Introduction
The marine biodiversity of the Pacific coast of Costa Rica is well known in some areas (i.e., Bahía Culebra, Golfo de Nicoya, Isla del Caño and Golfo Dulce). Other marine areas remain poorly studied, with little information available or far from complete, like the north Pacific of Costa Rica (Bahía Salinas and Golfo de Santa Elena) (Cortés, 2016; Cortés, 2017; Wehrtmann & Cortés, 2009). The same occurs regarding coastal ecosystems since some, such as coral reefs, have been relatively well studied (Alvarado, Beita-Jiménez, Mena, Fernández-García & Guzmán-Mora, 2015; Alvarado, Beita-Jiménez, Mena, Fernández-García, Guzmán-Mora, & Cortés, 2016; Alvarado Beita-Jiménez, Mena, Fernández-García, Cortés, Sánchez-Noguera, Jiménez, & Guzmán-Mora, 2018; Cortés, 1996/1997); while in others the research and publications on marine biodiversity are scarce (such as sandy beaches and rocky coast) (Dexter, 1974; Madrigal-Castro, Cabrera-Peña, Monge-Esquivel & Pérez-Acuña, 1984; Ortega, 1987; Sibaja-Cordero & Cortés, 2008; Sibaja-Cordero, Camacho-García & Vargas-Castillo, 2014).
The Área de Conservación Guanacaste (ACG) is one of eleven Conservation Areas within Costa Rica and it was declared a UNESCO World Heritage Site in 1999 (SINAC, 2012; UNESCO, 2020). The marine sector of ACG, recognized as a conservation priority site for marine and coastal biodiversity within Marine Ecological Unit Papagayo in the north Pacific of Costa Rica (Alvarado, Herrera, Corrales, Asch, & Paaby, 2011; SINAC-MINAET, 2008), has a high species richness and high diversity coastal and marine ecosystems, such as mangrove forest, beaches, bays and coves, rocky intertidal zones, mud flats, rocky subtidal sites, coral reefs, rhodolith beds, deep areas, shoals, and several isolated islands (Cortés, 2017; Cortés & Joyce, 2020). During the trade winds season (December to April-May; Rodríguez, Alfaro & Cortés, 2021) it is possible to find marine ecosystem dominated by the brown macroalgae Sargassum liebmannii J.A. Agardh 1847, that grow significantly as a consequence of the upwelling that brings deep cold, nutrient-rich and CO2-rich waters to the surface (Cortés, Samper-Villarreal & Bernecker 2014).
In ACG, including Bahía Salinas (BSa) and Golfo de Santa Elena (GSE), there are some publications on invertebrates and fish communities in coral reef, rocky reef and sandy areas (Alvarado et al., 2018; Cordero-Umaña & Santidrián-Tomillo, 2020). Thus, the aim of the present study is to increase the knowledge of marine biodiversity in this conservation area, specifically on BSa and GSE, and on poorly studied ecosystems like rocky reefs and Sargassum beds. The study provides a checklist of invertebrates and conspicuous fishes species that occur on shallow (6-14 m depth) rocky reefs and Sargassum beds in ACG. In addition, patterns of geographic distribution, number of species, species density and abundance are discussed.
Material and methods
Study site: The study was conducted during January and February 2013 in BSa and GSE, within ACG in the North Pacific of Costa Rica (Fig. 1, Table 1).
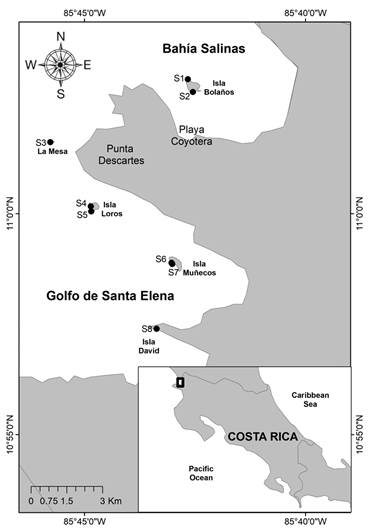
Fig. 1 Sampling sites in the North Pacific of Costa Rica. Rocky reefs: S1, S2, S3, S4, S5, S7 and S8; Sargassum beds: S6.
Table 1 Localities and survey sites in the North Pacific of Costa Rica. UVC: Underwater Visual Census; n: size of the sample.
| Locality | Survey site | ID | Geographic coordinates | Depth (m) | UVC method | n |
| Bahía Salinas | Isla Bolaños 1 | S1 | 11°03’02.65’’N 85°42’39.62’’W | 9 | Band transect | 3 |
| Isla Bolaños 2 | S2 | 11°02’45.19’’N 85°42’32.78’’W | 9 | Timed swim | 1 | |
| Golfo de Santa Elena | La Mesa | S3 | 11°01’37.13’’N 85°45’46.44’W | 9 | Band transect | 6 |
| Isla Loro 1 | S4 | 11°00’10.14’’N 85°44’51.43’W | 9 | Band transect | 6 | |
| Isla Loro 2 | S5 | 11°00’03.14’’N 85°44’50.86’’W | 13.5 | Timed swim | 1 | |
| Isla Muñecos 1 | S6 | 10°58’53.37’’N 85°43’01.78’’W | 9 | Band transect | 6 | |
| Isla Muñecos 2 | S7 | 10°58’51.67’’N 85°43’00.82’’W | 11 | Timed swim | 1 | |
| Isla David | S8 | 10°57’23.76’’N 85°43’21.98’’W | 6 | Timed swim | 1 |
ID: Code of the survey sites in the map.
Field methodology: In BSa we visited two sites, while in the GSE six. The marine ecosystems monitored were shallow (6 to 13.5 m depth) rocky reefs (boulders < 30 cm, small rocks < 30 cm - 1 m, big rocks 1 m - 3m, and rocks > 3m) and Sargassum beds (rocky substrate covered with 70% to 100% Sargassum) (Fig. 1, Table 1). Underwater Visual Census (UVC) methods using SCUBA were conducted at the eight sites during two sampling times (January and February 2013). In January, four sites were visited (S2, S5, S7 and S8). At each site the timed swim method, 50 to 60-minute, were done to register the invertebrates and conspicuous fish species. While in February, the other four sites (S1, S3, S4 and S6) were visited. Census followed standard band transect methodology described in previous studies (Alvarado et al., 2015; Alvarado et al., 2016). In each marine ecosystem, 10 m long transects, parallels to shore line and separated 10 m between them, were conducted by a single diver swimming to register the composition and abundance of invertebrates and conspicuous fishes. Three to six transects were performed at each site. Two runs were made along each transect. The first run was done while the line was being unrolled, recording all conspicuous fishes (> 5 cm total length) in the water column. We focused on conspicuous species rather than cryptobenthic species, or small fishes (< 5 cm in total length) that are behaviorally cryptic and are difficult to quantify by visual surveys due to their close association with the substratum (Allen, Bouvier & Jensen, 1992). In the second run, the diver recorded all invertebrates (>2.5 cm length) over the benthos and between crevices. For both UVC techniques, individuals under rocks were not registered to avoid altering the substrate. The categories were mollusks (gastropods and bivalves) and echinoderms (sea stars, sea urchins, sea cucumbers and brittle stars). Cryptic species (brittle stars) registered in transect bands method were not quantify to avoid an underestimation. For fishes, each band transect covered an area of 50 m2 (10 x 5 m) and for invertebrates an area of 20 m2 (10 x 2 m). Mean density (# ind m-2), relative abundance (# ind of one species/total # individuals), and frequency of occurrence (# transects observed/total # transects) were calculated for each species.
Fish species nomenclature and taxonomic designations followed Fricke, Eschemer & Van der Laan (2020) and Van der Laan, Fricke & Eschemeyer (2020), and for invertebrates the World Register of Marine Species (WoRMS Editorial Board, 2020). Fish species distribution were based on Froese & Pauly (2019) and Fricke et al. (2020), and categorized as follows: CT, Circumtropical; EP, Eastern Pacific; IP, Indo-Pacific; ETP, Eastern Tropical Pacific; WA, Western Atlantic. The IUCN Red List (IUCN 2020) was used to categorize the conservation status of each species recorded in this study as follows (in order of threat level): NE, not evaluated; DD, data deficient; LC, least concern. The categories mentioned correspond only to the species observed in this study.
Results
A total of 25 invertebrate species in 25 genera and 18 families were observed in BSa and GSE (Table 2) with the combined use of timed swim (n = 3) and band transects methods (n = 21). The most speciose invertebrate families were the Muricidae (3 spp.), Ophidiasteridae (2 spp.), Diadematidae (2 spp.), Toxoponeustidae (2 spp.), Cucumaridae (2 spp.) and Ophiocomidae (2 spp.). Hexaplex (2 spp.) was the most species rich genera, the rest of genera were represented by one specie. The highest number of species was observed in the rocky reef (23 spp.) while in Sargassum beds eight spp. (Table 2).
Table 2 Taxonomic list of invertebrates recorded in rocky reefs (RRs) and Sargassum beds (SBs) in the North Pacific of Costa Rica during January and February 2013, with the IUCN Red List classification. NE: Not Evaluated.
| Taxon | Marine Ecosystem | IUCN Red List | ||
| RRs | SBs | |||
| Phylum Mollusca | ||||
| Class Gastropoda | ||||
| Family Conidae | ||||
| Conus spp. Linnaeus, 1758 | S1, S5 | NE | ||
| Family Fasciolariidae | ||||
| Opeatostoma pseudodon (Burrow, 1815) | S3, S4, S8 | NE | ||
| Family Muricidae | ||||
| Babelomurex hindsi (Carpenter, 1857) | S2 | NE | ||
| Hexaplex erythrostomus (Swainson, 1831) | S3, S4 | NE | ||
| Hexaplex princeps (Broderip, 1833) | S1, S2, S4, S5 | S6 | NE | |
| Family Turbinellidae | ||||
| Vasum caestus (Broderip, 1833) | S6 | NE | ||
| Family Plakobranchidae | ||||
| Elysia diomedea (Bergh, 1894) | S4, S7 | S6 | NE | |
| Class Bivalvia | ||||
| Family Margaritidae | ||||
| Pinctada mazatlanica (Hanley, 1856) | S1, S2, S5 | NE | ||
| Family Spondylidae | ||||
| Spondylus limbatus G. B. Sowerbey II, 1847 | S1, S5 | NE | ||
| Phylum Echinodermata | ||||
| Class Asteroidea | ||||
| Family Mithrodiidae | ||||
| Mithrodia bradleyi Verrill, 1867 | S5, S7 | NE | ||
| Family Ophidiasteridae | ||||
| Pharia pyramidata (Gray, 1840) | S5 | NE | ||
| Phataria unifascialis (Gray, 1840) | S1, S3, S4, S5, S7 | NE | ||
| Family Oreasteridae | ||||
| Nidorellia armata (Gray, 1840) | S3 | NE | ||
| Class Echinoidea | ||||
| Family Arbaciidae | ||||
| Arbacia stellata (Blainville, 1825; ?Gmelin, 1791) | S1, S2, S3, S8 | NE | ||
| Family Diadematidae | ||||
| Astropyga pulvinata (Lamarck, 1816) | S7 | S6 | NE | |
| Diadema mexicanum A. Agassiz, 1863 | S1, S2, S3, S4, S5, S7, S8 | S6 | NE | |
| Family Cidaridae | ||||
| Eucidaris thouarsii (L. Agassiz & Desor, 1846) | S1, S2, S3, S4, S5, S7, S8 | NE | ||
| Family Toxopneustidae | ||||
| Toxopneustes roseus (A. Agassiz, 1863) | S1, S2, S3, S7, S8 | S6 | NE | |
| Tripneustes depressus A. Agassiz, 1863 | S2, S7 | S6 | NE | |
| Class Holothuroidea | ||||
| Family Cucumaridae | ||||
| Cucumaria flamma Solis-Marin & Laguarda-Figueras, 1999 | S2 | NE | ||
| Pseudocnus spp. Panning, 1949 | S2, S7 | NE | ||
| Family Holothuriidae | ||||
| Holothuria (Stauropora) fuscocinerea Jaeger, 1833 | S2 | NE | ||
| Class Ophiurodea | ||||
| Family Ophiocomidae | ||||
| Ophiocoma aethiops Lütken, 1859 | S4, S5 | NE | ||
| Ophiocomella alexandri (Lyman, 1860) | S6 | NE | ||
| Family Ophionereididae | ||||
| Ophionereis annulata (Le Conte, 1851) | S2 | NE | ||
Based on classification of the IUCN Red List, the conservation status of invertebrates species observed in BSa and GSE is unknow because those species have not been assessed by the IUCN (Table 2).
In the case of fishes, a total of 34 species in 30 genera and 15 families were observed (Table 3) with the combined use of timed swim (n = 3) and band transects methods (n = 21). The most speciose families were Serranidae (5 spp.), Pomacentridae (5 spp.), Labridae (5 spp.) and Tetraodontidae (4 spp.). Haemulon (3 spp.) and Halichoeres (3 spp.) were the most species rich genera. Two genera were represented by two species, and the rest by one species. The highest number of fish species was observed in the rocky reef (33 spp.), while in Sargassum beds only 10 species (Table 3).
Table 3 Taxonomic list of conspicuous fishes recorded in rocky reefs (RRs) and Sargassum beds (SBs) in the North Pacific of Costa Rica during January and February 2013, showing geographic distribution and IUCN Red List category. CT: Circumtropical; EP: Eastern Pacific; EP + IP: Eastern Pacific and Indo-Pacific; EP + WA: Eastern Pacific and Western Atlantic; ETP: Eastern Tropical Pacific. LC: Least Concern.
| Taxon | Marine Ecosystem | Distribution | IUCN Red List | |||
| RRs | SBs | |||||
| Order Myliobatiformes | ||||||
| Family Urotrygonidae | ||||||
| Urobatis halleri (Cooper, 1863) | S2, S5 | S6 | EP | LC | ||
| Order Anguilliformes | ||||||
| Family Muraenidae | ||||||
| Muraena lentiginosa Jenyms, 1842 | S4, S5, S7, S8 | EP | LC | |||
| Order Acanthuriformes | ||||||
| Family Pomacanthidae | ||||||
| Holacanthus passer Valenciennes, 1846 | S2, S3, S4, S5, S8 | EP | LC | |||
| Pomacanthus zonipectus (Gill, 1862) | S4, S8 | EP | LC | |||
| Family Chaetodontidae | ||||||
| Chaetodon humeralis Günther, 1860 | S1, S2, S3, S4, S5, S7, S8 | S6 | EP | LC | ||
| Johnrandallia nigrirostris (Gill, 1862) | S3, S4, S5, S7, S8 | EP | LC | |||
| Order Tetraodontiformes | ||||||
| Family Diodontidae | ||||||
| Diodon holocanthus Linnaeus, 1758 | S2, S3, S4, S5, S7, S8 | S6 | CT | LC | ||
| Family Tetraodontidae | ||||||
| Arothron hispidus (Linnaeus, 1758) | S5, S8 | EP + IP | LC | |||
| Arothron meleagris (Anonymus, 1798) | S2, S5 | EP + IP | LC | |||
| Canthigaster punctatissima (Günther, 1870) | S2, S5, S8 | S6 | ETP | LC | ||
| Sphoeroides lobatus (Steindachner, 1870) | S2 | S6 | EP | LC | ||
| Family Balistidae | ||||||
| Balistes polylepis Steindachner, 1876 | S5, S7, S8 | S6 | EP | LC | ||
| Order Carangiformes | ||||||
| Family Carangidae | ||||||
| Caranx caballus Günther, 1868 | S2, S3, S8 | EP | LC | |||
| Order Perciformes | ||||||
| Family Serranidae | ||||||
| Alphestes immaculatus Breder, 1936 | S1, S2, S7 | EP | LC | |||
| Cephalopholis panamensis (Steindachner, 1876) | S2, S3, S4, S5, S7 | EP | LC | |||
| Epinephelus labriformis (Jenyns, 1840) | S1, S2, S3, S5, S7, S8 | EP | LC | |||
| Paranthias colonus (Valenciennes, 1846) | S8 | EP | LC | |||
| Serranus psittacinus Valenciennes, 1846 | S6 | EP | LC | |||
| Family Apogonidae | ||||||
| Apogon pacificus (Herre, 1935) | S2, S5, S7, S8 | EP | LC | |||
| Family Lutjanidae | ||||||
| Lutjanus argentiventris (Peters, 1869) | S1, S2, S3, S4, S5, S7, S8 | EP | LC | |||
| Family Haemulidae | ||||||
| Haemulon maculicauda (Gill, 1862) | S2 | EP | LC | |||
| Haemulon scudderii Gill, 1862 | S8 | EP | LC | |||
| Haemulon steindachneri (Jordan & Gilbert, 1882) | S3, S4 | S6 | EP + WA | LC | ||
| Family Pomacentridae | ||||||
| Abudefduf troschelii (Gill, 1862) | S2, S4, S5, S7, S8 | EP | LC | |||
| Chromis atrilobata Gill, 1862 | S3, S4, S5, S7, S8 | EP | LC | |||
| Microspathodon dorsalis (Gill, 1862) | S3, S4, S5, S8 | EP | LC | |||
| Stegastes acapulcoensis (Fowler, 1944) | S2, S3, S4 | EP | LC | |||
| Stegastes flavilatus (Gill, 1862) | S2, S4, S5, S7, S8 | S6 | EP | LC | ||
| Family Labridae | ||||||
| Bodianus diplotaenia (Gill, 1862) | S2, S3, S4, S5, S7, S8 | EP | LC | |||
| Halichoeres chierchiae Di Caporiacco, 1948 | S3, S5 | EP | LC | |||
| Halichoeres dispilus (Günther, 1864) | S3, S4, S7, S8 | S6 | EP | LC | ||
| Halichoeres nicholsi (Jordan & Gilbert, 1882) | S5, S7 | EP | LC | |||
| Thalassoma lucasanum (Gill, 1862) | S1, S3, S5, S7, S8 | EP | LC | |||
| Family Scorpaenidae | ||||||
| Scorpaena mystes Jordan & Starks, 1895 | S7 | EP | LC | |||
Three percent of species are restricted to the Eastern Tropical Pacific (Gulf of California to Peru), whereas 94% occur throughout the Eastern Pacific (California, USA to Chile). Only four species are found outside the Eastern Pacific, one of which is circumtropical (Diodon holocanthus), one is amphioceanic (Haemulon steindachneri) and the remaining two occur in the Indo-Pacific (Arothron hispidus, A. meleagris) (Table 3).
Classification by IUCN Red List status revealed that the fish observed in BSa and GSE are not species in threatened categories, only of least concern (Table 3).
We counted a total of 1 964 invertebrate individuals with the use of band transects (n = 21). The highest number of individuals was recorded in the rocky reef, 1 862 ind., while in Sargassum beds only 102 ind. In the rocky reef, Diadema mexicanum accounted for 81.3% of all individuals observed. D. mexicanum (100%) and Eucidaris thouarsii (93%) ranked highest in frequency of occurrence, and three species were observed between 40 to 50% of the transects (Toxopneustes roseus, Opeatostoma pseudodon and Phataria unifascialis). D. mexicanum has the highest mean density (5.04 ± 2.02 ind m-2) (Table 4). While in Sargassum beds, T. roseus accounted for 87.3% of all individuals observed and ranked highest in frequency of occurrence (83%) and in mean density (0.74 ± 0.75 ind m-2) (Table 4).
Table 4 List of invertebrates observed in rocky reefs and Sargassum beds in the North Pacific of Costa Rica organized by total abundance, relative abundance, frequency of occurrence, and mean density (± SD).
| Species | Abundance (# inds) | Relative abundance (%) | Frequency (%) | Mean Density (#ind m-2) | SD |
| ROCKY REEFS | |||||
| Diadema mexicanum | 1513 | 81.3 | 1.00 | 5.04 | 2.02 |
| Eucidaris thouarsii | 192 | 10.3 | 0.93 | 0.64 | 0.57 |
| Toxopneustes roseus | 73 | 3.9 | 0.47 | 0.24 | 0.47 |
| Opeatostoma pseudodon | 43 | 2.3 | 0.47 | 0.14 | 0.24 |
| Arbacia stellata | 14 | 0.8 | 0.13 | 0.05 | 0.14 |
| Phataria unifascialis | 9 | 0.5 | 0.40 | 0.03 | 0.05 |
| Hexaplex princeps | 7 | 0.4 | 0.27 | 0.02 | 0.05 |
| Hexaplex erythrostomus | 3 | 0.2 | 0.13 | 0.01 | 0.03 |
| Spondylus limbatus | 3 | 0.2 | 0.07 | 0.01 | 0.04 |
| Pinctada mazatlanica | 2 | 0.1 | 0.07 | <0.01 | 0.03 |
| Conus sp. | 1 | 0.1 | 0.07 | <0.01 | 0.01 |
| Elysia diomedea | 1 | 0.1 | 0.07 | <0.01 | 0.01 |
| Nidorellia armata | 1 | 0.1 | 0.07 | <0.01 | 0.01 |
| SARGASSUM BEDS | |||||
| Toxopneustes roseus | 89 | 87.3 | 0.83 | 0.74 | 0.75 |
| Diadema mexicanum | 4 | 3.9 | 0.50 | 0.03 | 0.04 |
| Hexaplex princeps | 3 | 2.9 | 0.33 | 0.03 | 0.04 |
| Astropyga pulvinata | 3 | 2.9 | 0.33 | 0.03 | 0.04 |
| Vasum caestus | 1 | 1.0 | 0.17 | <0.01 | 0.02 |
| Elysia diomedea | 1 | 1.0 | 0.17 | <0.01 | 0.02 |
| Tripneustes depressus | 1 | 1.0 | 0.17 | <0.01 | 0.02 |
We counted a total of 622 fish individuals with the use of band transects (n = 21). The highest number of individuals was recorded in the rocky reef (539 ind.), while only 83 ind. in Sargassum beds. In the rocky reef, three species (Chromis atrilobata, Halichoeres dispilus and Stegastes acapulcoensis) accounting for 69.8% of all individuals observed. S. acapulcoensis (60%), Bodianus diplotaenia (60%) and Chaetodon humeralis (53%) ranked highest in frequency of occurrence, and five species were observed on 33% of transects. According to their mean density (Table 5), C. atrilobata (0.34 ± 0.52 ind m-2), H. dispilus (0.09 ± 0.21 ind m-2), S. acapulcoensis (0.07 ± 0.10 ind m-2) and H. steindachneri (0.05 ± 0.18 ind m-2) ranked highest. While in Sargassum beds, H. dispilus accounted for 77.1% of all individuals observed and ranked highest in frequency of occurrence (83%) and in mean density (0.21 ± 0.18 ind m-2) (Table 5).
Table 5 List of conspicuous fishes observed in rocky reefs and Sargassum beds in the North Pacific of Costa Rica organized by total abundance, relative abundance, frequency of occurrence, and mean density (± SD).
| Species | Abundance (# inds) | Relative abundance (%) | Frequency (%) | Mean density (#ind m-2) | SD |
| ROCKY REEFS | |||||
| Chromis atrilobata | 255 | 47.3 | 0.33 | 0.34 | 0.52 |
| Halichoeres dispilus | 65 | 12.1 | 0.33 | 0.09 | 0.21 |
| Stegastes acapulcoensis | 56 | 10.4 | 0.60 | 0.07 | 0.10 |
| Haemulon steindachneri | 40 | 7.4 | 0.27 | 0.05 | 0.18 |
| Thalassoma lucasanum | 23 | 4.3 | 0.13 | 0.03 | 0.10 |
| Bodianus diplotaenia | 19 | 3.5 | 0.60 | 0.03 | 0.03 |
| Chaetodon humeralis | 14 | 2.6 | 0.53 | 0.02 | 0.02 |
| Diodon holocanthus | 13 | 2.4 | 0.33 | 0.02 | 0.04 |
| Johnrandallia nigrirostris | 10 | 1.9 | 0.27 | 0.01 | 0.03 |
| Epinephelus labriformis | 9 | 1.7 | 0.40 | 0.01 | 0.02 |
| Abudefduf troschelii | 8 | 1.5 | 0.33 | 0.01 | 0.02 |
| Microspathodon dorsalis | 8 | 1.5 | 0.33 | 0.01 | 0.02 |
| Holacanthus passer | 4 | 0.7 | 0.20 | <0.01 | 0.01 |
| Stegastes flavilatus | 4 | 0.7 | 0.13 | <0.01 | 0.02 |
| Lutjanus argentiventris | 3 | 0.6 | 0.20 | <0.01 | 0.01 |
| Cephalopholis panamensis | 2 | 0.4 | 0.13 | <0.01 | 0.01 |
| Halichoeres chierchiae | 2 | 0.4 | 0.13 | <0.01 | 0.01 |
| Muraena lentiginosa | 1 | 0.2 | 0.07 | <0.01 | 0.01 |
| Pomacanthus zonipectus | 1 | 0.2 | 0.07 | <0.01 | 0.01 |
| Caranx caballus | 1 | 02 | 0.07 | <0.01 | 0.01 |
| Alphestes immaculatus | 1 | 0.2 | 0.07 | <0.01 | 0.01 |
| SARGASSUM BEDS | |||||
| Halichoeres dispilus | 64 | 77.1 | 0.83 | 0.21 | 0.18 |
| Chaetodon humeralis | 4 | 4.8 | 0.33 | 0.01 | 0.02 |
| Haemulon steindachneri | 4 | 4.8 | 0.17 | 0.01 | 0.03 |
| Serranus psittacinus | 3 | 3.6 | 0.33 | 0.01 | 0.02 |
| Sphoeroides lobatus | 2 | 2.4 | 0.33 | <0.01 | 0.01 |
| Stegastes flavilatus | 2 | 2.4 | 0.17 | <0.01 | 0.02 |
| Urobatis halleri | 1 | 1.2 | 0.17 | <0.01 | 0.01 |
| Diodon holocanthus | 1 | 1.2 | 0.17 | <0.01 | 0.01 |
| Canthigaster punctatissima | 1 | 1.2 | 0.17 | <0.01 | 0.01 |
| Balistes polylepis | 1 | 1.2 | 0.17 | <0.01 | 0.01 |
Discussion
The North Pacific of Costa Rica is a region of great research interest due to its high biological diversity and ocean-atmosphere phenomena, and for its great national importance due to economic activities that take place there (Cortés, 2014; Cortés, 2016). The scientific information about species in marine ecosystems in an area is critical to understand, conserve and management the biodiversity in an integral way (Margules & Pressey, 2000). Although our records of invertebrates and fish species in BSa and GSE are typical of the biogeographic province of the Eastern Tropical Pacific (ETP) of Costa Rica (Alvarado, Chacón-Monge, Solís-Marín, Pineda-Enríquez, Caballero-Ochoa, Solano-Rivera & Romero-Chaves, 2017; Alvarado & Chiriboga, 2008; Alvarado & Fernández, 2005; Alvarado, Solís-Marín & Ahearn, 2010; Alvarado et al., 2015; Alvarado et al., 2018; Cordero-Umaña & Santidrián-Tomillo, 2020; Cortés, 2017; Dominici-Arosemena, Brugnoli-Olivera, Cortés-Núñez, Molina-Ureña & Quesada-Alpizar, 2006; Murase, Angulo, Miyazaki, Bussing & López, 2014;), the distribution geographic record of invertebrates and fishes observed in this study contribute to the knowledge of marine biodiversity in conservation priority areas (North Pacific of Costa Rica) and/or sites (marine sector of conservation areas) identified in the country (Alvarado et al., 2011; Cortés, 2012; Cortés, 2014; Cortés, 2017; SINAC-MINAET, 2008). Moreover, it contributes to the research efforts on the biodiversity of marine ecosystems of the Pacific shores of Costa Rica (Cortés, 2012; Cortés, 2016) that have received little (e.g., rocky reefs) (Cordero-Umaña & Santidrián-Tomillo, 2020; Dominici-Arosemena et al., 2006; Espinoza & Salas 2005) or scarce attention (e.g., Sargassum beds) compared with others, e.g., coral reefs (Alvarado & Chiriboga, 2008; Alvarado et al., 2015; Alvarado et al., 2018; Cortés, 1996 1997).
Our estimates of number of macroinvertebrates and conspicuous fish species would be larger with more spatial and temporal replication. Because estimates are sample-design dependent, comparisons to number of species at other sites where different sampling designs were used should be considered only suggestive. At the rocky reefs, for mollusks (gastropods and bivalves) and echinoderms (sea stars, sea urchins, sea cucumber and brittle stars) we recorded a lower number of species (23) than reported in rocky reefs in the Gulf of Papagayo (46 species) (Cordero-Umaña, & Santidrián-Tomillo, 2020) and in coral reefs (35 to 37 species) in the ETP of Costa Rica (Alvarado et al., 2015; Alvarado et al., 2018). On the other hand, the number of fish species (33) is less than reported previously (46 to 81 species) by Cordero-Umaña & Santidrián-Tomillo (2020), Dominici-Arosemena et al. (2006) and Espinoza & Salas (2005) in rocky reefs in the North Pacific of Costa Rica. Our lower number of species maybe due to our lower sampling compared to previous studies, our results suggest that the number of macroinvertebrates and fish species in rocky reef could be higher at BSa and GSE.
In the case of Sargassum beds, our study is the first biodiversity report for this marine ecosystem in the ETP of Costa Rica. With eight macroinvertebrates (mollusks and echinoderms groups) and ten fish species, the number of species recorded are lower than reported in Sargassum beds for these faunistic groups (44 to 73 species) from others latitudes, like the Gulf of California in México (Foster, McConnico, Lundsten, Wadsworth, Kimball, Brooks et al., 2007; Suárez-Castillo, 2008; Suárez-Castillo, 2014; Suárez-Castillo, Riosmena-Rodríguez, Hernández-Carmona, Méndez-Trejo, López-Vivas, Sánchez-Ortiz et al., 2013). Our results suggest that the number of invertebrates and fish species could be higher in GSE, this too possibly due lower sampling effort than used in other studies, and because we visit the Sargassum beds in February, before the algae reached their maximum length (102.0 cm in April) (Cortés et al., 2014). The mean length of S. liebmanni thallus observed was 7.67 cm (Alvin Suárez, personal observation). Sargassum species are considered foundation species because they harbor a vast diversity of species because of the habitat produced by the algae itself (Foster et al., 2007; Suárez-Castillo, 2008; Suárez-Castillo, 2014).
The variation of nearshore flora and fauna is often attributed to habitat structure and seasonal changes in environmental conditions (Aburto-Oropeza & Balart, 2001; Beukhof, Dencker, Pecuchet & Lindegren, 2019; Dominici-Arosemena & Wolff, 2006; Foster et al., 2007; McCourt, 1985; Palacios & Zapata, 2014; Scrosati, 2001). However, without physical descriptors of marine ecosystems and temporal replication and more quantitative, seasonal records of species composition in these marine ecosystems from this study, the consistency of these temporal patterns and their causes are largely unknow. The communities of invertebrate and fish of both marine ecosystems have different species composition. At the rocky reefs, the black sea urchin, D. mexicanum, and slate pencil urchin, E. thouarssii, were the most abundant invertebrates (1 513 and 192 ind., respectively) and frequently observed (100% and 93%, respectively). This result suggest that our estimate is reasonable for this marine ecosystem, where it has been recognized that these two species are the most dominant grazers in the ETP (Guzmán & Cortés, 1993). It has been reported that these two echinoids exert a strong influence on the community structure (Andrew, 1989; Glynn, Wellington & Birkeland, 1979; Lawrence, 1975; Underwood, 1992), so the values of mean density of D. mexicanum (5.04 ind m-2) and E. thouarsii (0.57 ind m-2) observed in this study are important to consider the periodical monitoring of community structure in rocky reefs in BSa and GSE. The mean density value recorded for D. mexicanum in this study are higher than was has been previously recorded in the northern Pacific region of Costa Rica (0.20 to 2.19 ind m-2) (Alvarado, Cortés & Reyes-Bonilla, 2012). The fish species composition observed in this study, high abundance of C. atrilobata (255 inds.) and serranids, pomacentrids, labrids and tetraodontids fishes with highest number of fish species, is similar to recorded for rocky reef in the North Pacific of Costa Rica (Cordero-Umaña & Santidrián-Tomillo, 2020; Dominici-Arosemena et al., 2006; Espinoza & Salas, 2005), indicating that the estimate for our survey sites is reasonable for this type of community.
Comparisons of communities of invertebrate and fish species in Sargassum beds from this study with the observed in other sites in the northern Pacific region of Costa Rica can’t be done, because our work represent the first report of this type for this marine ecosystem in the region. The composition of invertebrates and fish species recorded are similar to observed in Sargassum beds in the Gulf of California (Foster et al., 2007; Suárez-Castillo, 2008; Suárez-Castillo, 2014; Suárez-Castillo et al., 2013) suggesting that our observations are typical for this marine ecosystem. However, it was possible to observe invertebrate species (Vasum caestus, Astropyga pulvinata) that have not been previously recorded in Sargassum beds, and differences in the invertebrate and fish species frequently observed and with highest mean densities compared with Sargassum beds in other sites (Suárez-Castillo, 2008; Suárez-Castillo et al., 2013). Therefore, taxonomic and community structure studies of the fauna in Sargassum beds in the North Pacific of Costa Rica are necessary.
The original approach of this research by one of the authors (A. Suárez) was to monitor the community of invertebrate and fish species in Sargassum beds in the northern Pacific region of Costa Rica, where has been reported the absence of S. liebmannni since 2013, following a population outbreak of the black sea urchin D. mexicanum (Cortés et al., 2014). The substantial decrease or total disappearance of these seaweed beds by multiple factors (seasonal patterns related to temperature changes, introduction of alien species, biotic interactions, among others) has been reported in many coastal areas around the world (Aburto-Oropeza, Sala, Paredes, Mendoza & Ballesteros, 2007; Britton-Simmons, 2004; Haraguchi & Sekida, 2008; Rivera & Scrosati, 2006; Viejo, 1997; Xuan-Vy & Huu-Dai, 2011). The losses of these foundation species and the shift from algal dominated substrate to bare substrate, as observed on rocky reefs in our survey sites in BSa and GSE (Alvin Suárez, personal observation), may have implications in the decline of marine biodiversity associated with them. Therefore, more studies of marine biodiversity in Sargassum beds in the North Pacific of Costa Rica are necessary.
Our study further evidences the poorly and scarce knowledge of communities of invertebrates and fish species in rocky reefs and Sargassum beds in the North Pacific of Costa Rica, respectively. Which require considerably more survey and field work to locate such marine ecosystems, better characterize their marine biodiversity that occur in them and better understand of the community structure of fauna and the factors that determine them. Information that is required to support management decisions that are backed by sound scientific knowledge. These marine ecosystems deserve special consideration in efforts to conserve biodiversity.
Ethical statement: authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.












 uBio
uBio 


