of the Instituto Mexicano del Seguro Social (I.M.S.S.) Monterrey, México. Polymer Eudragit® EPO was donated by Helm de México S.A. All chemicals and solvents were of analytical grade.
2.2. Ethicals: The hemolysis test was performed using human erythrocytes, under the approval of the ethics committee of the U.A.N.L. Under the Official Mexican Technical Standard: NOM-253-SSA1-2012.
2.3. Plant materials and extraction: Plant materials, i.e., C. longa rhizome (SKU: 205400-54) and B. vulgaris roots (SKU: 209142-54) originated from India were purchased in powdered form from Starwest Botanicals (Sacramento, CA, United States). The extraction of C. longa was performed with EtOH and the extraction of B. vulgaris was with MeOH. The extractions were performed with 100 g of each plant material, which were deposited in a 1 000 mL Erlenmeyer flask, in which 400 mL of solvent were added. The flasks were hermetically sealed to avoid evaporation of the solvent, kept at room temperature for 24 hr and in constant agitation away from light. After the time required for extraction, the solvent was separated from the rest of the plant material by filtration with Whatman No.1 paper (Merck® KGaA, Germany), then the extracts were concentrated in a rotary evaporator system (Heidolph Instruments GmbH & CO. KG, Germany) at a temperature of 30 °C and 100 rpm. The extraction yield (% Y) was calculated with the following formula:
2.4. Phytochemical tests: The following phytochemical tests were performed to identify the functional groups of each extract: Unsaturations, carbonyl group, sesquiterpene lactones, lactones, tannins, triterpenes-sterols, phenols, saponins, Shinoda (flavonoids), carbohydrates, and Dragendorff (alkaloids).
2.5. Strains: The E. histolytica HM1:IMSS and T. vaginalis GT15 strains were provided by C.B.R.N - I.M.S.S. and were grown and maintained in vitro in TYI-S-33 culture medium supplemented with 10 % DABS at 37 °C.
2.6. Preparation of polymer nanoparticles: The NPs was prepared by the nanoprecipitation technique (Fessi et al., 1989). For that purpose, 50 mg of Eudragit ® EPO polymer plus 5 mg of the extracts or main components were dissolved in 15 mL of EtOH, which was constantly injected in an aqueous phase (10 mL of bidistilled water), and then the solvent was removed by a rotary evaporator system (100 rpm / 30 ºC) to obtain an aqueous suspension of NPs. The particle size and polydispersity index (PDI) of the suspension were determined by photon correlation spectroscopy. An aliquot of the suspension was mixed with bi-distilled water and read in a Zetasizer Nano ZS (Malvern Instruments, United Kingdom) at 25 °C to determine its average particle size and PDI. The size of the nanoparticles was confirmed by scanning electron microscopy (SEM), where the conforming nanostructure and the degree of aggregation of the NPs were determined. Using the methodology previously described by Chong-Cerda et al., the percentage of extract or active trapped inside and outside the NPs was estimated (Chong-Cerda et al., 2020), for which the formulations were ultracentrifuged (Beckman-Coulter. United States. Allegra 64R Benchtop Centrifuge) at 20 000 rpm (4 °C / 3 hr). The encapsulation efficiency (% EE) was determined by the following formula:
Where EA corresponds to the entrapped amount into the NPs, and TA corresponds to the total amount of the extract or active ingredient.
2.7. Biological assays: The in vitro antiprotozoal activity of the extracts and the main components in free form and encapsulated into NPs was based on the methodology described by Elizondo et. al. (Elizondo-Luévano et al., 2018; Elizondo-Luévano et al., 2021c), in which they were tested against log-phase cultures of E. histolytica (20 000 trophozoites/mL), and T. vaginalis (100 000 trophozoites/mL) in TYI-S-33 culture medium with 10 % DABS at 37 °C. The concentrations of all treatments were evaluated at different concentrations to determine the IC50 of each treatment, respectively. The positive control consisted of metronidazole (reference drug) and only culture medium as negative control. IC50 was determined by the Probit test with a 95 % confidence interval.
2.8. Hemolysis test: Hemolysis was evaluated based on the hemolytic activity in erythrocytes (Elizondo-Luevano et al., 2020a). For this, 100 µL of the erythrocyte solution was added to a 96-well microplate after 30 min incubation, treated with 100 µL of the treatments (extracts and main components in free and nano-encapsulated form) diluted in PBS at a concentration range of 10 to 100 µg/mL. Distilled water was used as a positive control for hemolysis (C+) and untreated culture medium as a negative control (C-). The microplate was read at 540 nm in a microplate spectrophotometer (Thermo Fisher Scientific Inc., United States. Multiskan SkyHigh Spectrophotometer). The absorbance of the untreated erythrocyte suspension was used as blank. Percentage of hemolysis was calculated using the next formula:
2.9. Statistical analysis: Results expressed as the mean and standard error (SEM). Significant differences between groups were determined by the one-way ANOVA test, the fifty percent inhibitory concentration (IC50) was determined by the Probit test. The post-hoc Tukey´s test was performed to determine any statistical differences between treated and untreated controls. Student’s t-test was used to determine the differences between encapsulated and non-encapsulated treatments. Differences were significant at P < 0.05 with a 95 % confidence interval using the IBM-SPSS (IBM Corp, 2013). All experiments were performed in triplicate.
3.1. Phytochemical analysis: Phytochemical analysis indicates the presence of tannins, phenols, and flavonoids in the extract of C. longa and alkaloids in B. vulgaris. The extract yields of C. longa and B. vulgaris were 8.86 and 2.33 %, respectively.
3.2. In vitro antiprotozoal activity: All the treatments tested were biologically active analyzed by the Probit test (Table 1). The nanoencapsulated treatments were significantly (P < 0.001) more effective than the non-encapsulated treatments, it was observed that ExtBv was more effective than ExtCl either in free form or incorporated into NPs. However, by Tukey’s test, it was determined that BerNPs were the most effective treatment compared to the reference drug metronidazole showing IC50 values of 0.24 and 0.71 µg/mL respectively against E. histolytica and T. vaginalis.
Table 1 Antiprotozoal activity of C. longa and B. vulgaris extracts as well as curcumin and berberine in non-encapsulated form and encapsulated into NPs compared to the effects of the reference drug
| IC50 (µg/mL) | |||
| Treatment | E. histolytica | T. vaginalis | |
| Metronidazole (Reference drug) | 0.13 ± 0.03a | 0.07 ± 0.01a | |
| ExtCl | 361.43 ± 8.07h | 496.36 ± 10.52g | |
| ExtClNPs | 26.03 ± 2.31e | 102.29 ± 13.06e | |
| P t-student (F=12.71) | 0.009 | 0.025 | |
| ExtBv | 134.19 ± 11.20g | 192.08 ± 8.04f | |
| ExtBvNPs | 19.02 ± 1.16d | 46.30 ± 5.51d | |
| P t-student (F=12.71) | 0.042 | 0.034 | |
| Cur | 65.60 ± 5.17f | 51.11 ± 3.55d | |
| CurNPs | 9.48 ± 1.23c | 4.25 ± 1.04c | |
| P t-student (F=12.71) | 0.037 | 0.002 | |
| Ber | 3.28 ± 0.51b | 2.60 ± 1.12c | |
| BerNPs | 0.24 ± 0.10a | 0.71 ± 0.22b | |
| P t-student (F=12.71) | 0.004 | 0.003 | |
| P Anova (F = 27.91) | < 0.001 | < 0.001 | |
Values are shown as mean ± SD. Different letters within the same column are significantly different analyzed via the post-hoc Tukey’s test (P < 0.05).
Table 2 Size, polydispersity index (PDI) and percentage encapsulation (% EE) of the polymeric nanoparticles (NPs) of C. longa and B. vulgaris extracts, and their main components curcumin and berberine
| Formulation | Size (nm) | PDI | % EE |
| BlankNPs | 45.9a | 0.114b | - |
| ExtClNPs | 53.6b | 0.177d | 34.81b |
| ExtBvNPs | 54.4b | 0.190e | 25.94a |
| CurNPs | 66.5c | 0.133c | 83.59d |
| BerNPs | 73.4d | 0.086a | 76.48c |
| P Anova (F = 5.28) | < 0.05 | < 0.01 | < 0.01 |
Different letters within the same column are significantly different analyzed via the post-hoc Tukey’s test (P < 0.05).

Fig. 2 Morphology and size of the NPs by SEM at 1.0 kV. Their spherical structures are shown. A: NPsBlank, B.1: NPsExtCl, B.2: NPsExtBv, C.1: NPsCur, and C.2: NPsBer.
3.4. Hemolytic activity test: The hemolytic activity assay in human erythrocytes showed that none of the treatments (ExtCl, ExtBv, Cur, and Ber in free or nano-encapsulated form) were significantly (P < 0.001) less hemolytic than the positive control (C+) which showed 100 % hemolysis. The encapsulated treatments were less hemolytic than the non-encapsulated ones, and none of the treatments (non-encapsulated or NPs) showed hemolysis greater than 10 % even at 100 mg/mL (Table 3).
Table 3 Evaluation of the hemolysis test of extracts of C. longa, B. vulgaris, and their main components curcumin and berberine in unencapsulated and encapsulated form in polymeric nanoparticles (NPs)
| Positive control (C+) 100 ± 3.8 | Negative control (C-) 1.19 ± 0.3 | |||||||
| Hemolysis percentage (%) | ||||||||
| µg/mL | ExtCl | ExtCl NPs | ExtBv | ExtBv NPs | Cur | Cur NPs | Ber | Ber NPs |
| 10 | 0.49 ± 0.1* | 1.14 ± 0.4* | 1.23 ± 0.1 | 0.10 ± 0.0* | 0.13 ± 0.1* | 0.05 ± 0.0* | 1.43 ± 0.6 | 1.09 ± 0.2* |
| 20 | 0.72 ± 0.1* | 1.19 ± 0.2 | 1.25 ± 0.2 | 0.12 ± 0.1* | 1.01 ± 0.4* | 0.05 ± 0.0* | 1.88 ± 0.3 | 1.12 ± 0.3* |
| 40 | 2.19 ± 0.3 | 1.52 ± 0.4 | 2.81 ± 0.7 | 1.16 ± 0.1* | 1.17 ± 0.2 | 0.40 ± 0.1* | 2.29 ± 0.7 | 1.13 ± 0.1* |
| 60 | 2.43 ± 0.4 | 1.98 ± 0.1 | 2.95 ± 0.3 | 1.38 ± 0.1 | 1.26 ± 0.3 | 0.54 ± 0.1* | 4.13 ± 0.5 | 2.10 ± 0.2 |
| 80 | 3.64 ± 0.3 | 1.99 ± 0.3 | 5.65 ± 0.8 | 2.02 ± 0.2 | 1.33 ± 0.2 | 0.70 ± 0.1* | 7.03 ± 0.7 | 2.50 ± 0.3 |
| 100 | 3.79 ± 0.7 | 1.99 ± 0.6 | 6.13 ± 0.9 | 2.04 ± 0.2 | 1.51 ± 0.3 | 0.76 ± 0.1* | 7.12 ± 0.7 | 2.77 ± 0.5 |
| P t-student (F=2.57) | 0.011 | 0.003 | 0.019 | 0.016 | ||||
Results are given in percentage (%) of hemolysis. The values are shown as the mean ± SD. *Significant differences in comparison with the negative control (C-) (P < 0.05).
Discussion
Currently, some reports are indicating the antiparasitic activity of plant extracts like C. longa, Elettaria cardamomum, and Kalanchoe daigremontiana as well as their components such as alkaloids, essential oils, and flavonoids among other phytochemicals (Cárdenas Garza et al., 2021; D’Andrea, 2015; Elizondo-Luévano et al., 2021a; Rosmalena et al., 2019). The activity of these compounds is well documented against a wide range of diseases such as parasitosis (Elizondo-Luévano et al., 2020b). These products have also been shown to possess fewer side effects in humans. As an alternative, several medicinal plant extracts had been investigated to develop a new drug for human parasites (Castañeda et al., 2021; Pérez et al., 2017). E. histolytica and T. vaginalis have been commonly treated with metronidazole, despite being an effective molecule, increased treatment failure has been observed in these pathogens due to increased resistance, which is a cause for concern (Jarrad et al., 2016). Another option for the treatment of metronidazole-resistant strains is a longer treatment or with higher doses, although this drug in most cases is well-tolerated, it can cause severe side effects, such as mutagenesis and carcinogenesis (Kuriyama et al., 2011). Hence the importance of finding alternatives for the treatment of amoebiasis and trichomoniasis. For this, two research strategies were proposed. The first was the search for new active principles and the second was the search for new ways of administration and delivery systems such as NPs.
Previous studies on the chemical composition of C. longa and B. vulgaris have indicated that the most important active constituents are Cur and Ber respectively, which have documented antioxidant, anticancer, antibacterial, antiparasitic, and other effects (Balakrishna & Kumar, 2015; Imenshahidi & Hosseinzadeh, 2016; Karłowicz-Bodalska et al., 2017; Vaughn et al., 2016). Cur, an active compound present in the rhizome of the C. longa, has been found to have in vitro antiprotozoal activity against Leishmania spp. malaria, G. lamblia, T. vaginalis, and Schistosoma mansoni (Elizondo-Luévano et al., 2020; Magalhães et al., 2009). Ber, an active compound present in B. vulgaris, it is considered to have antiparasitic potential against E. histolytica, G. lamblia, T. vaginalis, Leishmania spp., and anthelmintic against Strongyloides venezuelensis, Schistosoma mansoni, and Echinococcus granulosus (Elizondo-Luévano et al., 2021a; Elizondo-Luévano et al., 2021b; Mahmoudvand et al., 2014; Wright et al., 2000). Although Cur and Ber are the natural products with good antiprotozoal activity, these are the molecules with very low bioavailability, which limits their use; therefore, NPs seem to be an effective route of administration. For this purpose, firstly, the extracts of the rhizome of C. longa and the root of B. vulgaris were obtained, the yields obtained were 8.86 and 2.33 %, respectively. We observed that our results coincide with what was expected since in the case of C. longa considering the phenols and flavonoids, and for B. vulgaris, the alkaloids were the compounds that stood out for their presence.
By Probit statistical analysis, the IC50 of each treatment evaluated against E. histolytica and T. vaginalis in comparison to the reference drug was calculated (Table 2). For the C. longa treatments, the IC50 of the crude extract was 361.43 (E. histolytica) and 496.36 (T. vaginalis) µg/mL, and for its incorporation into NPs, decreased to 26.03 (E. histolytica) and 102.29 (T. vaginalis) µg/mL. The IC50 determined evaluating the extract of B. vulgaris, were 134.19 (E. histolytica) and 192.08 (T. vaginalis) µg/mL, once encapsulated the NPs presented IC50 of 19.02 (E. histolytica) and 46.30 (T. vaginalis) µg/mL. It was possible to observe the higher activity of ExtBv compared to ExtCl, however, when the extracts were incorporated into NPs, the IC50 decreased significantly (P < 0.001) in all cases. Therefore, it was found that both C. longa and B. vulgaris extracts presented more effective antiprotozoal activity in nano-encapsulated form.
Nanoparticles have been considered powerful tools for the treatment of infections due to the possibility of being endocytosed by cells, which could benefit the release of the active in the case of intracellular infections and due to the possibility of controlling and prolonging the release of the encapsulated molecules, nanoparticles could be very useful in the prevention or treatment of infections (Sasidharan & Saudagar, 2020). Such is the case of CurNPs and BerNPs, which were shown to be more effective than their non-encapsulated counterparts (Table 2) with IC50 of 9.48 (E. histolytica) and 4.25 (T. vaginalis) µg/mL for CurNPs, and 0.24 (E. histolytica) and 0.71 (T. vaginalis) µg/mL when BerNPs were evaluated, being far superior compared to Cur and Ber in non-encapsulated form. The most effective treatment was BerNPs behaving significantly like metronidazole. It was also determined that Cur was more effective than ExtClNPs against T. vaginalis, and Ber was more effective than ExtBvNPs when evaluated against both parasites. Ber is present in a wide variety of medicinal plants such as A. mexicana, B. aristata, and B. vulgaris and possesses a variety of pharmacological properties (Raju et al., 2019). There is also evidence that Ber is active against filarial, protozoa, and helminths (Elizondo-Luevano et al., 2020b; Elizondo-Luévano et al., 2018; Mahmoudvand et al., 2014; Rana & Misra-Bhattacharya, 2013).
There are no previous reports in the literature concerning the use of C. longa and B. vulgaris NPs or Cur and Ber NPs for the treatment of amoebiasis and trichomoniasis. However, there are studies where nanoparticles have been used as drug carriers to treat parasitosis in in vivo models, where efficient uptake, sustained release and enhanced therapeutic effects were observed. This is due to the size of the NPs, which results in a high concentration with its payload at the site of the medication (Sun et al., 2019). The activity of NPs opens the possibility to the combination of drugs that promote synergistic action or minimize the possibility of resistance of certain strains to drugs (Panic et al., 2014). There are other kinds of nanoparticles, such as silver nanoparticles with the aqueous extract of C. longa (Shabanzadeh et al., 2013), and nanometric complexes using ß-cyclodextrin incorporating the extract of B. vulgaris however, there are few studies on the incorporation of natural extracts into NPs (Hadaruga et al., 2010). In this work, we were able to produce NPs incorporating the crude extracts of C. longa and B. vulgaris independently, as well as Cur and Ber, using the cationic copolymer Eudragit® EPO of pharmaceutical-grade approved, it provides greater purity, stability, and reproducibility to the formulations, compared to natural polymers (Khoee & Yaghoobian, 2009). The characterization included the determination of the mean particle size, PDI, particle morphology, and the entrapment efficiency of the actives.
A significant difference (P < 0.05) was determined between the NPs with the extracts, the NPs with the actives, and the blank (Table 3). BlankNPs had a size and PDI of 45.9 nm and 0.114, respectively. The NPs containing C. longa extract had a size of 53.6 nm with a PDI of 0.177, those containing B. vulgaris extract presented size of 54.4 nm and a PDI of 0.190, while the NPs with Cur and Ber presented sizes of 66.5 and 73.4 nm as well as PDIs of 0.133 and 0.086, respectively. The sizes increase when the pure actives were nano-encapsulated, however, the PDIs decrease in comparison with the nano-encapsulated extracts. This indicates as the sizes of the NPs increased the homogeneity of the nano-formulations increased (Lancheros et al., 2014). These results were reproducible and meet the objective of having a particle size less than 200 nm and PDI less than 0.2. We sought to obtain a size smaller than 200 nm, since the smaller the particle size, the greater the surface interaction area, and this size allows sterilization by filtration (Banerjee et al., 2016). SEM confirmed the hemispherical morphology and smooth surface (Fig. 2). In the nanoparticulate systems, 34.81 (ExtClNPs), 25.94 (ExtBvNPs), 83.59 (CurNPs) and 76.48 (BerNPs) % EE were obtained (Table 3). A significant increase (P < 0.01) is noted in the encapsulation of Cur and Ber compared to crude extracts. Previous studies have reported entrapment efficiencies ranging from 74 to 98.5 % encapsulating pure actives such as curcumin, quercetin, piperine, and silybin by nanoprecipitation method (Abolhassani et al., 2020).
The investigation of the action of natural or synthetic products on erythrocytes is important to determine any hemolytic activity because it is an indicator of general cytotoxicity and bioactivity, this type of in vitro test is commonly employed for cyto-toxicological evaluations (Majidzadeh et al., 2020). In the present study, we tested concentrations between 10-100 µg/mL and none of the treatments (nano-encapsulated and not) were significantly (P < 0.05) hemolytic compared to the positive control which had 100 % of hemolytic activity (Table 3). In the hemolysis bioassay, we observed that the low hemolytic activity depends on the concentration of each treatment (hemolysis was concentration-dependent), so the dose is directly proportional to the hemolytic damage. However, when we evaluated the treatments based on nanoparticles, it was possible to observe a significant reduction in the nanoencapsulated treatments, this may be because the Eudragit® polymers are innocuous, and the low hemolytic activity is due to the release of the active ingredients or extracts to the medium where they are, or by mechanical stress during centrifugation (Ofridam et al., 2021). As can be seen in graph 3, Ber turned out to be the most active treatment presenting average hemolysis of 7.21 % (not encapsulated) and 2.77 % (NPs), when evaluated at 100 ppm, however, these results are below the data presented in Table 2 where we determined the IC50 values, where for the case of BerNPs it was 0.24 (E. histolytica) and 0.71 (T. vaginalis) µg/mL, respectively. Consequently, the use of NPs opens the possibility of new and better ways of administering active ingredients with biological activity (Meerasa et al., 2012; Raju et al., 2019). Since the NPs did not prove to be haemolytic, their evaluation can be considered for evaluation purposes in an in vivo model.
In this work, extracts of the rhizome of C. longa and roots of B. vulgaris, as well as their main components, i.e., curcumin, and berberine were successfully nano-encapsulated by the nanoprecipitation technique. NPs showed increased antiprotozoal activity against T. vaginalis and E. histolytica compared to non-encapsulated treatments giving a guideline to their potential use as adjuvants in the release of natural products for the treatment of parasitosis. None of the NPs-based treatments showed in vitro cytotoxicity on human erythrocytes. However, the mechanisms of action and the in vivo efficacy of Cur and Ber-loaded NPs need to be evaluated in an animal-infected model.
Ethical statement: the authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.












 uBio
uBio